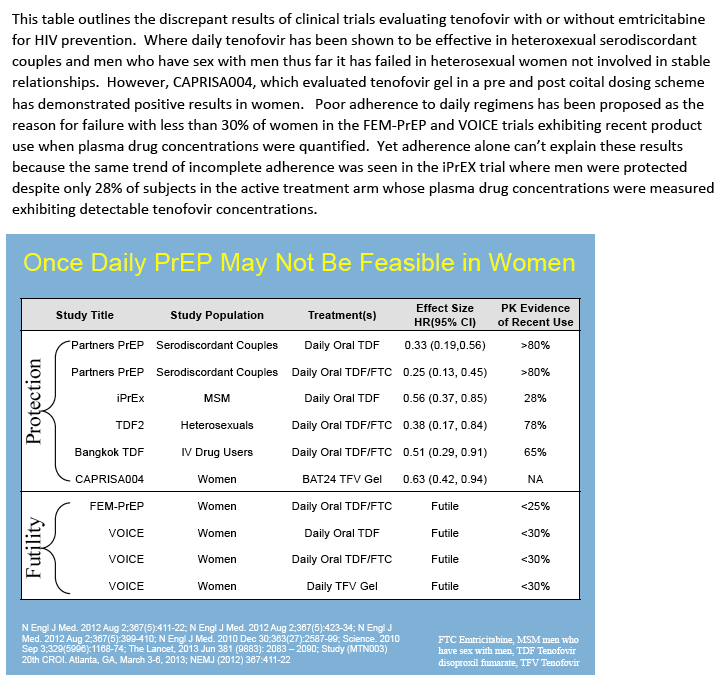
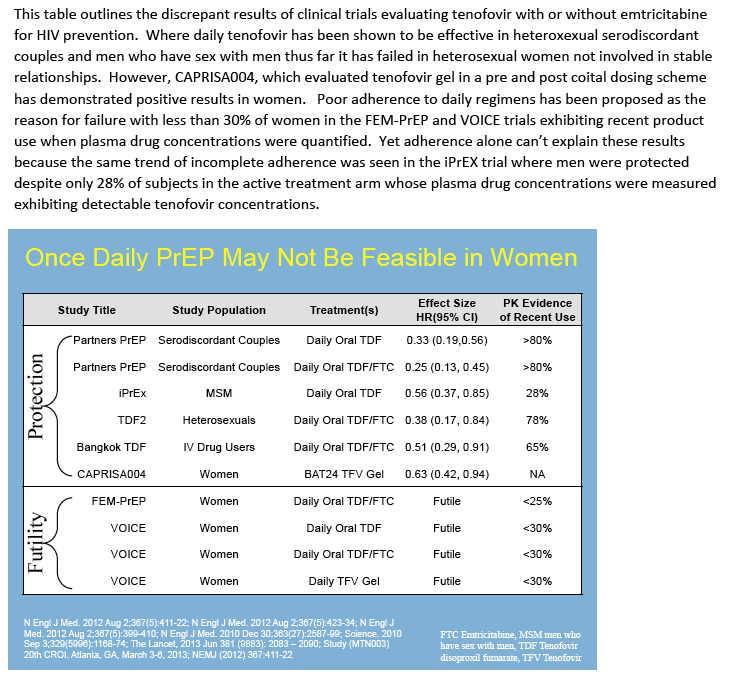
 |
 |
 |
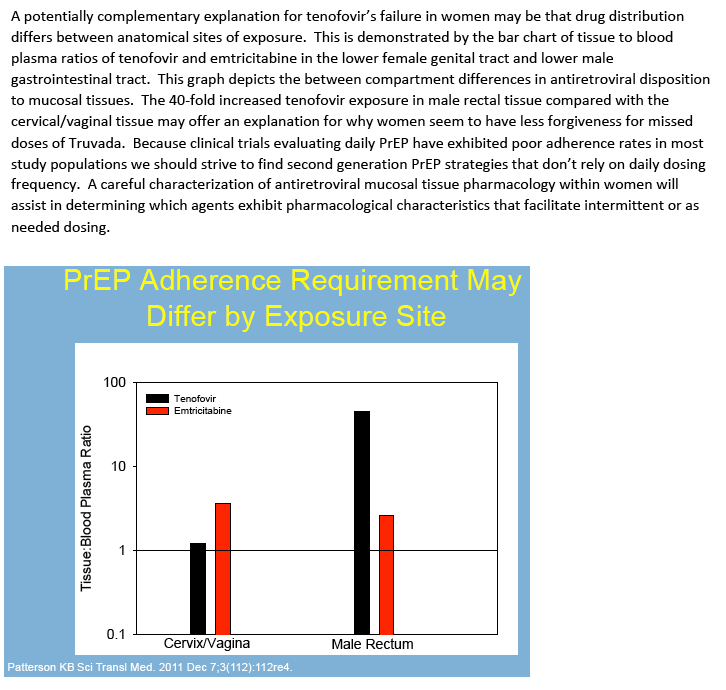
| |
Mucosal Tissue Pharmacokinetics of Maraviroc and Raltegravir in Women: Implications for Chemoprophylaxis [PrEP, Mucosal Tissue Compartments PK]
|
| |
| |
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis
Therapy, 19- 21 May 2014, Washington, DC
Mackenzie L. Cottrell, Heather M.A. Prince, Craig Sykes,
Nicole White, Stephanie Malone, Evan S. Dellon,
Ryan D. Madanick, Nicholas J. Shaheen, Michael G. Hudgens,
Jacob Wulff, Kristine B. Patterson, Angela D.M. Kashuba
University of North Carolina
UNC Eshelman School of Pharmacy and School of Medicine

