 |
 |
 |
| |
Cascade of Care for Hepatitis C Virus (HCV) Infection within the U.S. Department of Veterans Affairs (VA)
|
| |
| |
Reported by Jules Levin
DDW 2015 - May 16-19 2015 Wash DC
Marissa Maier1 , David Ross1 , Maggie Chartier1 , Pamela S. Belperio2 , Lisa I. Backus2
1 Office of Public Health/Clinical Public Health, Washington, District of Columbia, United States; 2 Office of Public Health/Population Health, Palo Alto, California, United States

Background With new efficacious HCV treatments available, a population health approach is useful to assess a healthcare system's ability to identify infected individuals, link them to care, provide antiviral treatment, and achieve sustained virologic response (SVR).
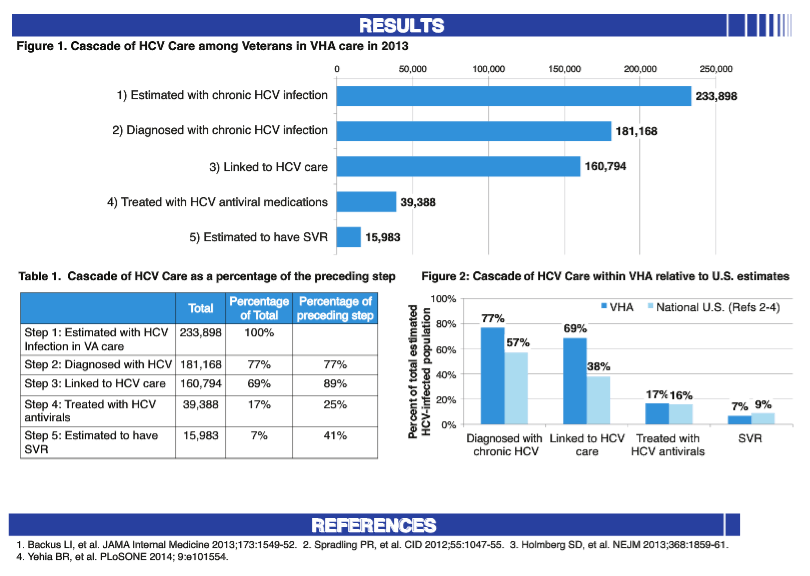
Methods The Cascade of HCV Care for all Veterans in VA care in 2013 (as defined by at least one outpatient visit) has five steps:
(1) Number with HCV infection-We used VA's Corporate Data Warehouse to determine HCV screening, RNA confirmatory testing, infection prevalence and per year incident diagnosis rates. The estimated total number with HCV infection reflects the sum of 1) those already identified with a detectable HCV viral load or genotype, 2) estimated additional cases if RNA testing were performed on those with positive HCV antibody results without RNA testing and 3) estimated additional cases if all unscreened Veterans were screened based on the incident diagnosis rate in each sex, birth cohort and race/ethnicity stratum in care in 2013 multiplied by the ratio of the 2013 to 2012 rate.
(2) Diagnosed with HCV-known detectable HCV viral load or genotype.
(3) Linked to HCV care-entered into the HCV Clinical Case Registry which requires manual review of cases and HCV entered on the problem list.
(4) Antiviral treatment-receipt of any HCV antiviral medication including boceprevir, consensus interferon, interferon, peginterferon, ribavirin or telaprevir from VA at any time up to and including 31 December 2013.
(5) Achieved SVR-undetectable HCV RNA on at least one test 12 weeks or more after the end of antiviral treatment. The SVR rate among those evaluable for SVR was then applied to those still on treatment or recently completing treatment to determine the number of patients expected to have an SVR.
Results Overall, 5,596,178 Veterans had a VA outpatient visit in 2013 and 3,135,396 (56.0%) had VA HCV screening. Of the 218,111 Veterans with positive antibody screens, 208,893 (95.8%) had RNA testing for HCV infection. Of those with positive antibody screens who received RNA-based testing, 161,044 (77.1%) had HCV infection. An additional 20,124 Veterans had HCV infection based on RNA testing without preceding antibody testing. Altogether 181,168 individuals were diagnosed with HCV infection. An estimated 45,623 additional cases of HCV infection would be found with screening of the entire unscreened population in VA care in 2013. An estimated additional 7,107 individuals with HCV infection would be identified by applying the observed prevalence (77.1%) in those with RNA testing to the 9,218 with positive HCV antibody tests who lacked RNA testing. Results for each step of the Cascade appear in the Table.
Conclusion VA has diagnosed the majority of patients with HCV and linked them to HCV care. The largest gap in the Cascade of HCV Care is in the initiation of antiviral treatment.

|
| |
|
 |
 |
|
|