 |
 |
 |
| |
Estimated Time to Protection and Duration of Protection With Daily TDF/FTC PrEP
|
| |
| |
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, May 19-21, 2014, Washington, DC
Mark Mascolini
Authors comment: We think the important conclusion is 7 days of dosing appears sufficient to reach therapeutic drug concentrations for MSM.
We think that the "duration of action" (ie drug concentrations lingering after dosing stops) may be more difficult to interpret because people should not stop dosing while they may be being exposed to HIV.
Tenofovir/emtricitabine (TDF/FTC) preexposure prophylaxis (PrEP) may fully protect a person from HIV after 7 days of dosing, according to an analysis of 21 healthy men and women meeting a protective threshold calculated from iPrEx trial data [1]. Protection from HIV could last up to a week after TDF/FTC stops, University of Colorado researchers and colleagues estimated.
Several trials now show that daily TDF/FTC substantially lowers the risk of HIV acquisition in people who take this two-in-one pill faithfully. But as recently released CDC PrEP guidelines indicate, time to full protection with TDF/FTC PrEP and duration of protection after stopping the drugs remain unknown.
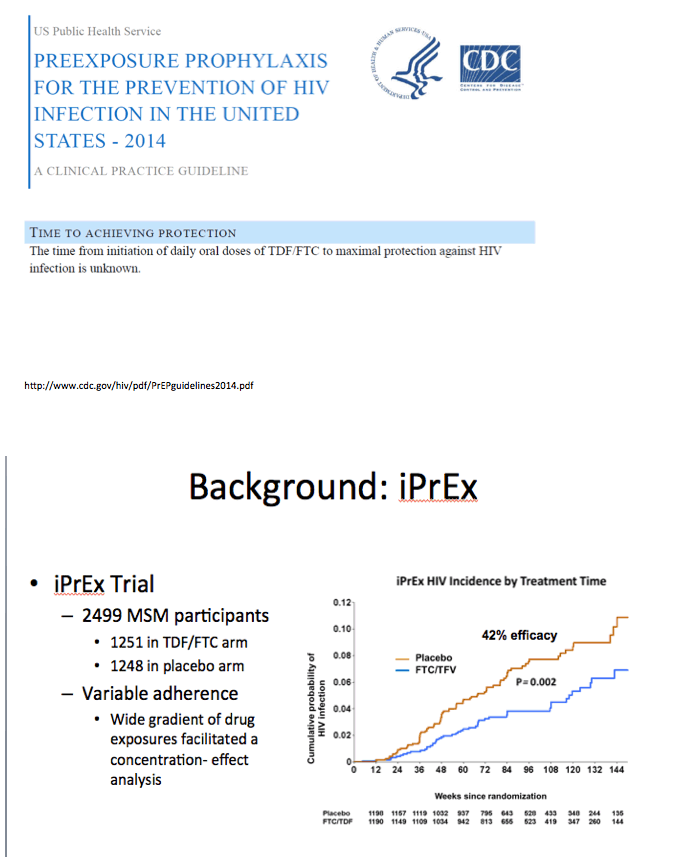
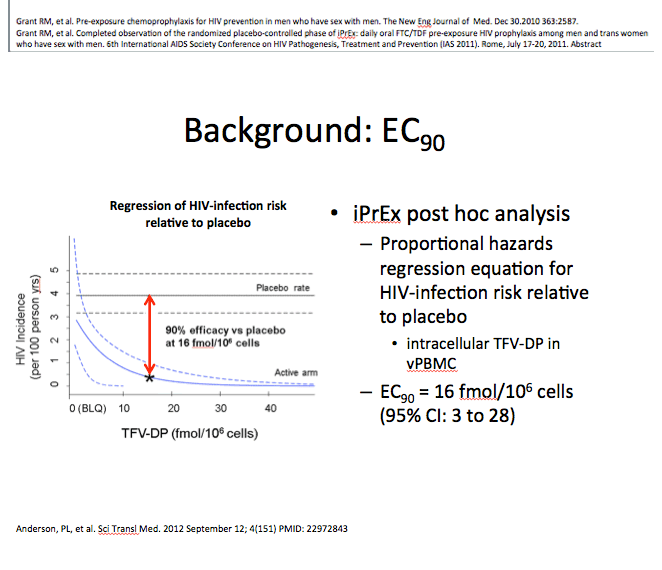
Enrolling men who have sex with men (MSM) and transgender women, iPrEx was the first placebo-controlled trial to establish the protective potential of daily oral TDF/FTC in high-risk HIV-negative people [2]. Further work by iPrEx investigators yielded an estimate that a tenofovir-diphosphate level of 16 fmol per million peripheral blood mononuclear cells (PBMCs) lowered HIV acquisition risk 90% compared with placebo, thus establishing a 90% effective concentration (EC90) [3]. (Tenofovir-diphosphate is the active metabolite of tenofovir.) The new study aimed to determine the number of days of TDF/FTC needed to reach the EC90 and how long tenofovir-diphosphate levels remain above the EC90 after dosing stopped.
