 |
 |
 |
| |
Sofosbuvir+GS-5816 GT 1-6: ASTRAL-1, ASTRAL-2, ASTRAL-3, ASTRAL-4
|
| |
| |
- SOF+GS5816+Rbv in GT3/cirrhosis
- PK analysis SOF/LDV, HIV ARTs in HIV coinfected
- GS9857+SOF+GS5816, 6 & 8 weeks
- NS5A pre-treatment global prevalence & effect on treatment outcome using deep sequencing
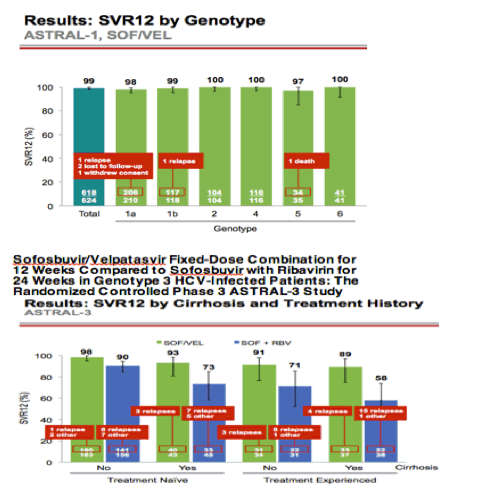
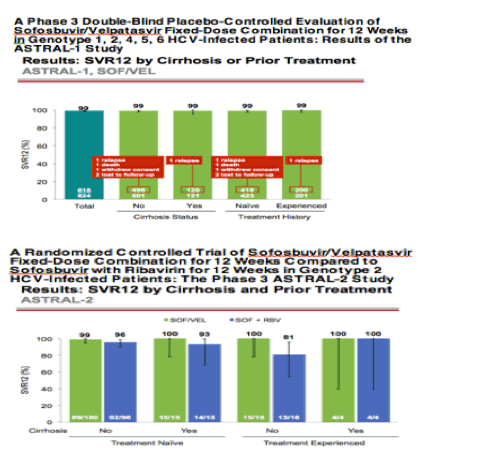
AASLD: A Phase 3 Double-Blind Placebo-Controlled Evaluation of Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks in Genotype 1, 2, 4, 5, 6 HCV-Infected Patients: Results of the ASTRAL-1 Study - (11/17/15)
published: http://www.natap.org/2015/AASLD/AASLD_52.htm
AASLD: A Randomized Controlled Trial of Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks Compared to Sofosbuvir with Ribavirin for 12 Weeks in Genotype 2 HCV-Infected Patients: The Phase 3 ASTRAL-2 Study - (11/30/15)
AASLD: Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks Compared to Sofosbuvir with Ribavirin for 24 Weeks in Genotype 3 HCV-Infected Patients: The Randomized Controlled Phase 3 ASTRAL-3 Study - (11/30/15)
AASLD: Sofosbuvir/Velpatasvir Fixed-Dose Combination for the Treatment of HCV in Patients With Decompensated Liver Disease: the Phase 3 ASTRAL-4 Study / ASTRAL 1, 2 and 3 - (11/30/15)
------------------------
published:
Sofosbuvir and Velpatasvir for HCV Genotype 2 and 3 Infection - (11/30/15)
Sofosbuvir and Velpatasvir for HCV in Patients with Decompensated Cirrhosis (ASTRAL-4) - (11/30/15)
-----------------------
AASLD: Pharmacokinetic Analyses of Ledipasvir/Sofosbuvir and HIV Antiretroviral Regimens in Subjects With HCV/HIV Coinfection - (11/16/15)
--------------------------
AASLD: Sofosbuvir Plus Velpatasvir (GS-5816) Combination Therapy for Treatment-Experienced Patients With Genotype 1 or 3 Hepatitis C Virus Infection: A Randomized Trial - (11/17/15)
GT 3 with cirrhosis recd with & without Rbv
Phase 2 Study:
The SVR12 rates in treatment-experienced patients with genotype 3 HCV infection and cirrhosis who received sofosbuvir plus 100 mg of velpatasvir without and with ribavirin were 88% and 96%, respectively.
--------------------------
AASLD: Sofosbuvir/Velpatasvir + GS-9857 for 6 or 8 Weeks in Genotype 1 or 3 HCV-Infected Patients - (11/16/15)
-----------------------
AASLD: Prevalence of Pre-Treatment NS5A Resistance Associated Variants in Genotype 1 Patients Across Different Regions Using Deep Sequencing and Effect on Treatment Outcome with LDV/SOF - (11/16/15)
------------------
|
| |
|
 |
 |
|
|