 |
 |
 |
| |
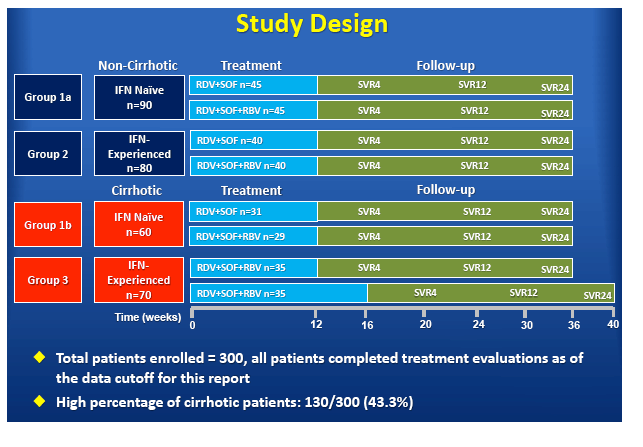
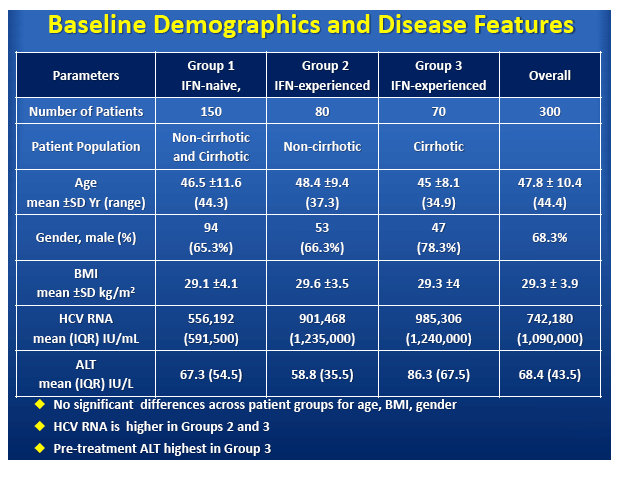
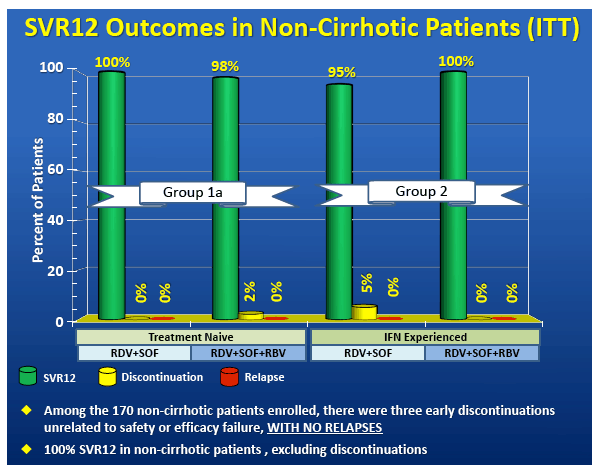
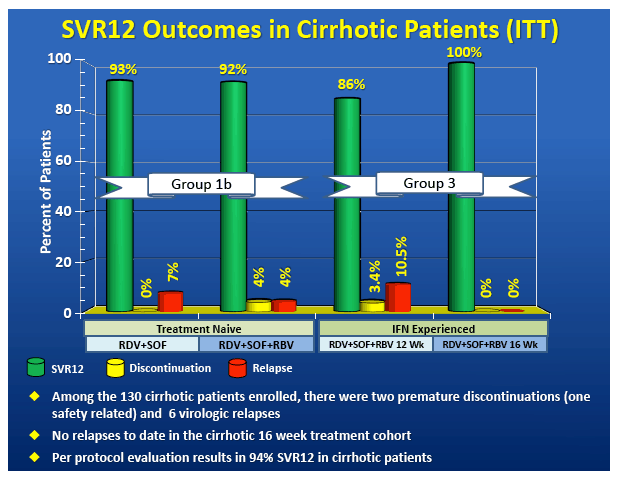
High Virologic Response Rate in Egyptian HCV-Genotype 4 Patients Treated with Ravidasvir (PPI-668) and Sofosbuvir: Results of a Large Multicenter Phase 3 Registrational Trial
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Gamal Esmat1, Maissa El Raziky1, 2, Asmaa Gomaa3, Tamer Elbaz1, Mahmoud M. Abouelkhair1, 2, Alyaa Sabry3, Hadeel Gamal El Deen1, Mohamed K. Ashour1, 2, Mohammed Abdel-Hamid4, Ola Nada5, Sherine Helmy6, Hanaa Abdel-Maguid6, Richard Colonno7, Nathaniel Brown7, Eric Ruby7, Pamela Vig7, Imam Waked3
1Cairo University, 2Cairo Fatemic Hospital, 3National Liver Institute, 4Minia University, 5Ain Shams University, 6Pharco Pharmaceuticals and 7Presidio Pharmaceuticals







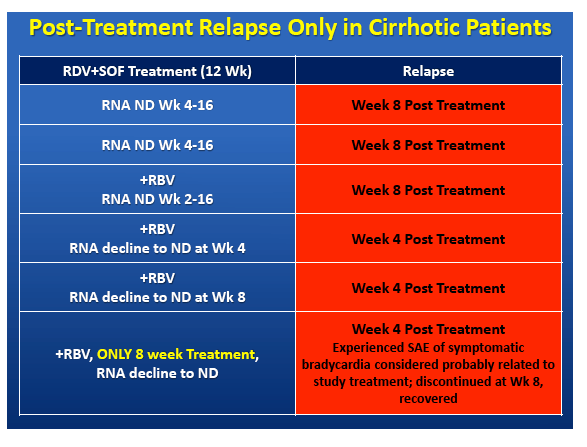

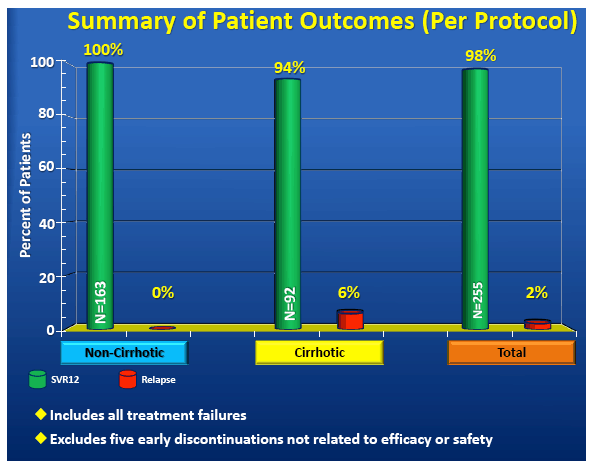

|
| |
|
 |
 |
|
|