 |
 |
 |
| |
A novel microdose approach to assess bioavailability, intestinal absorption, gut metabolism, and hepatic clearance of simeprevir in healthy volunteers
|
| |
| |
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HIV & Hepatitis Therapy, Washington DC; May 19-21, 2014
Sivi Ouwerkerk-Mahadevan,1 Jan Snoeys,1 Alex Simion,2 Ellen Scheers,1 Maria Beumont Mauviel2
1Janssen Research & Development, Beerse, Belgium;
2Janssen Infectious Diseases BVBA, Beerse, Belgium

Program abstract-
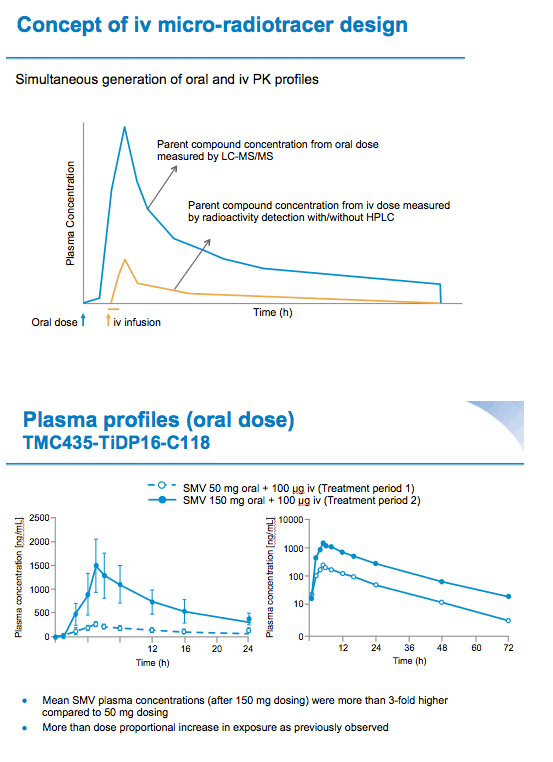
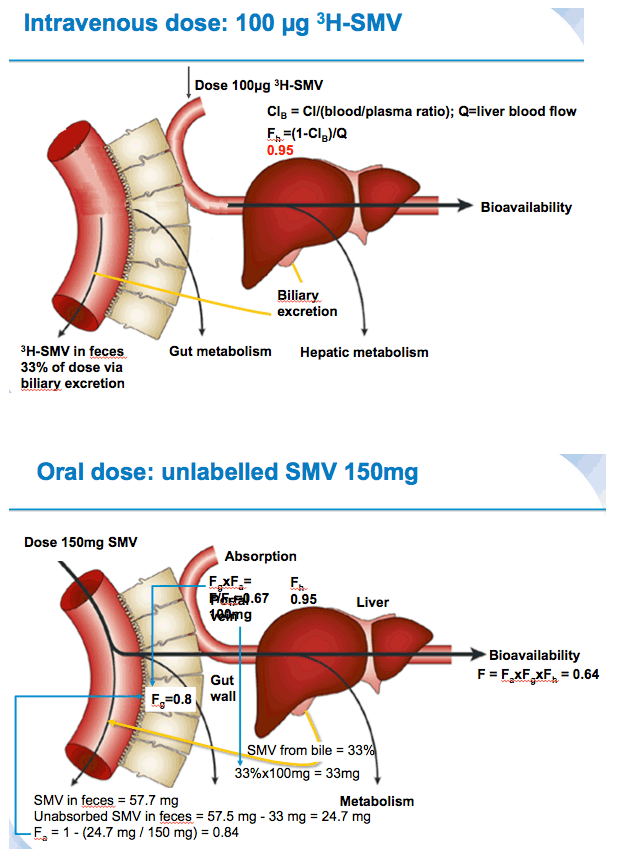
Background: Simeprevir (SMV, TMC435) is a one pill, once-daily (QD), hepatitis C virus (HCV) NS3/4A protease inhibitor approved in Japan, Canada, and the USA, and under regulatory review in Europe for treatment of chronic HCV genotype 1 infection. This sequential, single-dose Phase I study assessed the pharmacokinetics (PK) and absolute bioavailability (Fabs) of single oral SMV doses of 50 mg and 150 mg followed by an intravenous (i.v.) microdose of [3H]-SMV 100 μg in healthy volunteers. The proportion of the i.v. dose excreted in urine and faeces, and information from the oral dose was used to determine the contribution of intestinal absorption, and intestinal- and hepatic-metabolism in the PK of simeprevir.
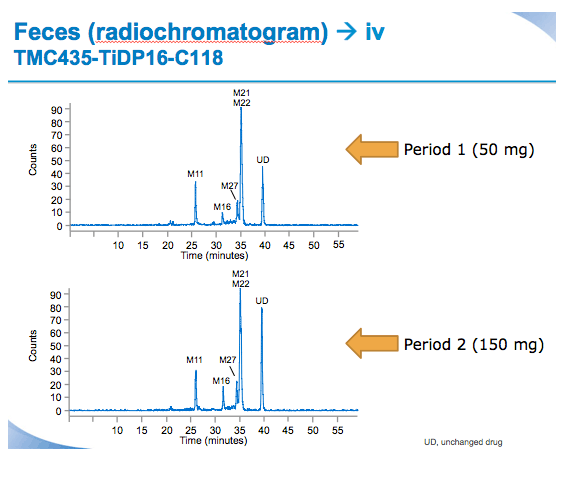
Materials and Methods: Six healthy male volunteers (median age 47.0 years) received two sequential treatments separated by a washout period. Treatment Period 1: single oral dose of SMV 50 mg followed 5 hours later by a single 10-minute i.v. infusion of 100 μg [3H]-SMV (100μCi). Treatment Period 2: single oral dose of SMV 150 mg followed 5 hours later by [3H]-SMV (dosing as before). SMV and [3H]-SMV plasma PK profiles, and total plasma radioactivity were determined for both treatments 0-72 hours after oral SMV administration. Urine per 24-hour interval and faeces were collected for at least 96 hours after oral SMV administration. Urine and faeces were analysed for total radioactivity and metabolite profiling was performed on faeces. Safety was assessed by laboratory examination, electrocardiogram, vital and clinical signs.
