 |
 |
 |
| |
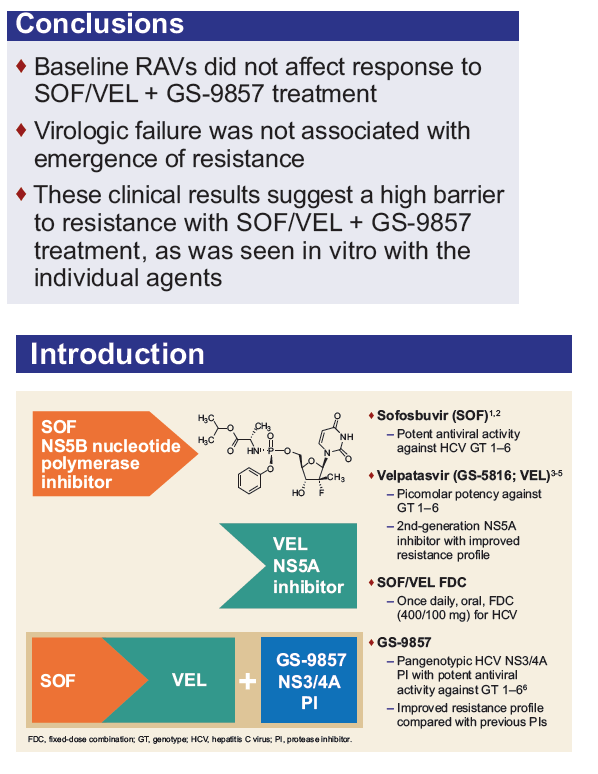
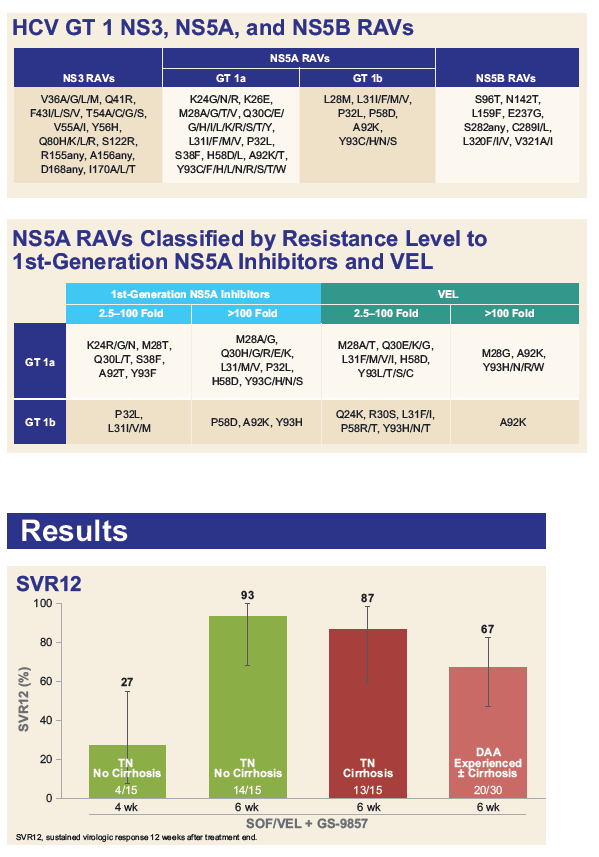
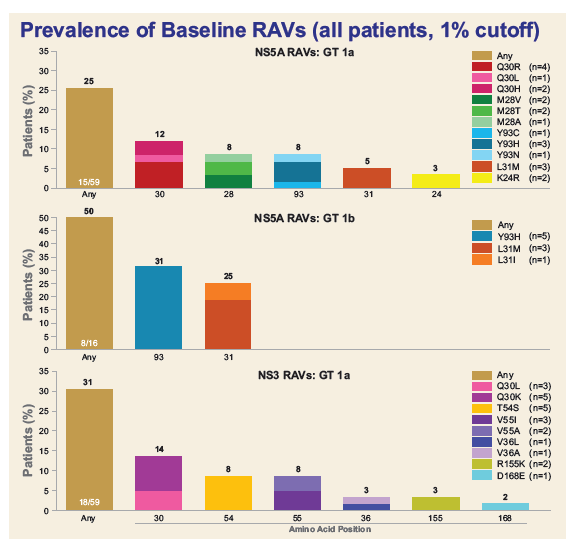
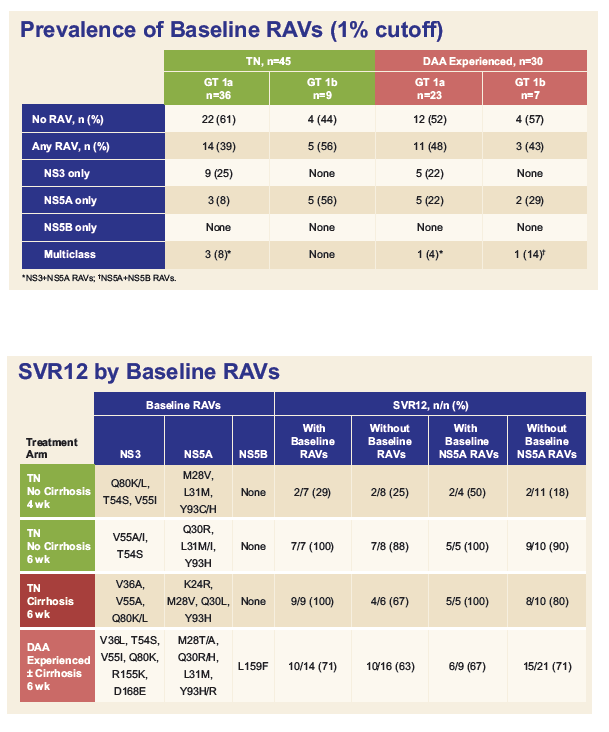
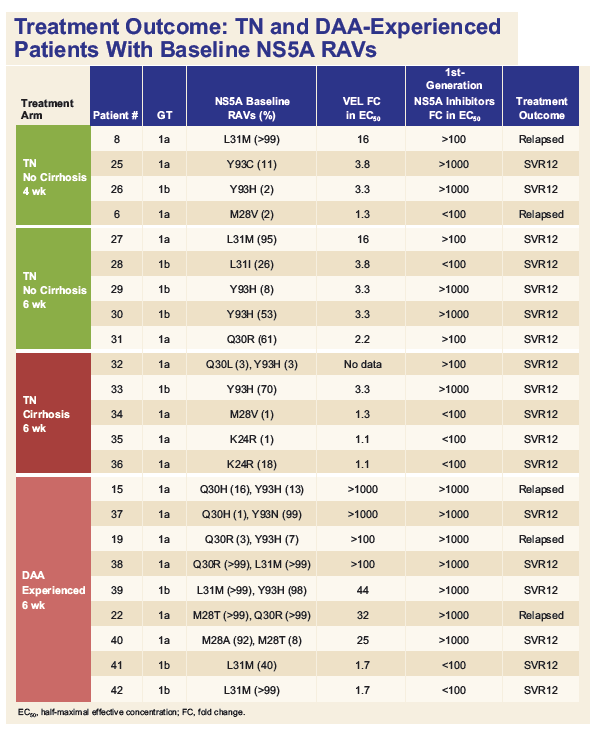
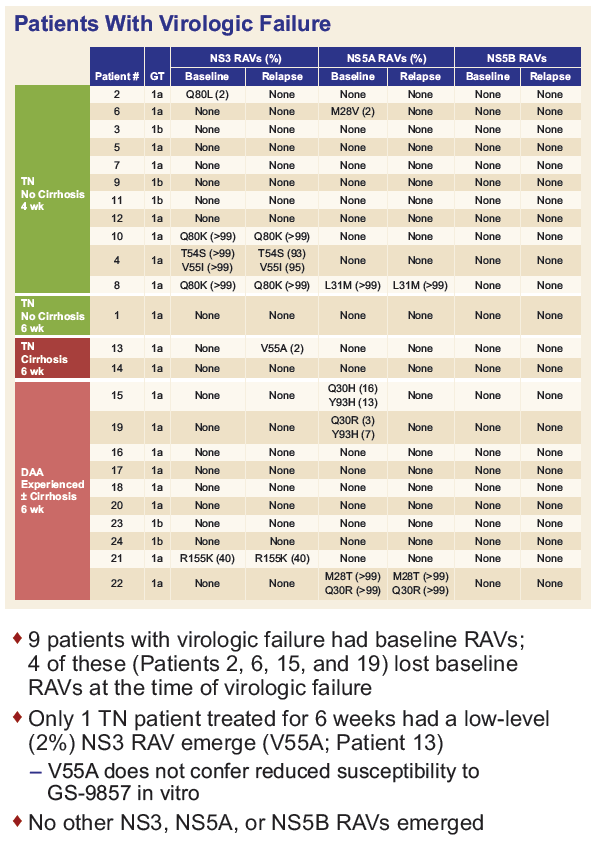
Resistance Analysis of Treatment-Naïve and DAA-Experienced Genotype 1 Patients With and Without Cirrhosis Who Received Short-Duration Treatment With Sofosbuvir/Velpatasvir + GS-9857
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Ed Gane,1 Evguenia S. Svarovskaia,2 Rob Hyland,2 Luisa M. Stamm,2 Hadas Dvory-Sobol,2 Diana M. Brainard,2 Krishna Chodavarapu,2 Michael D. Miller,2 Hongmei Mo,2 Christian Schwabe1
1Auckland Clinical Studies Ltd, Auckland, New Zealand; 2Gilead Sciences, Inc., Foster City, CA
AASLD: Sofosbuvir/Velpatasvir + GS-9857 for 6 or 8 Weeks in Genotype 1 or 3 HCV-Infected Patients - (11/16/15)


|
| |
|
 |
 |
|
|