 |
 |
 |
| |
Pharmacokinetics of ATV/r or DRV/r in + 2 NRTIs among HIV-1-naive Patients with Severe Immunosuppression - IMEA 040-DATA sub-study
|
| |
| |
Unbound ATV and DRV Stable for 48 Weeks in Patients With Low CD4s
15th European AIDS Conference, October 21-24, 2015, Barcelona
Mark Mascolini
Unbound fractions of ritonavir-boosted atazanavir and darunavir remained largely stable through 48 weeks in people starting these protease inhibitors (PIs) with a sub-200 CD4 count in France [1]. Total darunavir minimum concentration (Cmin) fell from week 2 to week 12 despite good adherence, a finding suggestion autoinduction of darunavir metabolism. Most study participants in both groups reached and maintained a viral load below 50 copies.
About one third of people diagnosed with HIV in France have a CD4 count below 200. Ritonavir-boosted PIs are often prescribed for people with advanced HIV infection, and French national guidelines recommend atazanavir/ritonavir or darunavir/ritonavir as preferred PIs for antiretroviral-naive people. Researchers at Hopital Bichat in Paris and collaborators at other centers conducted this study to assess the pharmacokinetics of once-daily atazanavir/ritonavir (300/100 mg) and darunavir/ritonavir (800/100 mg) with two nucleosides (either tenofovir/emtricitabine or abacavir/lamivudine) in antiretroviral-naive adults with a CD4 count below 200.
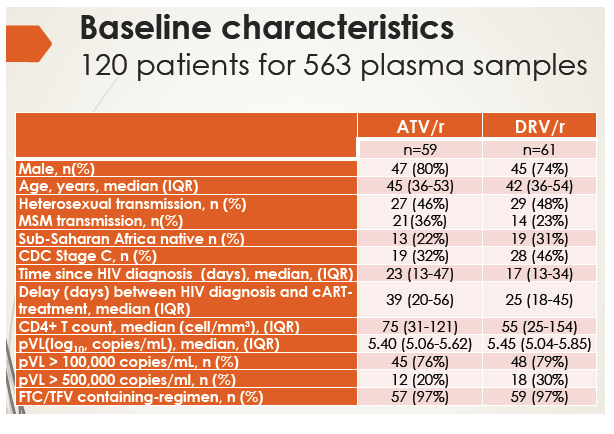
In this randomized noncomparative, 48-week study, researchers measured both total and unbound antiretroviral Cmin 24 hours after dosing at weeks 2, 4, 12, 24, 36, and 48 in 59 people starting atazanavir/ritonavir and 61 starting darunavir/ritonavir. Median ages in the atazanavir and darunavir groups were 45 and 42, and 80% and 74% were men. Proportions of black Africans were 22% in the atazanavir group and 31% in the darunavir group. Respective median times between HIV diagnosis and therapy were 39 and 25 days. Median CD4 count stood at 75 in the atazanavir group and 55 in the darunavir group, and three quarters had a pretreatment viral load above 100,000 copies. Almost everyone took tenofovir/emtricitabine.
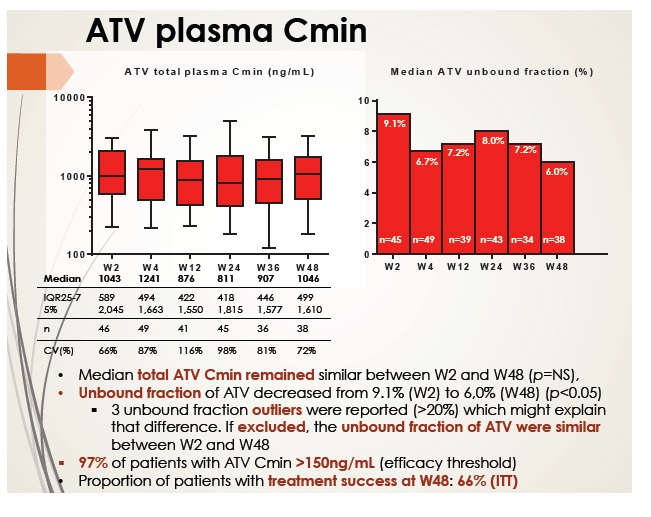
Median total atazanavir Cmin remained stable through 48 weeks of treatment, with Cmin exceeding the efficacy cutoff of 150 ng/mL in 97% of patients. Median unbound atazanavir fraction dropped from 9.1% at week 2 to 6.0% at week 48. But when the analysis excluded outliers (all with unbound fractions above 20%), the unbound fraction difference between weeks 2 and 48 disappeared.
