 |
 |
 |
| |
HCV Treatment 2014 and Beyond - New Era of Hepatitis C Management
|
| |
| |
from Jules: in his talk Dr Kim reviews new oral HCV therapies & a variety of current issues including study results and cirrhotic and treatment-experienced patients.
IDSA: HCV Treatment Cascade: Is Eradication Possible? - Shruti H. Mehta, PhD MPH (10/10/14)
Reported by Jules Levin
IDSA, Philadelphia, PA
October 9, 2014
Arthur Y. Kim, MD
Massachusetts General Hospital
Harvard Medical School



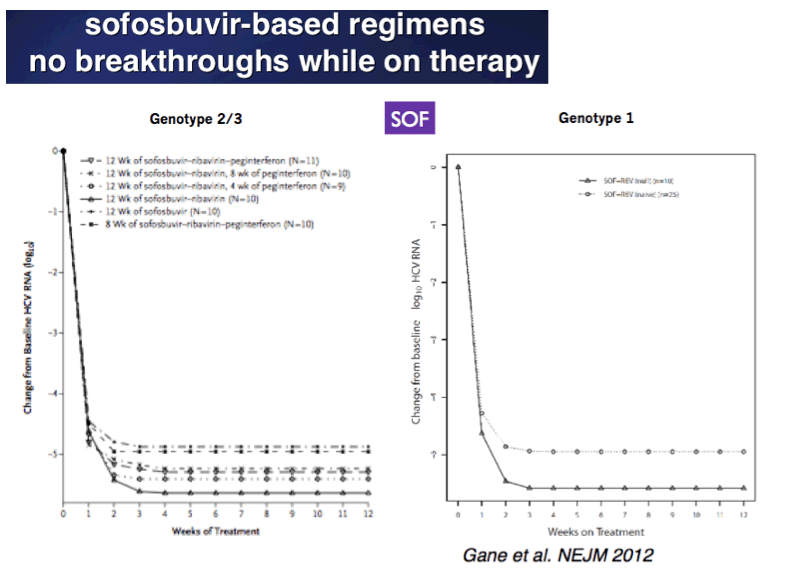
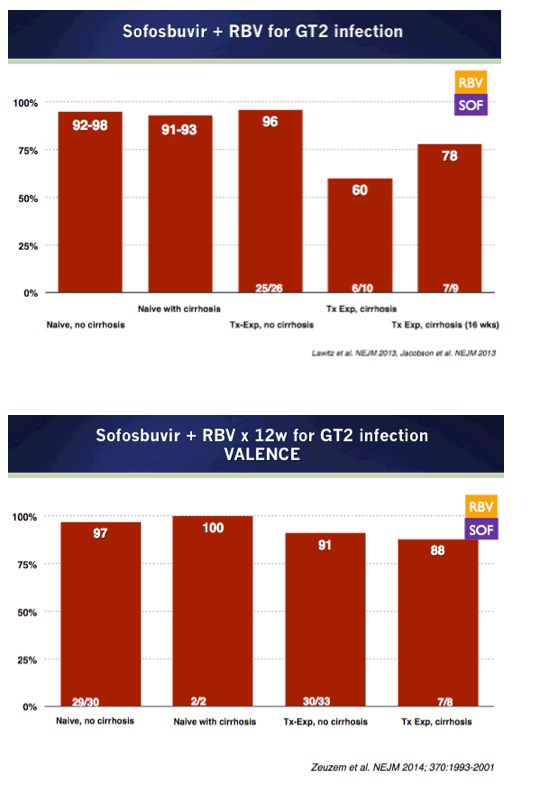
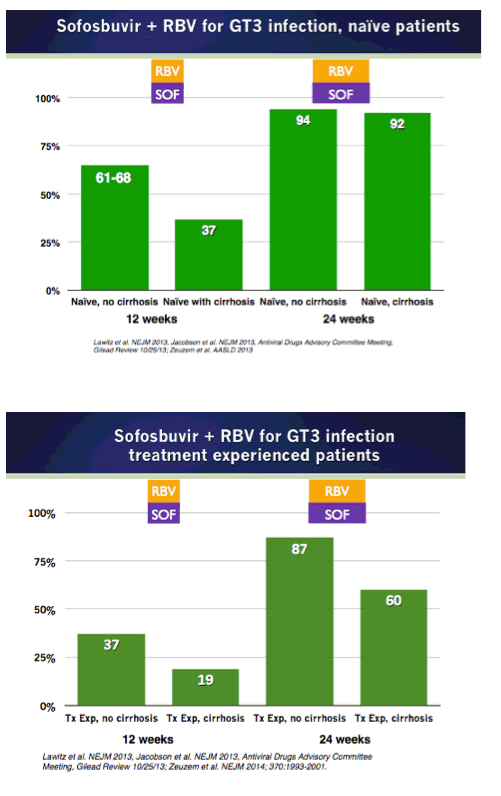
