 |
 |
 |
| |
Unbound Darunavir Above Wild-Type EC50 With Once-Daily Dose in Pregnancy
|
| |
| |
Slides Below
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, May 19-21, 2014, Washington, DC
Mark Mascolini
Levels of unbound (active) darunavir remained more than 10-fold above the unbound 50% effective concentration (EC50) for wild-type (nonmutant) HIV in the second and third trimesters of pregnancy among women taking a once-daily darunavir/ritonavir dose of 800/100 mg [1]. No infants became infected with HIV.
Previous research in 14 pregnant women taking 600/100 mg of darunavir/ritonavir twice daily found decreased total darunavir exposure--but not unbound darunavir exposure--during pregnancy compared with postpartum [2]. The finding suggested no need for dose adjustment when pregnant women take this twice-daily dose.
The new study extended this research to women taking 800/100 mg of darunavir/ritonavir once daily with other antiretrovirals [1], the dose licensed for people with no antiretroviral experience or no darunavir resistance mutations. Study participants were at least 18 years old, in the second trimester, and taking a once-daily darunavir regimen. Janssen researchers measured total and unbound darunavir plasma concentrations and total ritonavir before dosing and 1, 2, 3, 4, 6, 9, 12, and 24 hours after dosing.
When entering the study, 10 of 17 women (59%) had a viral load below 50 copies and 6 had a viral load below 400 copies. Seven women were white, 5 black, 2 Hispanic, and 3 of other or multiple races. Median age at screening stood at 24 years and ranged from 18 to 33. Median body mass index measured 28 kg/m(2) and ranged from 21 to 50.
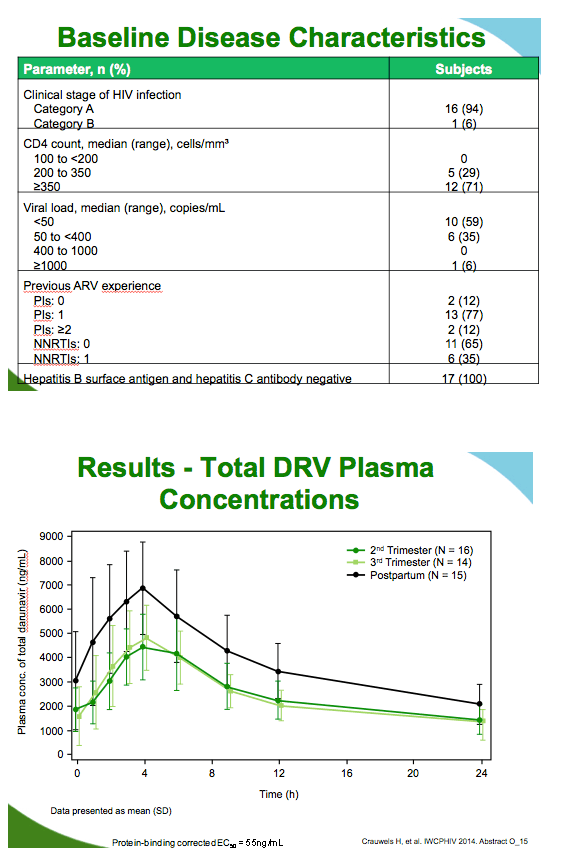
Both total and unbound darunavir concentrations were lower in the second and third trimesters than postpartum, as indicated by the following least squares means ratios for area under the concentration-time curve (AUC), minimum concentration (Cmin), and maximum concentration (Cmax):
Total darunavir second trimester
AUC: 0.66 (90% confidence interval [CI] 0.60 to 0.74)
Cmin: 0.68 (90% CI 0.56 to 0.83)
Cmax: 0.66 (90% CI 0.59 to 0.75)
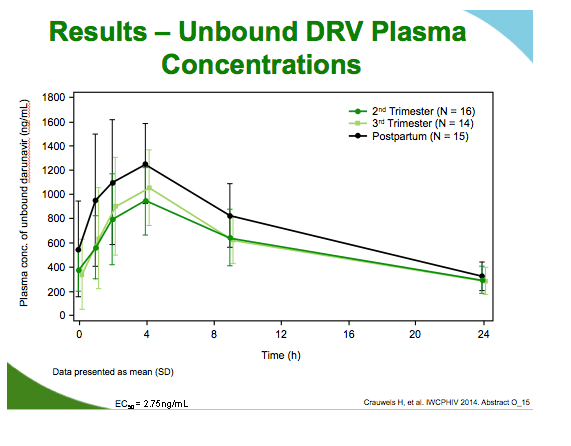
Unbound darunavir second trimester