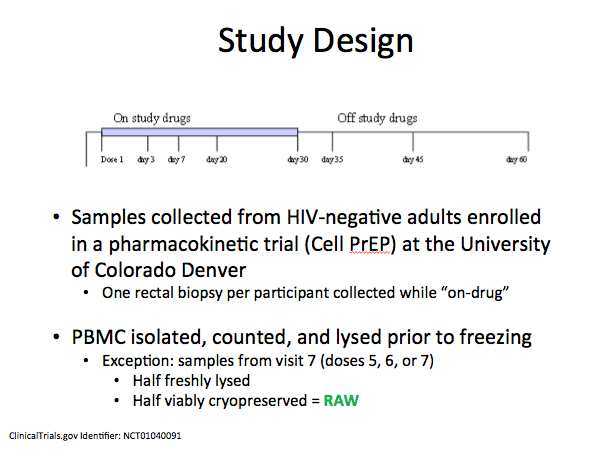
The analysis involved 21 HIV-negative people--11 men and 10 women--who took daily TDF/FTC for 30 days then remained in follow-up for another 30 days after stopping the drug. Researchers collected PBMCs at the first dose; on treatment days 3, 7, 20, and 30; and 5, 15, and 30 days after dosing stopped. Rectal biopsies collected once during an on-drug visit yielded mononuclear cells. After each daily dose of TDF/FTC, the researchers calculated the proportion of people reaching the iPrEx EC90. They also calculated inferred HIV risk reduction using the regression model developed earlier [3]. Finally, they determined a pharmacokinetic concentration-time profile in rectal tissue.
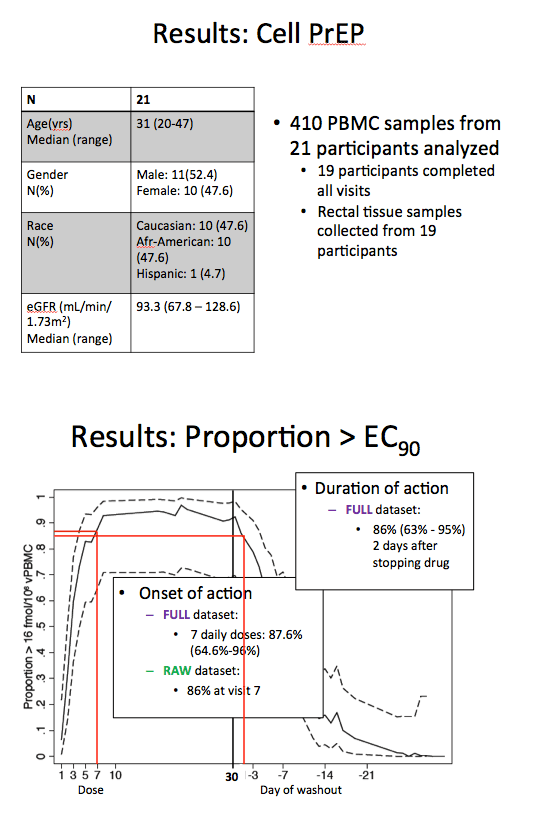
Median age of the 11 men and 10 women stood at 31 years and median weight at 81.1 kg. Ten participants were Caucasian, 10 were African American, and 1 was Hispanic. Everyone had healthy kidney function (eGFR range 67.8 to 128.6 mL/min). Nineteen people completed all study visits and had rectal biopsies. Tenofovir-diphosphate concentrations did not differ between men and women after the first dose or after steady-levels were reached.
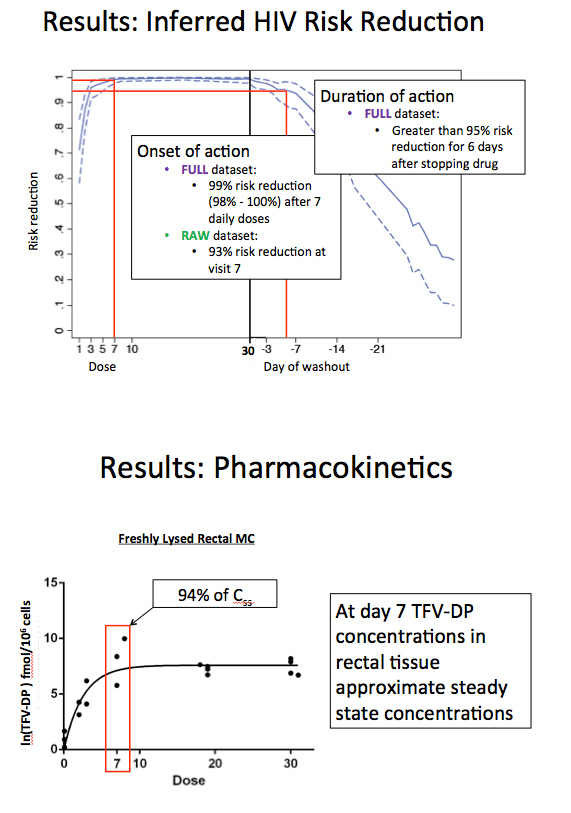
In the full dataset, after 7 daily doses of TDF/FTC 87.6% (95% confidence interval [CI] 64.6 to 96) reached the previously determined EC90. Nearly the same proportion--86% (95% CI 63 to 95)--remained above the EC90 for 2 days after stopping TDF/FTC.