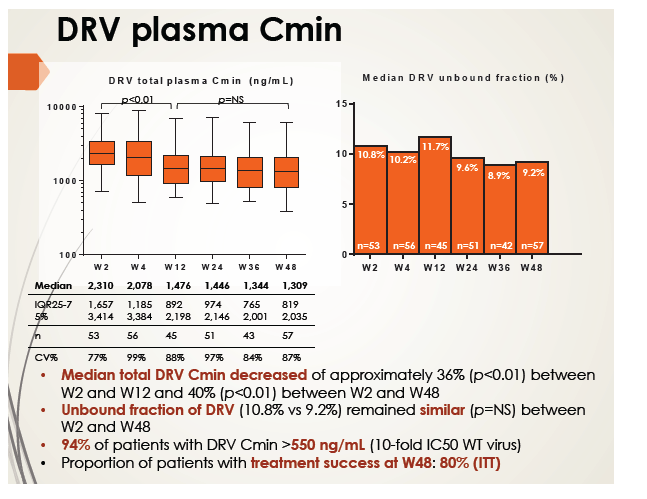
Median total darunavir Cmin fell approximately 36% between week 2 and week 12 (P < 0.01) and about 40% from week 2 to week 48 (P < 0.01). But most patients (94%) maintained a darunavir Cmin above the efficacy cutoff of 550 ng/mL, and the unbound darunavir fraction did not vary much between week 2 and week 48 (10.8% and 9.2%). The researchers called the drop in total darunavir Cmin "difficult to explain" in light of good overall adherence, but they suggested it could reflect autoinduction of darunavir metabolism.
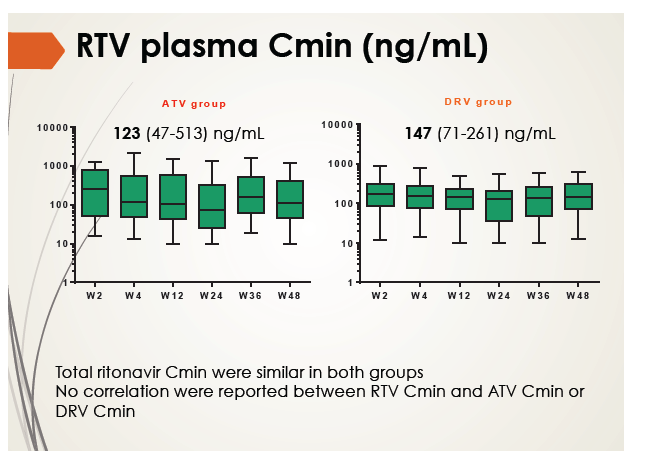
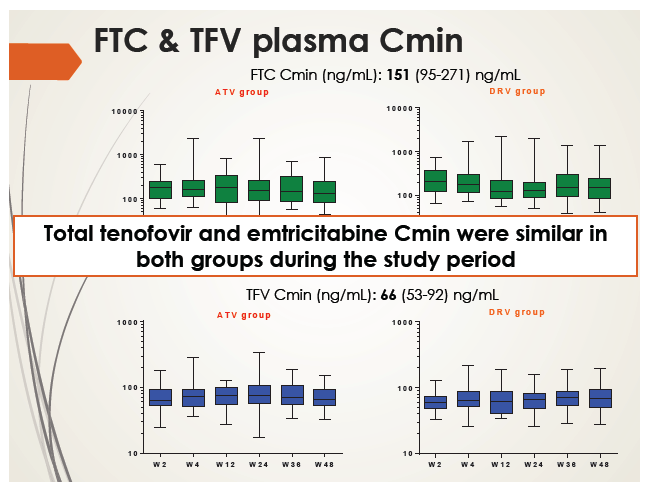
Ritonavir Cmin was similar in the atazanavir and darunavir groups and did not change substantially through 48 weeks. Total tenofovir and emtricitabine Cmin were also similar in the two study groups and remained stable over 48 weeks. Tenofovir and emtricitabine concentrations were consistent with historical data.