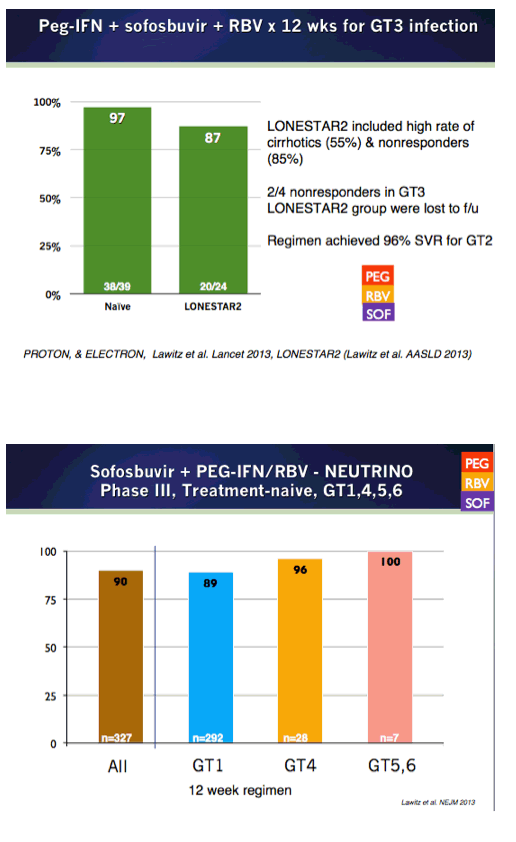
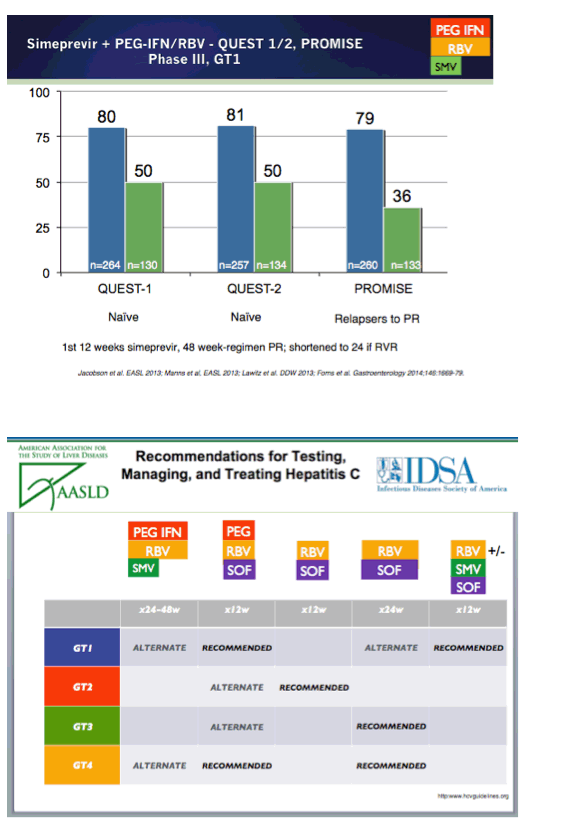
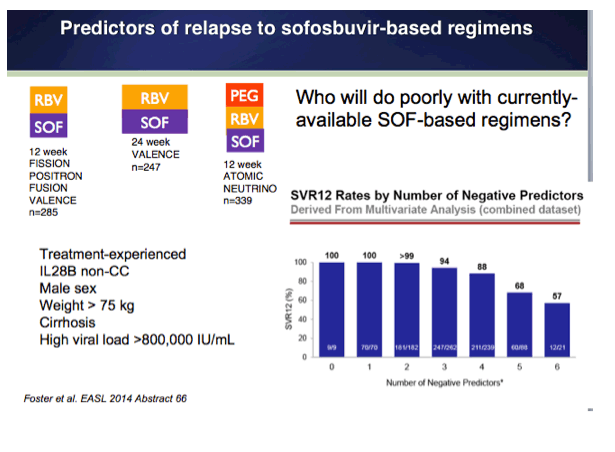
EASL/2014: Sofosbuvir-Based Regimens Are Associated With High SVR Rates Across Genotypes and Among Patients With Multiple Negative Predictive Factors - (04/14/14)
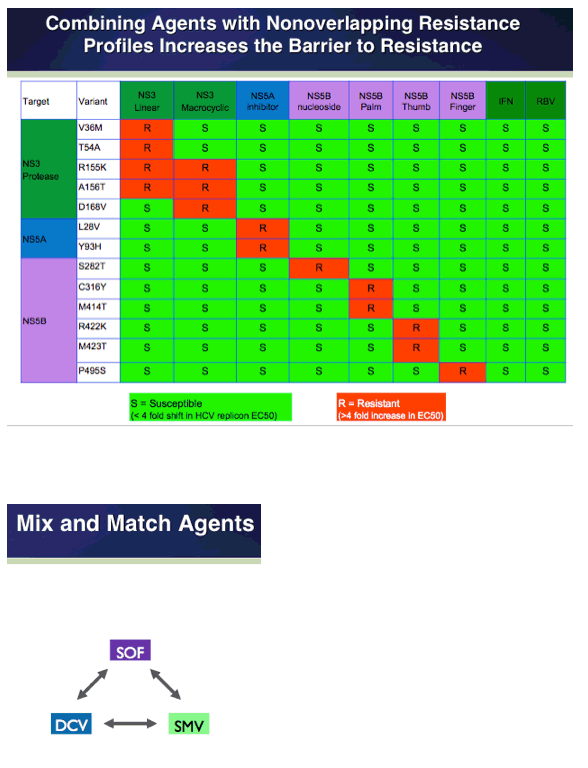
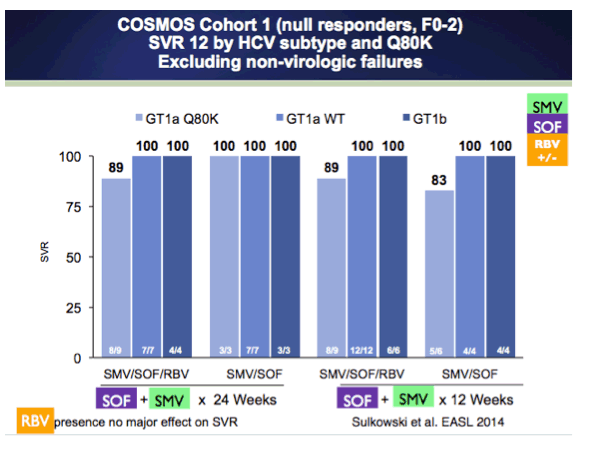
COSMOS - Simeprevir+Sofosbuvir
EASL: Once-daily simeprevir (TMC435) plus sofosbuvir (GS-7977) with or without ribavirin in HCV genotype-1 prior null responders with METAVIR F0-2: COSMOS study subgroup analysis - (04/14/14)