Mucosal tissue pharmacokinetics of maraviroc and raltegravir in women: implications for chemoprophylaxis.
M.L. Cottrell1, H.M.A. Prince1, C. Sykes1, N. White1, S. Malone1, E.S. Dellon2, R.D. Madanick2, N.J. Shaheen2, M.G. Hudgens3, J. Wulff3, K.B. Patterson2, A.D.M. Kashuba1
1University of North Carolina, UNC Eshelman School of Pharmacy, Chapel Hill, USA; 2University of North Carolina, School of Medicine, Chapel Hill, USA; 3University of North Carolina, Gillings School of Global Public Health, Chapel Hill, USA
Background: The poor adherence rates demonstrated by heterosexual women in recent PrEP trials necessitate second-generation PrEP strategies that allow for intermittent or as-needed dosing. PK/PD modeling will inform the design of these dosing strategies, but better characterization of antiretroviral exposure in mucosal tissues is needed. This investigation was performed to examine the single dose pharmacokinetic profile and dose proportionality of maraviroc(MVC) and raltegravir(RTG) as potential PrEP agents.
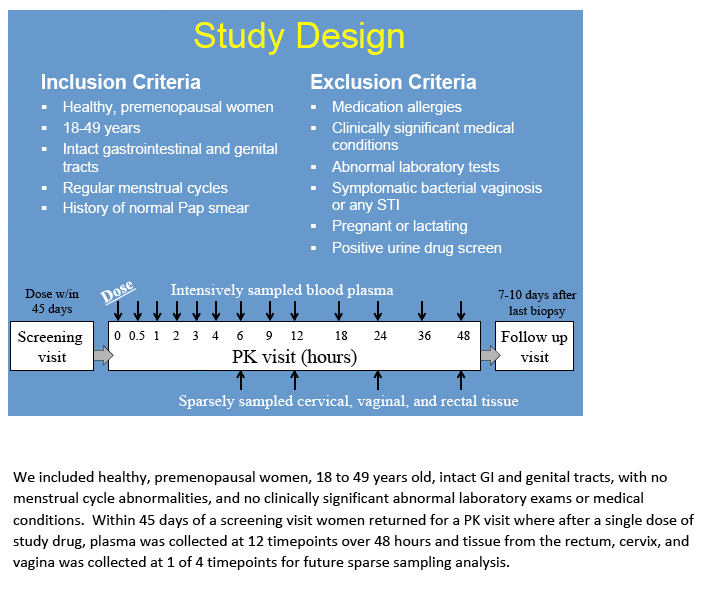
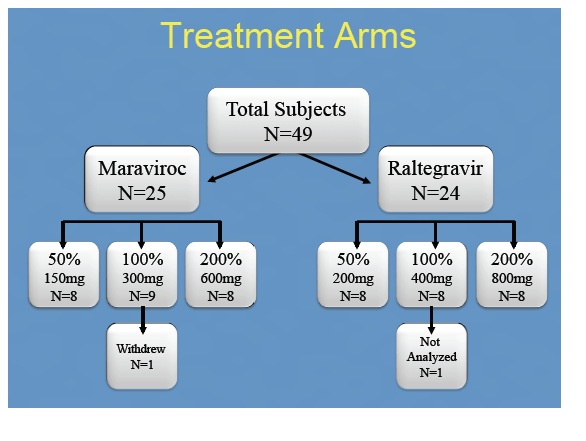
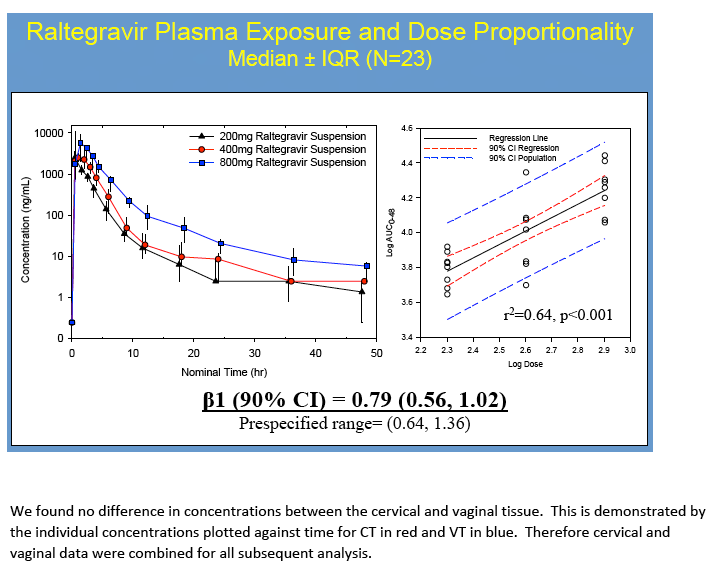
Materials & Methods: 48 healthy women were given single oral doses of MVC or RTG at 50%, 100% or 200% of the licensed treatment doses. 13 plasma samples were collected over 48h. Vaginal tissue(VT), cervical tissue(CT) and rectal tissue(RT) were collected at 6, 12, 