 |
 |
 |
| |
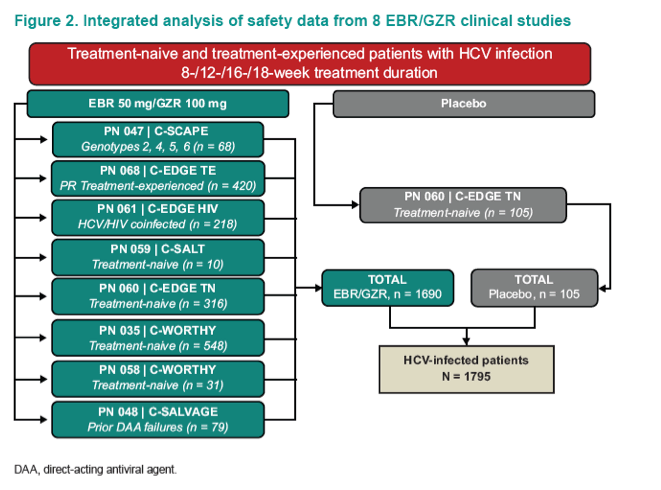
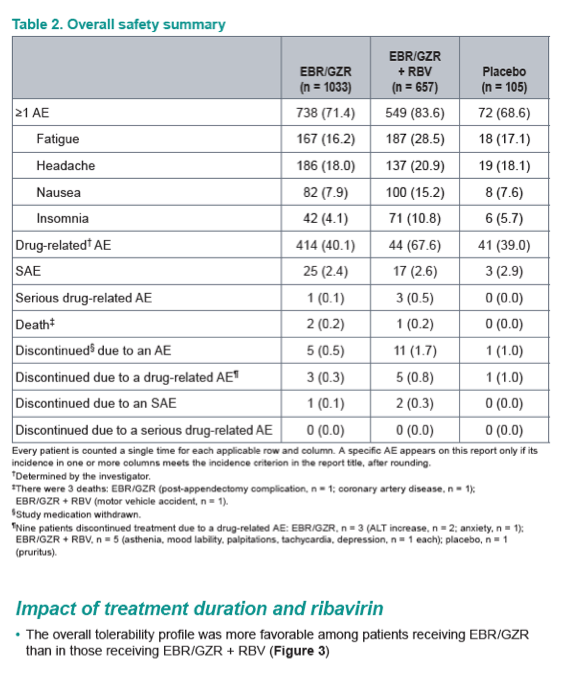
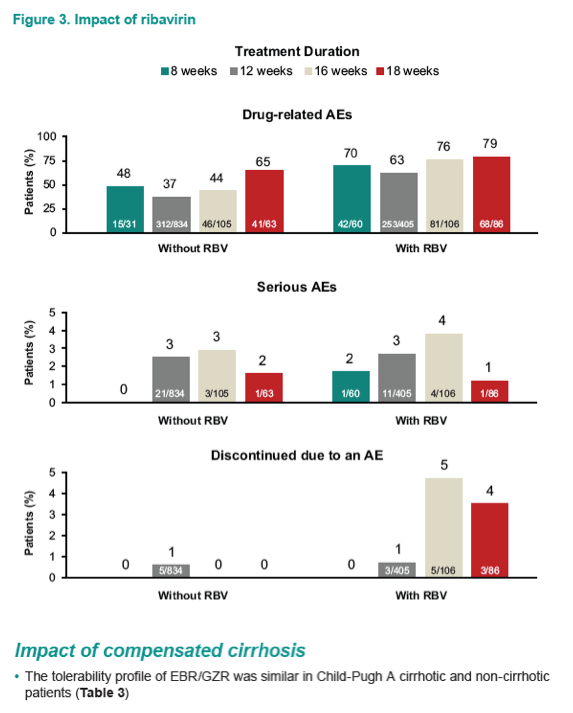
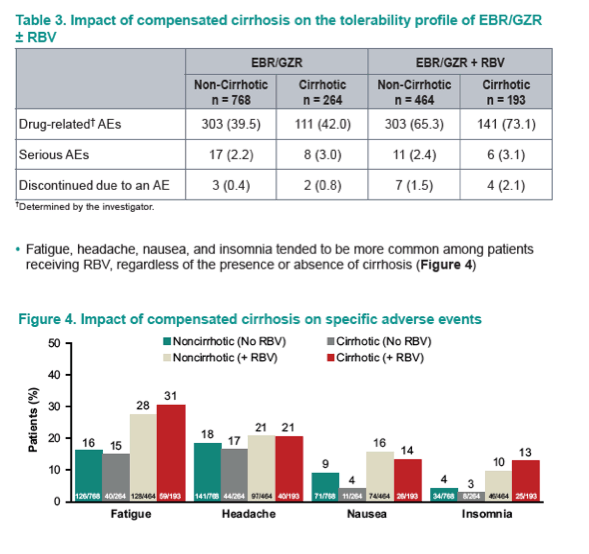
Safety and Tolerability of Elbasvir/Grazoprevir in Patients With Chronic Hepatitis C: An Integrated Analysis of Phase 2-3 Trials
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Dusheiko GM1; Manns MP2; Vierling JM3; Reddy KR4; Sulkowski M5; Kwo P6; Lawitz E7; Brown DD8; Klopfer SO8; Robertson M8; Wahl J8; Barr E8; Haber B8; Charles ED8*
1University College London, Institute of Liver and Digestive Health, Royal Free Hospital, London, UK; 2Medical School of Hannover, Hannover, Germany; 3Baylor St. Luke's Medical Center, Houston, TX, USA; 4University of Pennsylvania, Philadelphia, PA, USA; 5Johns Hopkins University School of Medicine, Baltimore, MD, USA; 6Indiana University School of Medicine, Indianapolis, IN, USA; 7The Texas Liver Institute, University of Texas
Health Science Center, San Antonio, TX, USA; 8Merck & Co., Inc., Kenilworth, NJ, USA. *Former employee of Merck & Co., Inc.






|
| |
|
 |
 |
|
|