AUC: 0.76 (90% CI 0.67 to 0.85)
Cmin: 0.87 (90% CI 0.69 to 1.10)
Cmax: 0.75 (90% CI 0.65 to 0.87)
Total darunavir third trimester
AUC: 0.65 (90% CI 0.57 to 0.74)
Cmin: 0.50 (90% CI 0.35 to 0.73)
Cmax: 0.69 (90% CI 0.63 to 0.77)
Unbound darunavir third trimester
AUC: 0.80 (90% CI 0.71 to 0.89)
Cmin: 0.62 (90% CI 0.43 to 0.90)
Cmax: 0.84 (90% CI 0.74 to 0.96)
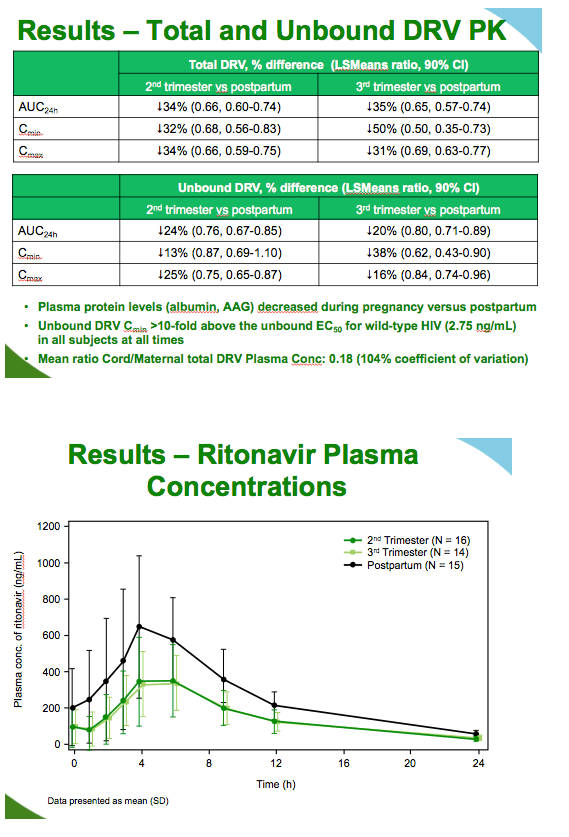
Although lower during pregnancy than postpartum, unbound darunavir levels remained more than 10-fold above the unbound EC50 for wild-type virus, 2.75 ng/mL, in all women at every measure. Ritonavir concentrations were about 45% lower during the second and third trimesters than postpartum. Albumin concentrations were 20% to 27% lower during pregnancy than postpartum, while alpha1-acid glycoprotein levels were 46% lower during pregnancy.
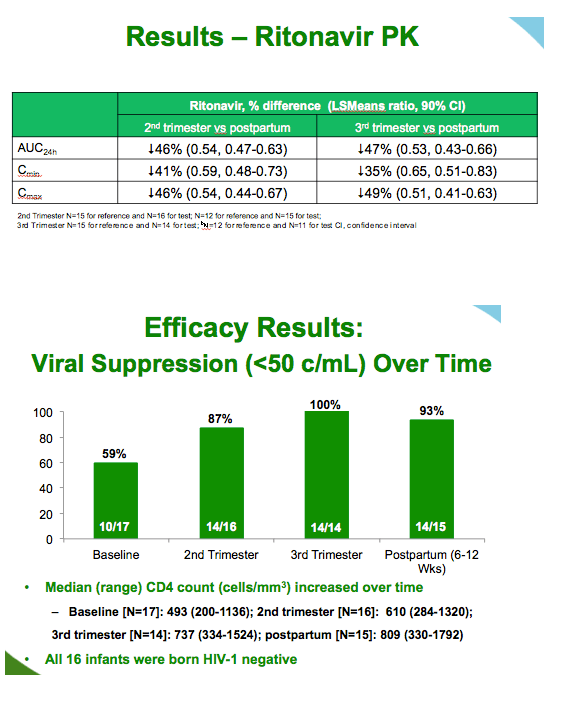
Proportions of women with a viral load below 50 copies rose from 59% (10 of 17) at baseline, to 87% (14 of 16) in the second trimester, 100% (14 of 14) in the third trimester, and 93% (14 of 15) after delivery. Median CD4 count rose over time, from 493 at baseline to 809 postpartum. No women transmitted HIV to an infant.
Six serious adverse events that occurred during treatment were considered related to pregnancy. One such event, gestational diabetes, was judged possibly related to darunavir/ritonavir. Three of 16 infants were born before 37 weeks gestation, at weeks 33, 36, and 36.
The Janssen team suggested that lower darunavir concentrations in pregnancy could reflect pregnancy-related dilution of albumin or alpha1-acid glycoprotein, lower ritonavir concentrations, or both mechanisms. They proposed that "no dose adjustment is required for darunavir/ritonavir 800/100 mg once-daily in pregnant women." The researchers cautioned, though, that clinicians should watch for "additional considerations or use of concomitant medications" that may further lower darunavir exposure during pregnancy.
References
1. Crauwels H, Baugh B, Ryan B, et al. Total and unbound pharmacokinetics of once-daily darunavir/ritonavir in HIV-1-infected pregnant women. 15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. May 19-21, 2014. Washington, DC. Abstract O_15.