24, and 48hrs for pooled sample analysis. Drug concentrations were measured by LC-MS/MS. Concentrations were analyzed with WinNonlin® and SigmaPlot®. Dose proportionality was declared if the 90% confidence interval(CI) around the slope(ß1) of log-transformed AUC0-48h÷dose was within 0.64-1.36. Linear regression determined if plasma concentrations predicted mucosal tissue exposure. Data are reported as median(min,max).
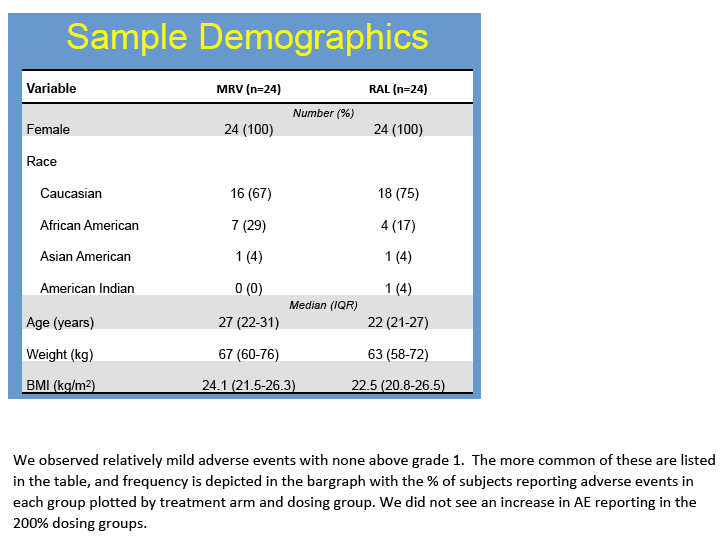
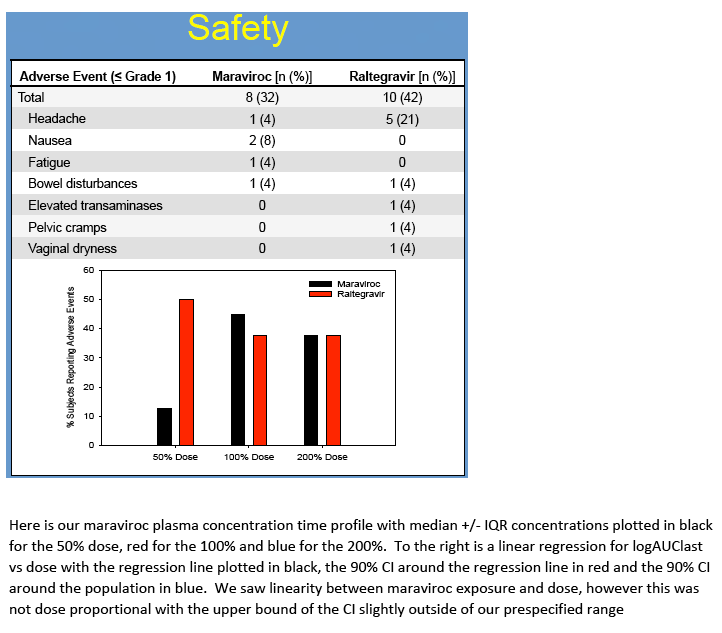
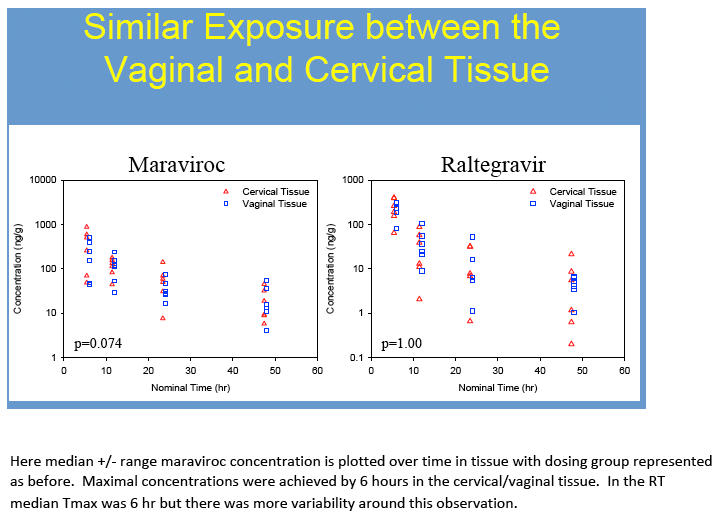
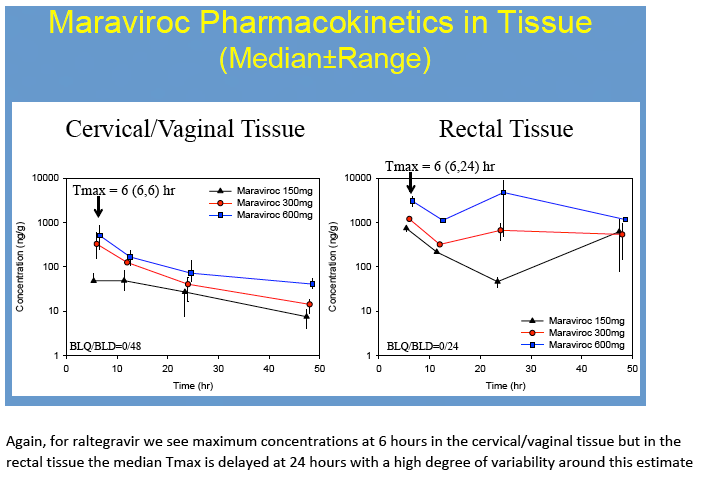
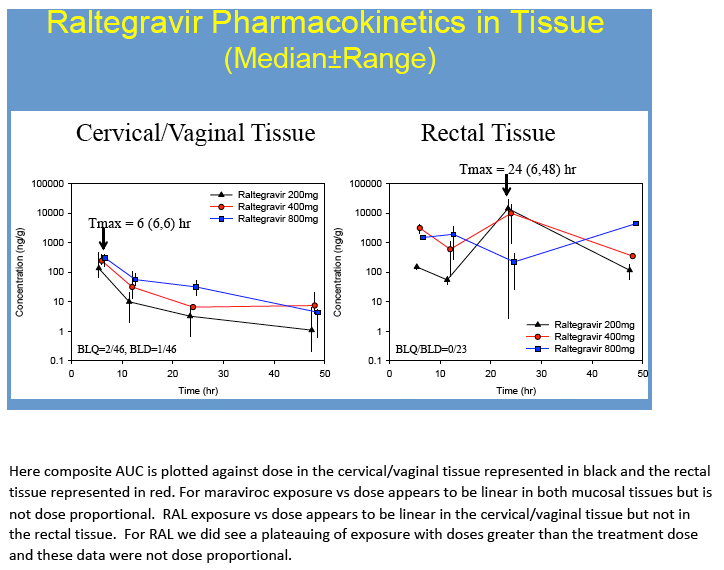
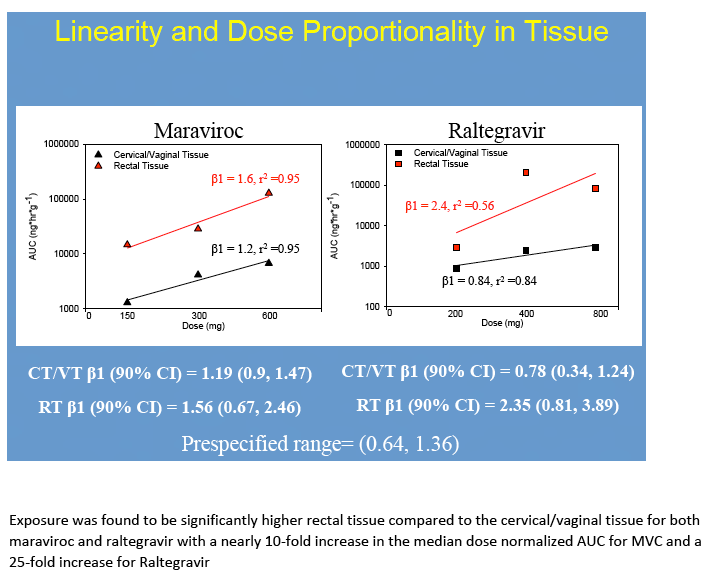
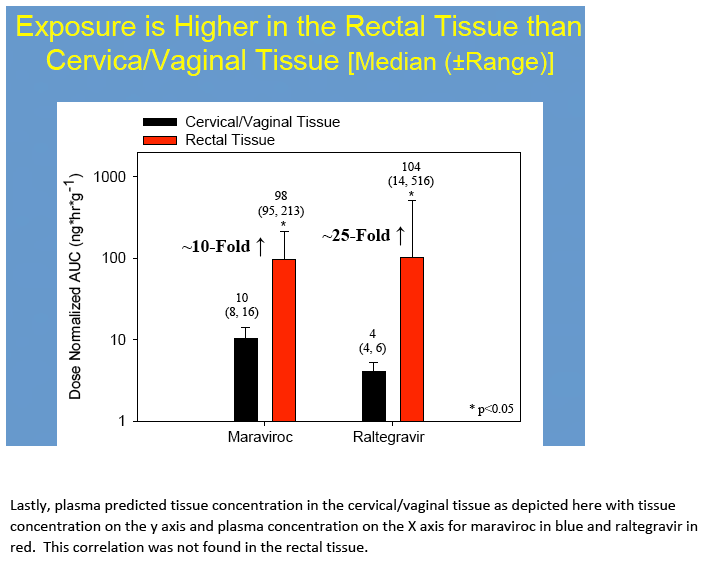
Results: No difference in MVC or RTG exposure was noted between VT and CT(Tmax, Cmax, and AUC0-48h,p>0.4), and the data were combined for subsequent analyses. MVC and RTG demonstrate rapid distribution to VT/CT(Tmax=6(6,6)h for MRV and RTG). Distribution to RT was more variable(Tmax= 6(6,24)h for MVC and 24(6,48)h for RTG). Cmax in VT/CT for MVC at 50/100/200% doses were 53/351/535ng*g-1 and for RTG were 127/329/274ng*g-1. Cmax in RT for MVC at 50/100/200% doses were 740/1201/4749ng*g-1 and for RTG were 151/9893/4394ng*g-1. Compared to VT/CT, RT Cmax was 8-fold higher for MRV and 15-fold higher for RTG. Likewise, RT AUC0-48h was 10-fold higher than VT/CT for MRV and 28-fold higher for RTG.
