 |
 |
 |
| |
Effect of Chronic Kidney Disease on the Pharmacokinetics of Ombitasvir, Paritaprevir, Ritonavir and Dasabuvir in Subjects with HCV Genotype 1 Infection
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Diana Shuster1, Rajeev Menon1, Daniel E. Cohen2, Doerthe Eckert3, Amit Khatri1
1AbbVie Inc., Clinical Pharmacology and Pharmacometrics, North Chicago, IL, USA; 2AbbVie Inc., Infectious Disease Development, North Chicago, IL, USA; 3AbbVie Deutschland GmbH & Co.KG, Clinical Pharmacology and Pharmacometrics, Ludwigshafen, Germany
AASLD: RUBY-I: Ombitasvir/Paritaprevir/Ritonavir + Dasabuvir ± Ribavirin in Non-Cirrhotic HCV Genotype 1-Infected Patients With Severe Renal Impairment or End-Stage Renal Disease - (12/16/15)
AASLD/2014: The Pharmacokinetics and Safety of the Direct Acting Antiviral Regimen of ABT-450/r, Ombitasvir with/without Dasabuvir in Subjects with Mild, Moderate and Severe Renal Impairment Compared to Subjects with Normal Renal Function.....http://www.natap.org/2014/AASLD/AASLD_55.htm



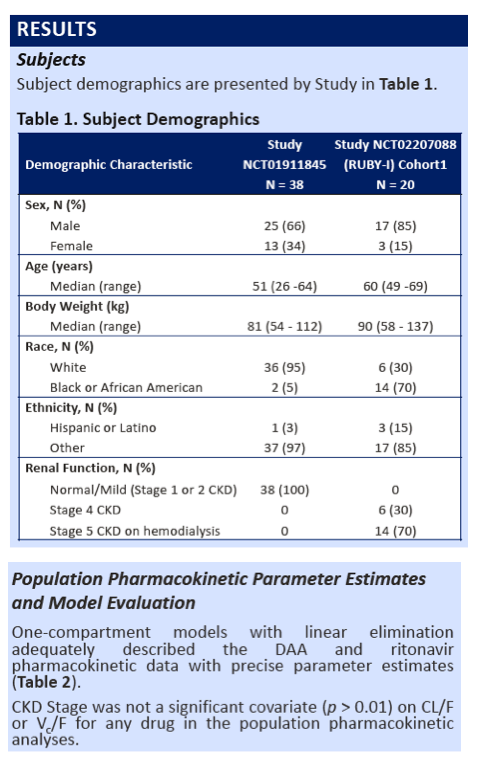
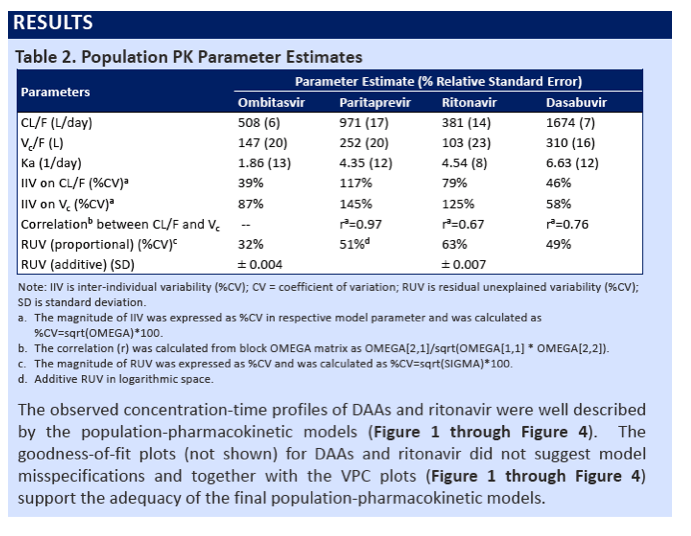
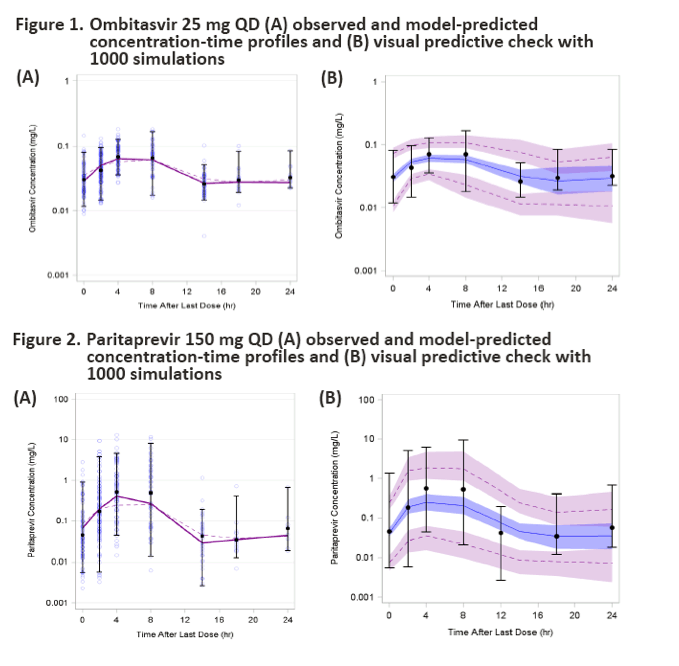
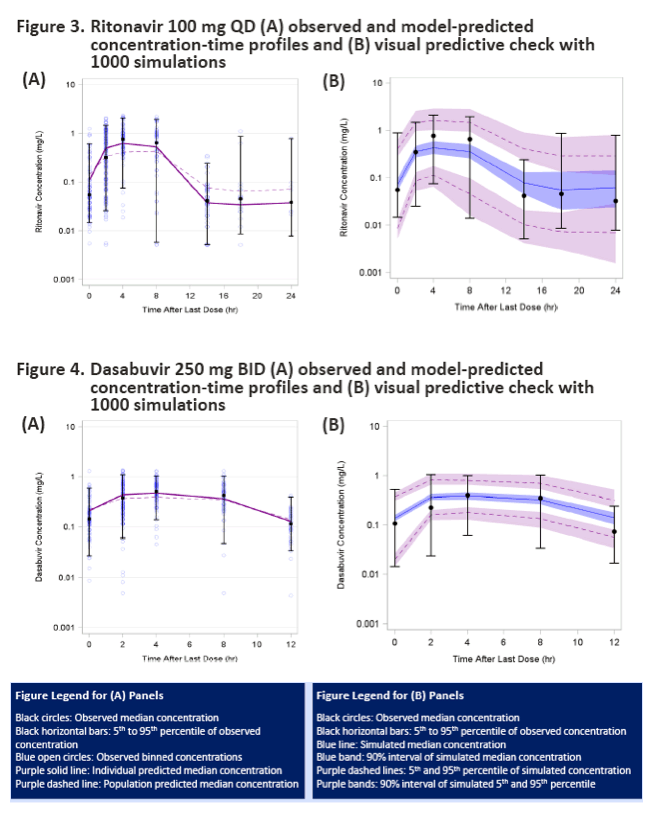
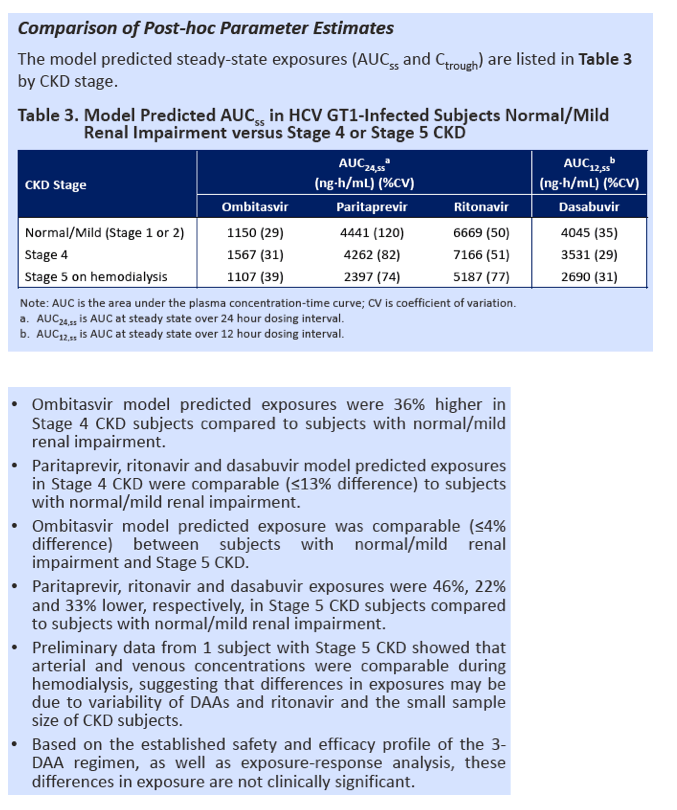

|
| |
|
 |
 |
|
|