2. Zorrilla CD, Wright R, Osiyemi OO, et al. Total and unbound darunavir pharmacokinetics in pregnant women infected with HIV-1: results of a study of darunavir/ritonavir 600/100 mg administered twice daily. HIV Med. 2014;15:50-56. http://onlinelibrary.wiley.com/doi/10.1111/hiv.12047/pdf
---------------
Total and Unbound Pharmacokinetics of Once-Daily Darunavir/ritonavir in HIV-1-Infected Pregnant Women
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. Washington DC, May 19-21, 2014
Crauwels HM,1 Baugh B,2 Ryan B,3 Zorrilla C,4 Osiyemi OO,5 Yasin S,6 Brown K,2 Verboven P,1 Hillewaert V,1 Kakuda TN3
1Janssen Research & Development, Beerse, Belgium; 2. Janssen Therapeutics, Titusville, NJ, US; 3. Janssen Research & Development, Titusville, NJ, US; 4. University of Puerto Rico School of Medicine, San Juan, Puerto Rico; 5. Triple O Research Institute PA, West Palm Beach, FL, US; 6. University of Miami School of Medicine, Miami, FL, US


Program abstract:
Introduction: Antiretroviral therapy during pregnancy reduces risk of mother-to-child transmission (MTCT). Physiologic changes during pregnancy can affect pharmacokinetics. A previous analysis of darunavir/ritonavir 600/100mg twice-daily showed lower total but not unbound darunavir exposure in HIV-1-infected pregnant women, and no MTCT. This study was extended to HIV-1-infected pregnant women receiving once-daily darunavir/ritonavir.
Materials & Methods: Phase IIIb study, HIV-1-infected pregnant women ≥18 years old, in 2nd trimester of pregnancy, receiving darunavir/ritonavir 800/100mg once-daily with other antiretrovirals. Fifty-nine percent (10/17 patients) had viral suppression (<50 copies/mL) at study entry. Darunavir (total and unbound) and ritonavir (total) plasma concentrations were evaluated predose, 1, 2, 3, 4, 6, 9, 12 and 24 hours postdose (3, 6, 12h timepoints only analyzed for total concentrations), during the 2nd and 3rd trimesters, and postpartum. Total darunavir and ritonavir plasma concentrations were determined using a validated HPLC-MS/MS assay. Darunavir unbound fraction was determined by separation through ultrafiltration of 14C darunavir-fortified plasma samples and liquid scintillation counting. Pharmacokinetic parameters were derived using non-compartmental analysis. Safety and efficacy were evaluated at each visit.
Results: 17 women (5 Black, 2 Hispanic, 7 White, 3 Other) enrolled; 16 had evaluable pharmacokinetic data. Total darunavir AUC24h, Cmin and Cmax were lower by 34% (LSMeans ratio, 90% CI: 0.66, 0.60-0.74), 32% (0.68, 0.56-0.83) and 34% (0.66, 0.59-0.75) during the 2nd trimester and by 35% (0.65, 0.57-0.74), 50% (0.50, 0.35-0.73) and 31% (0.69, 0.63-0.77) during the 3rd trimester, versus postpartum. Unbound darunavir AUC24h, Cmin and Cmax were lower by 24% (0.76, 0.67-0.85), 13% (0.87, 0.69-1.10) and 25% (0.75, 0.65-0.87) during the 2nd trimester, and by 20% (0.80, 0.71-0.89), 38% (0.62, 0.43-0.90) and 16% (0.84, 0.74-0.96) during the 3rd trimester, versus postpartum. Unbound darunavir was >10-fold above the unbound EC50 for wild-type HIV (2.75 ng/mL) in all subjects at all times. Ritonavir pharmacokinetic parameters decreased by approximately 45-50% during the 2nd and 3rd trimesters, versus postpartum. Albumin and α1-acid glycoprotein (AAG) concentrations were 20-27% and 46% lower, respectively, during pregnancy versus postpartum (median % decrease). Viral suppression was maintained in >88% of subjects at all times during gestation. CD4+ counts increased in the 2nd and 3rd trimesters and 6-12 weeks postpartum, with lower values observed in-between (2-5 weeks postpartum). Six serious adverse events were reported in the treatment phase, all considered pregnancy-related; only 1 (gestational diabetes) was considered possibly related to darunavir/ritonavir. Three of 16 infants were born prior to 37 weeks (33, 36 and 36 weeks), there were no birth defects, and all infants were HIV-1-negative (standard PCR testing).
Conclusions: Total darunavir decreased during pregnancy possibly due to pregnancy-related albumin and/or AAG dilution and/or decreased ritonavir concentrations. The decrease in unbound darunavir during pregnancy was less pronounced than for total darunavir, although larger than previously reported in HIV-1-infected pregnant women treated with twice-daily darunavir/ritonavir. Unbound darunavir was >10-fold above the wild-type EC50, high viral suppression rates were maintained throughout the study, and there was no MTCT. The regimen was well tolerated. No dose adjustment is required for darunavir/ritonavir 800/100mg once-daily in pregnant women.


|
| |
|
 |
 |
|
|