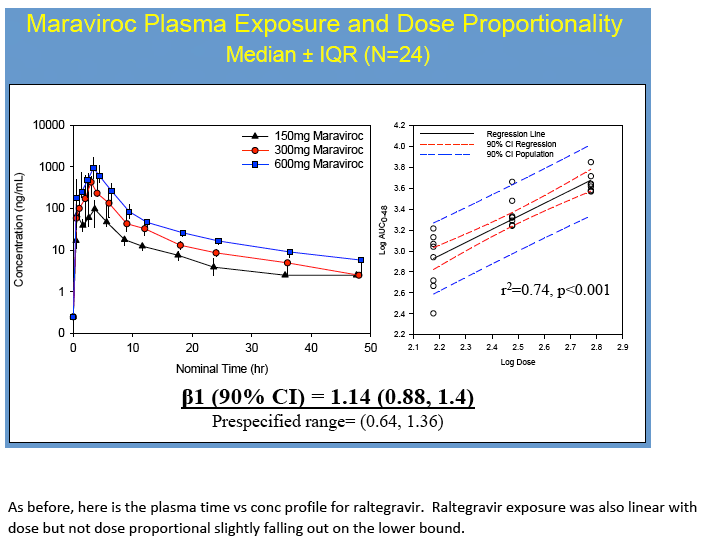
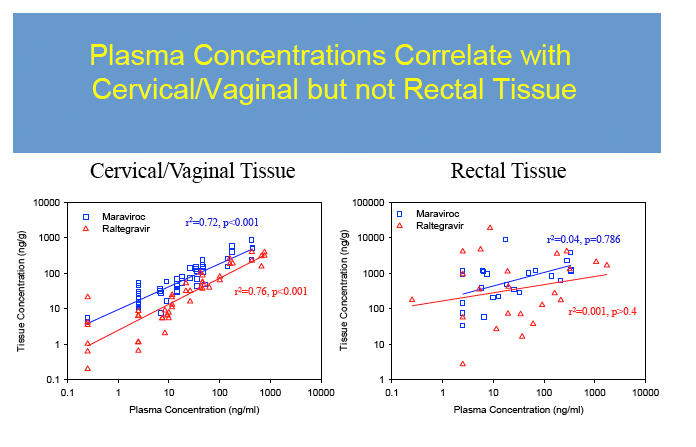
No dose-exposure relationship met statistical dose proportionality criteria. However, MVC demonstrated a linear relationship between dose and AUC0-48h in CT/VT (ß1=1.2,r2=0.95) and RT(ß1=1.6,r2=0.95). A linear relationship was found for RTG in VT/CT(ß1=0.84,r2=0.84) but not RT (ß1=2.4,r2=0.56), where it plateaued with higher doses. VT/CT concentrations were correlated with plasma for MVC (r2=0.72, p<0.001) and RTG (r2= 0.76, p≤0.001). RT concentrations did not correlate with plasma(r2=0.04 for MVC and r2=0.001 for RTG, p≥0.4).
Conclusions: To our knowledge, this is the first report of MRV and RTG PK in multiple mucosal tissues in women. Our findings indicate MVC and RTG rapidly distribute to the FGT making them potentially well-suited for chemoprophylaxis in women. Different distribution patterns were seen in RT. As previously determined in separate studies of men and women, MRV and RTG exposure was 2-11-fold higher in RT than in VT/CT. While MRV exposure in VT/CT and RT increased 5-9-fold across a 4-fold dosing range, RTG did not. This could indicate saturation of RTG transport in tissues, as we have recently found MDR1 transporter expression in VT/CT and RT. These tissue data will be used to create PK models for modeling and simulation studies of intermittent chemoprophylaxis doses. Characterizing the tissue concentrations required for protection is an important next step in discerning these agents' role for HIV prevention.
from authors:



|
| |
|
 |
 |
|
|