 |
 |
 |
| |
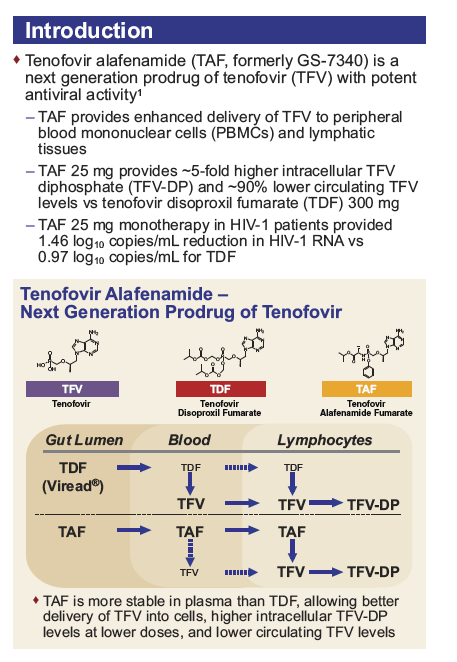
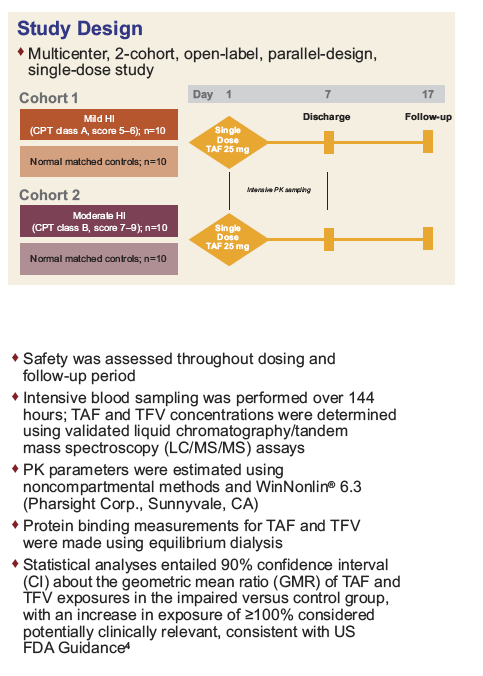
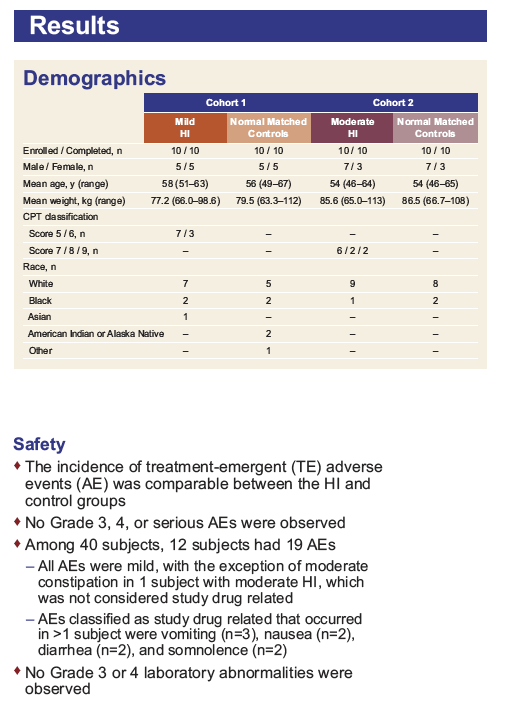
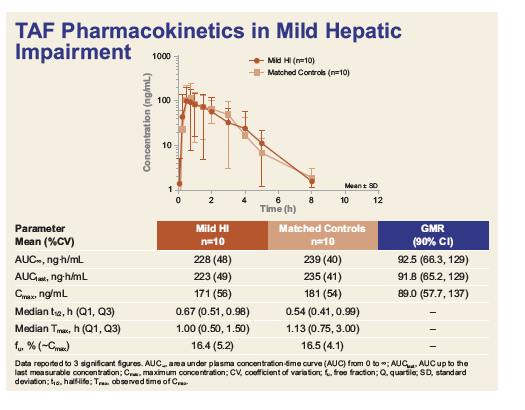
Pharmacokinetics and Safety of Tenofovir Alafenamide in Subjects With Mild or Moderate Hepatic Impairment
|
| |
| |
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, May 19-21, 2014, Washington, DC
Rebecca Begley,1 Hal Martin,1 Eileen B. Lawson,1 Xuelian Wei,1 Mona Vimal,1 Thomas C. Marbury,2 Brian P. Kearney1
1Gilead Sciences, Inc., Foster City, California; 2Orlando Clinical Research Center, Orlando, Florida

48 Week Study of Tenofovir Alafenamide (TAF) vs. Tenofovir Disoproxil Fumarate (TDF), Each in a Single Tablet Regimen (STR) with Elvitegravir, Cobicistat, and Emtricitabine [E/C/F/TAF vs. E/C/F/TDF] for Initial HIV Treatment......http://www.natap.org/2013/ICAAC/ICAAC_54.htm