EASL: Simeprevir plus sofosbuvir with/without ribavirin in HCV genotype-1 prior null-responder / treatment-naïve patients (COSMOS study): primary endpoint (SVR12) results in patients with METAVIR F3-4 (Cohort 2) - (04/14/14)
Janssen Initiates Phase 3 OPTIMIST Trials of Once-Daily Simeprevir in Combination with Once-Daily Sofosbuvir for the Treatment of Genotype 1 Chronic Hepatitis C - (04/03/14)
Virology analyses of simeprevir in Phase 2b and 3 studies - (04/17/14)
Deep sequencing analyses of minority baseline polymorphisms and persistence of emerging mutations in HCV genotype 1-infected patients treated with simeprevir - (04/17/14)
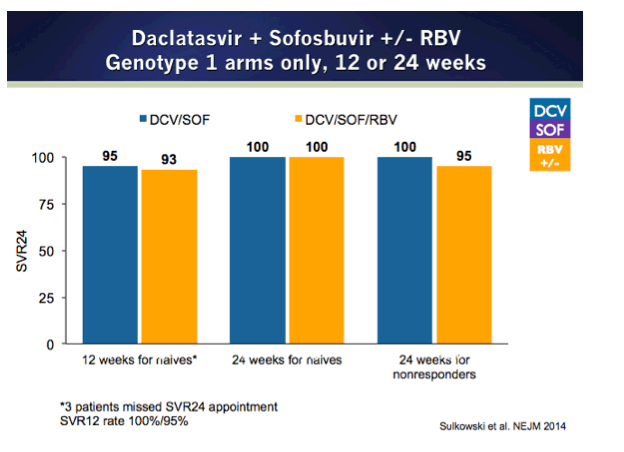
NEJM - Daclatasvir plus Sofosbuvir for Previously Treated or Untreated Chronic HCV Infection 12, 24 weeks GT1/2/3, FDA Approvals Oct 10 thru Dec This Fall - (10/02/14)
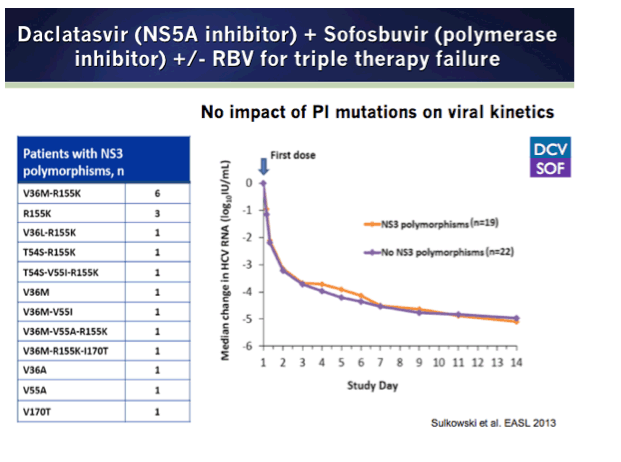
Phase 3 started early 2014
Phase III HIV/HCV Co-Infection Daclatasvir (DCV)+ Sofosbuvir (SOF).......in Treatment-naïve and Treatment-experienced Chronic Hepatitis C (Genotype 1, 2, 3, 4, 5, or 6)
Phase III Daclatasvir + Sofosbuvir in Cirrhotic Subjects and Subjects Post-liver Transplant..