Inferred risk reduction with the full dataset was 99% (95% CI 98 to 100) after 7 daily doses of TDF/FTC. Inferred risk reduction remained above 95% for 6 days after people stopped taking the drugs. Pharmacokinetic analysis indicated that concentrations of tenofovir-diphosphate in rectal tissue approximated steady-state concentrations.
Together these findings suggested to the researchers that nearly everyone taking TDF/FTC PrEP will attain protective concentrations of tenofovir after 7 days of dosing and that concentrations will remain protective about a week after a person stops taking TDF/FTC. But the University of Colorado researchers who conducted this study cautioned that the EC90 rests solely on data from MSM and transgender women enrolled in iPrEx. Jonathan Schapiro, a member of the Pharmacology Workshop organizing committee, noted that factors unmeasured in this analysis could contribute to protection and counseled against telling PrEP users that the 7 days to protection and 6-day duration are absolute values that apply to everyone.
References
1. Seifert S, Glidden DV, Meditz AL, et al. Estimated onset and duration of PrEP activity for daily TDF/FTC using the EC90 from iPrEx. 15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. May 19-21, 2014. Washington, DC. Abstract O_10.
2. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587-2599. http://www.nejm.org/doi/full/10.1056/NEJMoa1011205
3. Anderson PL, Glidden DV, Liu A, et al. Emtricitabine-tenofovir concentrations and pre-exposure prophylaxis efficacy in men who have sex with men. Sci Transl Med. 2012;4:151ra125. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3721979/

Program abstract:
Estimated onset and duration of PrEP activity for daily TDF/FTC using the EC90 from iPrEx
Background: Critical questions for the field of PrEP include the number of daily TDF/FTC doses required to establish protection from HIV acquisition (onset of action) and the number of days after stopping TDF/FTC that protection is maintained (duration of action). A substudy of the iPrEx trial estimated that 16 fmol/million cells of tenofovir-diphosphate (TFV-DP) in viably cryopreserved PBMC (vPBMC) was associated with a 90% reduction in HIV acquisition risk relative to placebo (EC90) in men who have sex with men (MSM). The aim of this study was to estimate the number of daily TDF/FTC doses required to reach the EC90 and how long the concentration remained above the EC90 after drug discontinuation from steady-state conditions.
Materials & Methods: HIV-negative adults were enrolled in an intensive pharmacokinetic study of daily TDF/FTC for 30 days. PBMC were collected at first dose, days 3, 7, 20, and 30, and off-drug on days 5, 15, and 30 of washout. Rectal biopsies were performed once during an on-drug visit and mononuclear cells were isolated, counted, and lysed. Paired vPBMC and freshly lysed PBMC were collected once per participant. vPBMC were harvested using the same procedures as in iPrEx and TFV-DP/FTC-TP were quantified in all samples with validated LC-MS/MS. The conversion between TFV-DP in vPBMC versus freshly lysed PBMC was determined using multiple imputation based on paired samples from the current and past studies. With each daily dose, the proportion of individuals achieving the iPrEx EC90 was assessed and the inferred HIV risk reduction was calculated with the exponential regression model from iPrEx [PMID: 22972843] using viable-corrected PBMC values.
Results: Twenty-one participants were included, 11 male, 10 female, median age: 31 years, weight: 81.1kg, race: 10 Caucasian, 10 African American, 1 Hispanic. Nineteen subjects completed all visits. No differences were observed between males and females in TFV-DP concentrations after the first dose or at steady state (p ≥ 0.29). After 8 doses, 93% (95% CI 71% - 98%) of participants reached the iPrEx EC90. At this time, the ln(TFV-DP) concentration was 96% of steady state in rectal mononuclear cells (1438 fmol/million cells). Greater than 90% of participants remained above the iPrEx EC90 until two days after stopping drug, at which time the percentage fell to 86% (63% - 95%). The inferred HIV risk reduction was 95% (92% - 98%) after 3 doses, 99% (98% - 100%) after 8 doses, and 98% (96% - 100%) two days after stopping drug. The inferred HIV risk reduction remained greater than 95% for 6 days after stopping drug from steady-state conditions.
Conclusions: These data indicate that high PrEP efficacy and high rectal tissue concentrations can be achieved in MSM after approximately one week of daily dosing. They also suggest that following daily dosing to steady state in MSM, a high level of protection may persist for several days after the last dose taken.



|
| |
|
 |
 |
|
|