Twenty-Eight Day Safety and Efficacy of Tenofovir Alafenamide (TAF) Fumarate in Chronic Hepatitis B (CHB) Patients........http://www.natap.org/2013/AASLD/AASLD_91.htm
Antiviral Activity of Tenofovir Alafenamide (TAF) against Major NRTI-Resistant Viruses: Improvement over TDF/TFV is Driven by Higher TFV-DP Loading in Target cells.......http://www.natap.org/2013/ResisWksp/ResisWksp_04.htm....[(IDRW)
International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies; June 4-8, 2013; Toronto, ON, Canada]
Lack of Effect of Tenofovir Alafenamide (TAF) on Primary Osteoblasts In Vitro at Clinically Relevant Drug Concentrations......http://www.natap.org/2013/ICAAC/ICAAC_15.htm

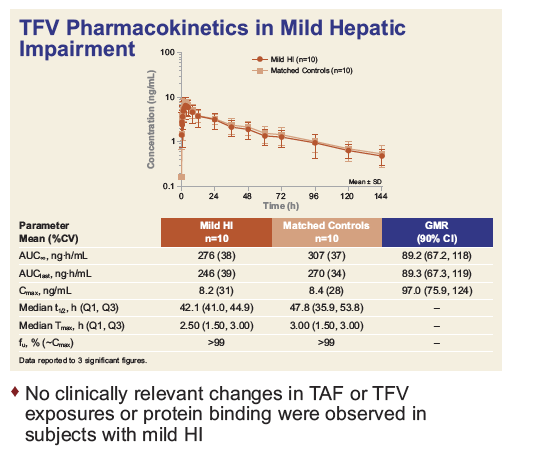
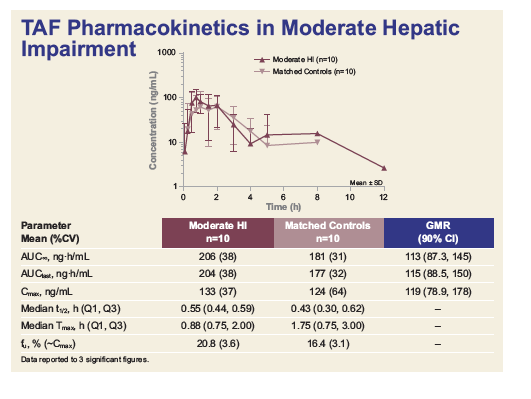
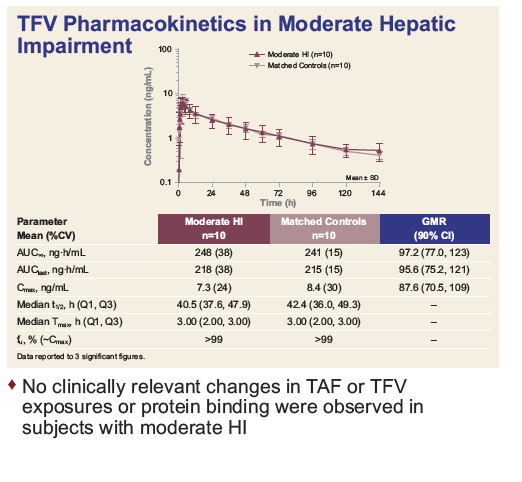
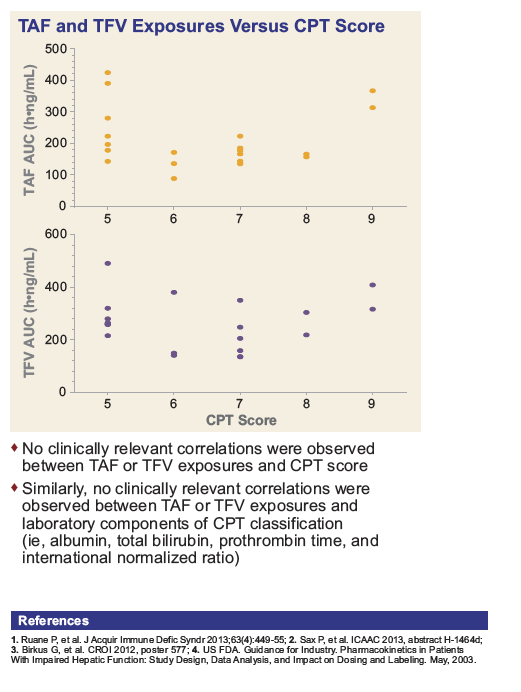
|
| |
|
 |
 |
|
|