Phase III Daclatasvir and Sofosbuvir for Genotype 3 Chronic HCV
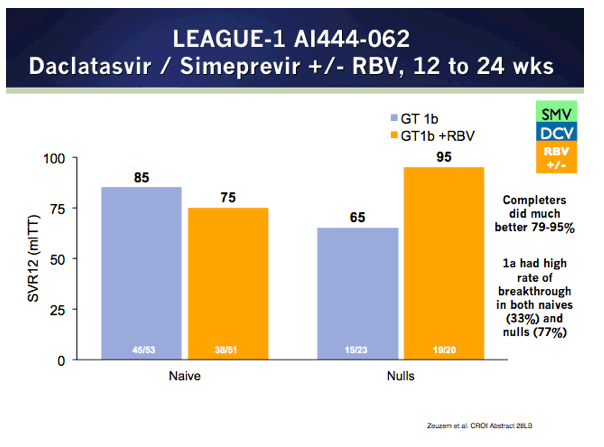
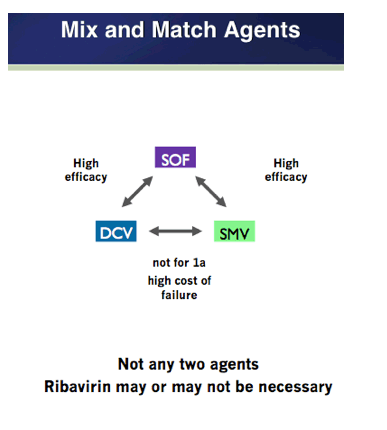
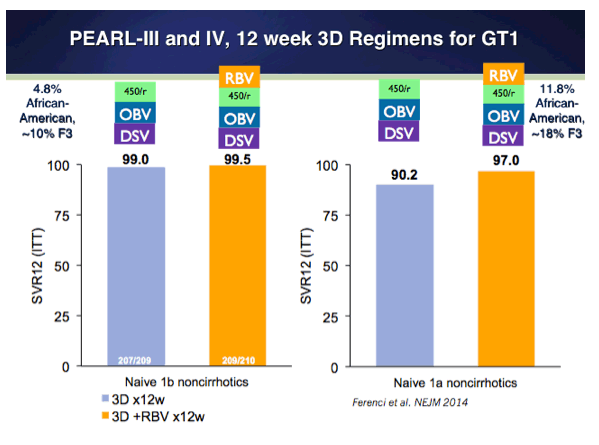
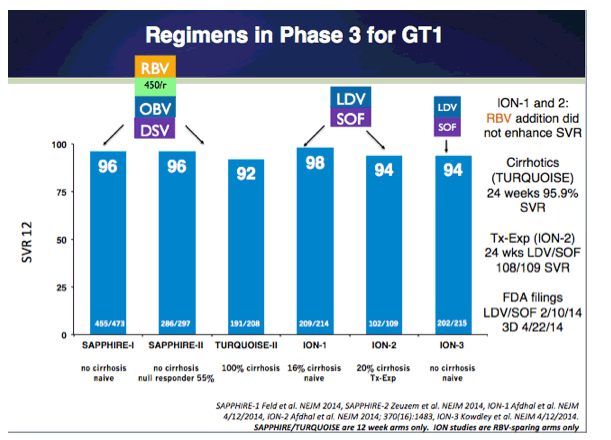
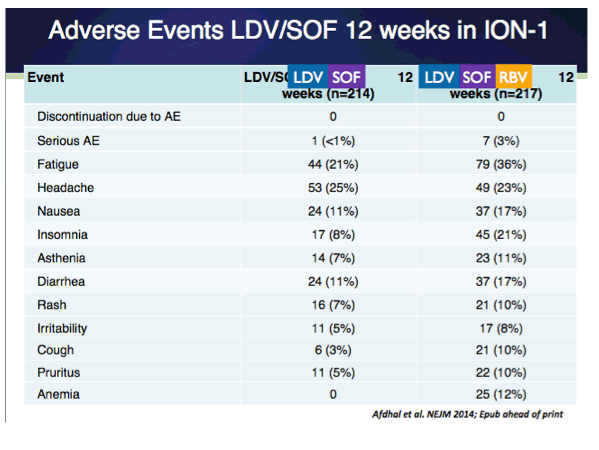
EASL: All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir With Or Without Ribavirin for 12 or 24 Weeks in Treatment-Naive Genotype 1 HCV-Infected Patients: the Phase 3 ION-1 Study - (04/14/14)
EASL: All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir With or Without Ribavirin for 12 or 24 Weeks in Treatment-Experienced Genotype 1 HCV-Infected Patients: The Phase 3 ION-2 Study - (04/14/14)
EASL: Ledipasvir/Sofosbuvir With and Without Ribavirin for 8 Weeks Compared to Ledipasvir/Sofosbuvir for 12 Weeks in Treatment-Naïve Noncirrhotic Genotype-1 HCV-Infected Patients: The Phase 3 ION-3 Study - (04/11/14)
NEJM Publications:
ION-1 Ledipasvir and Sofosbuvir for Untreated HCV Genotype 1 Infection - (04/14/14)