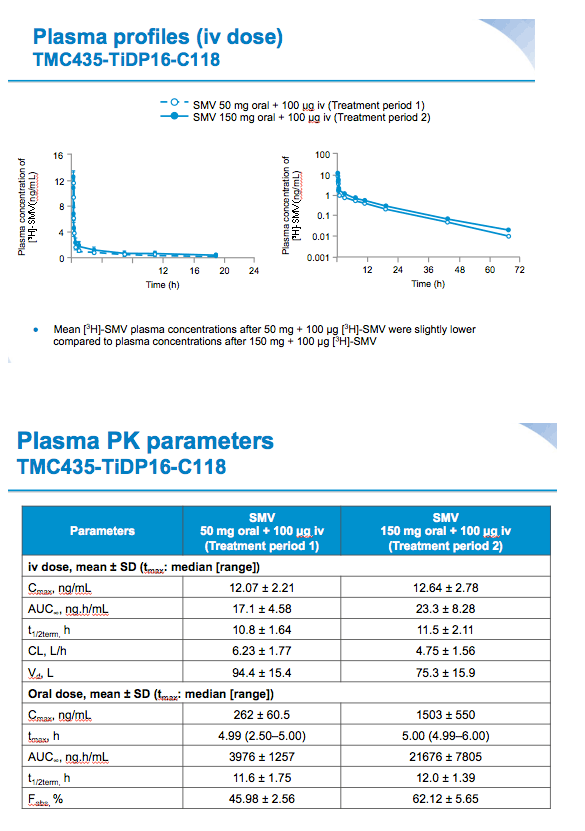
Results: Systemic clearance was saturable between single oral doses of SMV 50 mg and 150 mg, decreasing from 6.23 L/h to 4.75 L/h, respectively. Volume of distribution decreased from 94.4 L for the 50 mg dose to 75.3 L for the 150 mg dose. Elimination half-lives were 10.8 h and 11.5 h, respectively, for the 50 mg and 150 mg doses. These changes are consistent with saturation of hepatic uptake of simeprevir at the 150 mg dose. The SMV plasma exposure was more than dose-proportionally higher for SMV 150 mg compared with 50 mg.
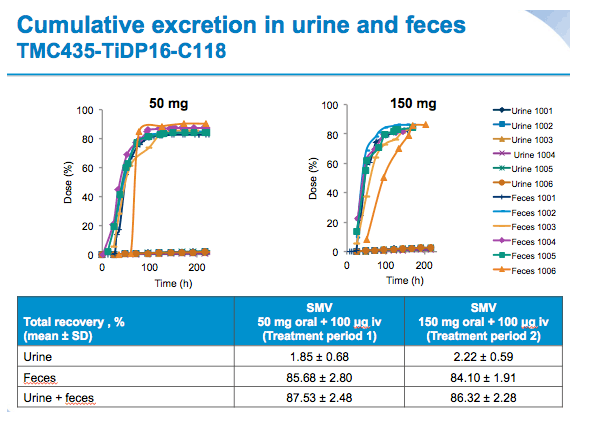
With both treatments, radioactivity was excreted mainly in the faeces (~85% of total radioactive dose) and only ~2% of the total radioactive dose was recovered in urine.
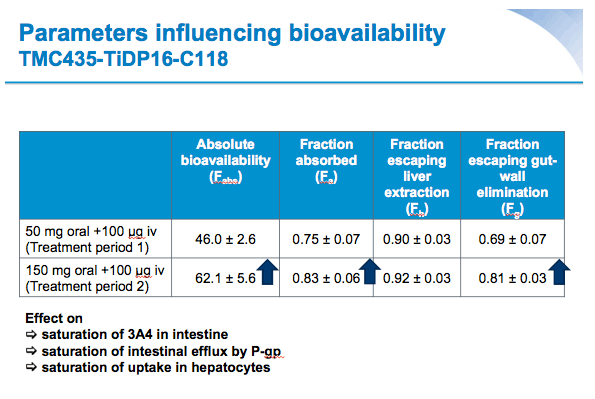
Mean values for fraction absorbed (Fa) (0.75±0.07 for 50 mg, 0.83±0.06 for 150 mg) and fraction escaping gut-wall elimination (Fg) (0.69±0.07 for 50 mg and 0.81±0.03 for 150 mg) were higher for SMV 150 mg versus SMV 50 mg. Mean Fabs of SMV was higher after a single oral dose of 150 mg vs 50 mg (62.1% versus 46.0%, respectively).
Both treatments were generally well tolerated in healthy male volunteers, with no serious adverse events (AEs), fatal AEs, grade 3/4 AEs or AE-related treatment discontinuations.
Conclusions: The absolute bioavailability of orally administered SMV was dose-dependent; 46% at 50 mg and 62% at 150 mg. Intestinal absorption, metabolism and hepatic uptake were saturable, with a greater fraction absorbed and lower fraction metabolized at the higher dose.
This study was funded by Janssen.



|
| |
|
 |
 |
|
|