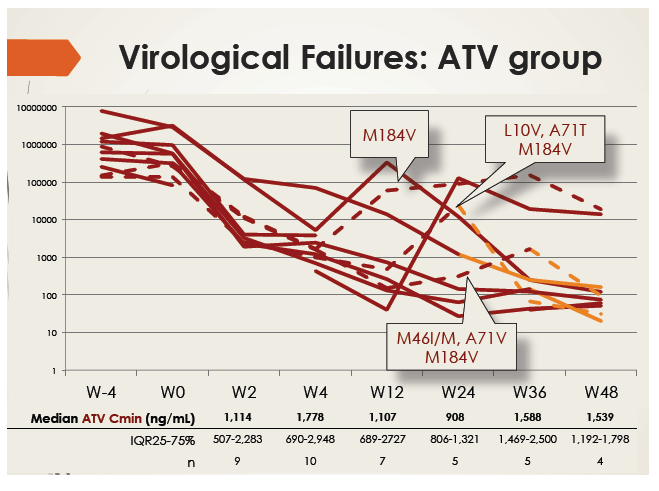
In an intention-to-treat analysis, 66% of patients in the atazanavir group had an undetectable viral load at week 48. Among the 10 people with virologic failure, 3 reached a viral load below 50 copies and rebounded without emergence of resistance mutations. Two people with atazanavir-related mutations switched to darunavir. All of these patients had adequate atazanavir Cmins. All but 2 patients with virologic failure maintained a viral load below 400 copies, and those 2 had poor adherence.
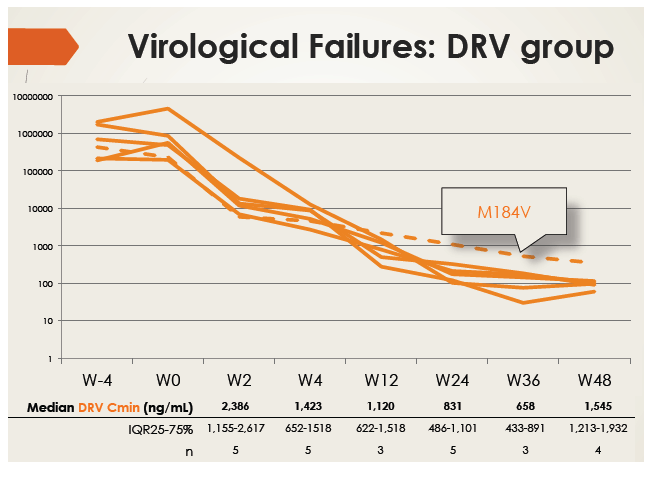
Intention-to-treat analysis rated 80% in the darunavir group as treatment successes at week 48. Among 6 patients who failed to reach the virologic endpoint, 1 did attain a viral load below 50 copies but rebounded. Five of the 6 maintained a viral load around 100 copies, and the sixth had a detectable emtricitabine-related M184V mutation. No one with virologic failure had a darunavir Cmin below the efficacy threshold of 550 ng/mL.
Reference
1. Le M, Slama L, Assoumou L, et al. Pharmacokinetics of once-daily ritonavir boosted atazanavir or darunavir in combination with a dual nucleos(t)ide analogs backbone among HIV-1-naive patients with severe immunosuppression: IMEA 040-DATA sub-study. 15th European AIDS Conference, October 21-24, 2015, Barcelona. Abstract PS9/6.
----------------
Reported by Jules Levin







|
| |
|
 |
 |
|
|