ION-2 Ledipasvir and Sofosbuvir for Previously Treated HCV Genotype 1 Infection - (04/14/14)
ION-3 Ledipasvir and Sofosbuvir for 8 or 12 Weeks for Chronic HCV without Cirrhosis - (04/11/14)
The Prevalence of HCV NS5A, Nucleoside and Protease Inhibitor Resistance Associated Variants and the Effects on Treatment with Ledipasvir/Sofosbuvir ± RBV in the Phase 3 ION Studies......"lower SVR rates were not observed in treatment-naive subjects or with longer 24 week LDV/SOF treatment"
http://www.natap.org/2014/ResisWksp/ResisWksp_07.htm
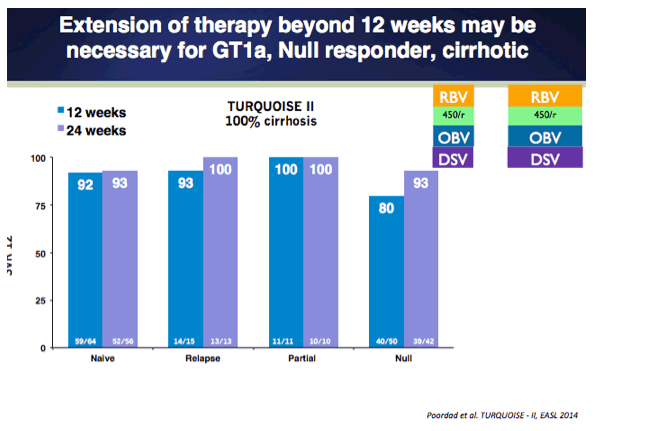
EASL: TURQUOISE-II: SVR12 RATE OF 92-96% IN 380 HEPATITIS C VIRUS GENOTYPE 1-INFECTED ADULTS WITH COMPENSATED CIRRHOSIS TREATED WITH ABT-450/r/ABT-267 AND ABT-333 PLUS RIBAVIRIN - (04/14/14)
NEJM - TURQUOISE-II ABT-450/r-Ombitasvir and Dasabuvir with Ribavirin for Hepatitis C with Cirrhosis - Phase 3 TURQUOISE-II - (04/14/14)
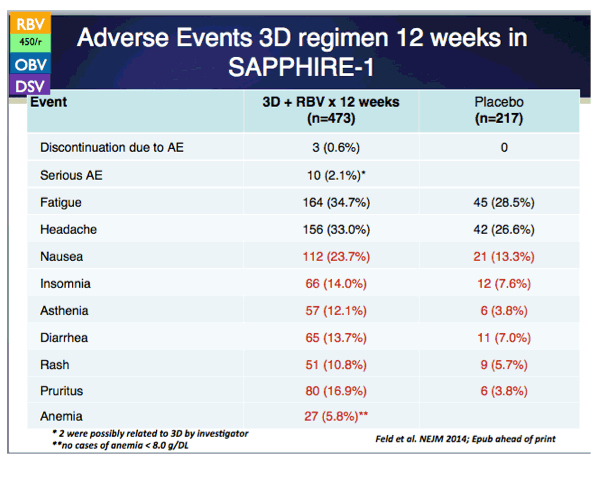
EASL: SAPPHIRE-I: PHASE 3 PLACEBO-CONTROLLED STUDY OF INTERFERON-FREE, 12-WEEK REGIMEN OF ABT-450/r/ABT-267, ABT-333, AND RIBAVIRIN IN 631 TREATMENT-NAïVE ADULTS WITH HEPATITIS C VIRUS GENOTYPE 1 - (04/11/14)
EASL: SAPPHIRE-II: PHASE 3 PLACEBO-CONTROLLED STUDY OF INTERFERON-FREE, 12-WEEK REGIMEN OF ABT-450/r/ABT-267, ABT-333, AND RIBAVIRIN IN 394 TREATMENT-EXPERIENCED ADULTS WITH HEPATITIS C VIRUS GENOTYPE 1 - (04/10/14)
NEJM Publications:
SAPPHIRE-I Treatment of HCV with ABT-450/r-Ombitasvir and Dasabuvir with Ribavirin (in previously untreated patients with HCV genotype 1 infection and no cirrhosis) - (04/11/14)
SAPPHIRE-II Retreatment of HCV with ABT-450/r-Ombitasvir and Dasabuvir with Ribavirin - (04/10/14) NEJM
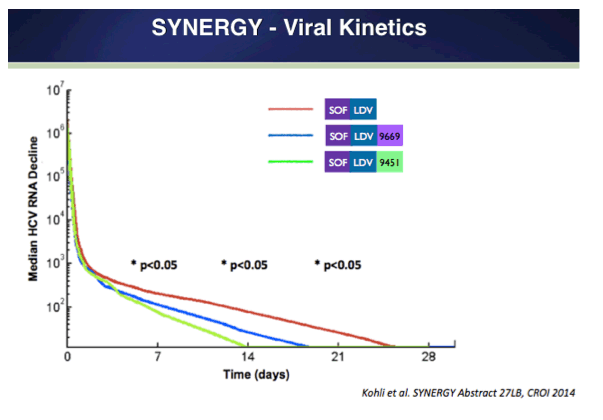
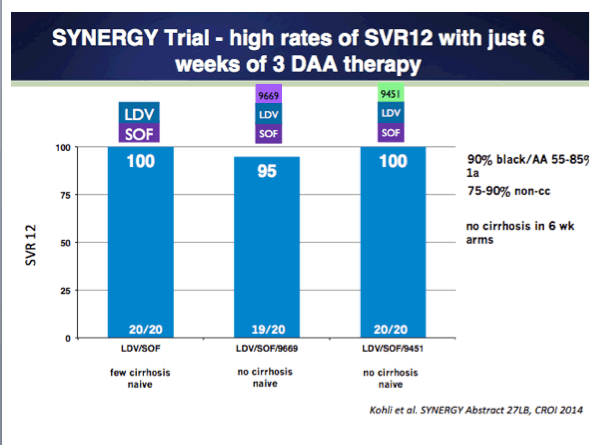
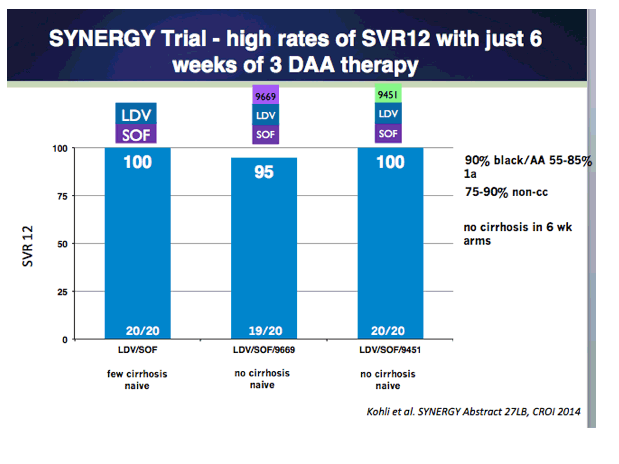
Combination Oral, Ribavirin Free, Antiviral Therapy to Optimize Treatment Outcomes for Hepatitis C Treatment Naïve Patients: Interim Results from the NIAID SYNERGY Trial (6 & 12 weeks therapy)......http://www.natap.org/2013/AASLD/AASLD_30.htm

|
| |
|
 |
 |
|
|