 |
 |
 |
| |
HIV-1-RNA Decay and Dolutegravir Concentrations in
Semen of Patients Initiating a First Antiretroviral Regimen
|
| |
| |
Reported by Jules Levin
EACS 2015 Oct 21-25 Barcelona, Spain
Arkaitz Imaz1, Javier Martinez-Picado2,3, Jordi Niubo4, Angela D. M. Kashuba5, Elena Ferrer1, Nerea Rozas1, Laura Acerete1, Antonia Vila1, Dan Ouchi2, Craig Sykes5, Daniel Podzamczer1
1. HIV and STD Unit, Department of Infectious Diseases, Hospital Universitari de Bellvitge, L'Hospitalet de Llobregat, Barcelona, Spain
2. AIDS Research Institute IrsiCaixa, Institut d'Investigacio en Ciencies de la Salut Germans Trias i Pujol, Universitat Autònoma de Barcelona, Badalona, Spain
3. Institucio Catalana de Recerca i Estudis Avançats (ICREA), Barcelona, Spain
4. Department of Microbiology, Hospital Universitari de Bellvitge, L'Hospitalet de Llobregat, Barcelona, Spain
5. UNC Center for AIDS Research, UNC Eshelman School of Pharmacy, University of North Carolina at Chapel Hill, Chapel Hill, NC, USA

Program Abstract
HIV-1-RNA Decay and Dolutegravir Concentrations in Semen of Patients Initiating a First Antiretroviral Regimen
A. Imaz1, J. Martinez-Picado2,3, J. Niubo4, A.D. Kashuba5, E. Ferrer1, N. Rozas1, L. Acerete1, A. Vila1, D. Ouchi2, C.S. Sykes5, D. Podzamczer1
1Hospital Universitari de Bellvitge, HIV and STD Unit, Department of Infectious Diseases, L'Hospitalet de Llobregat, Spain, 2AIDS Research Institute IrsiCaixa, Institut d'Investigacio en Ciencies de la Salut Germans Trias i Pujol, Universitat Autònoma de Barcelona, Badalona, Spain, 3Institucio Catalana de Recerca i Estudis Avançats (ICREA), Barcelona, Spain, 4Hospital Universitari de Bellvitge, Department of Microbiology, L'Hospitalet de Llobregat, Spain, 5University of North Carolina, UNC Center for AIDS Research, UNC Eshelman School of Pharmacy, Chapel Hill, United States
Objectives: To quantify HIV-1 RNA decay and dolutegravir (DTG) concentrations in semen of HIV-1 infected patients initiating antiretroviral therapy (ART) with DTG plus abacavir/lamivudine (ABC/3TC).
Methods: Prospective, single-arm, open-label study including 15 HIV-1 infected, ART-naïve men initiating DTG+ABC/3TC. HIV-1 RNA was measured in paired seminal (SP) and blood plasma (BP) samples at baseline, days 3, 7, and 14, and weeks 4, 12 and 24. The HIV-1 RNA decay rate in plasma and seminal fluid was assessed using nonlinear mixed-effects models. Total DTG concentration at the end of the dosing interval (C24h) was quantified in in SP and BP at weeks 4 and 24 by a validated liquid chromatography-tandem mass spectrometry method.
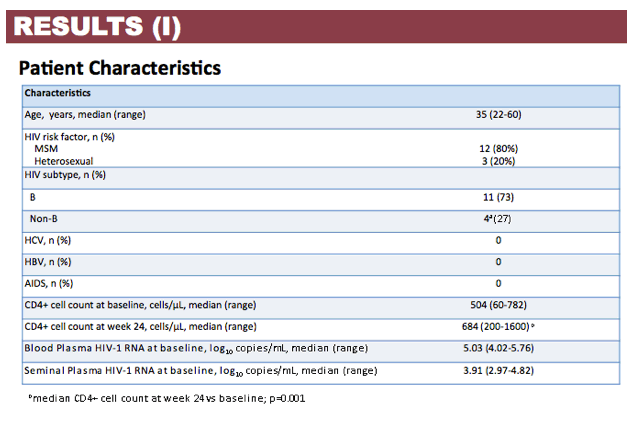
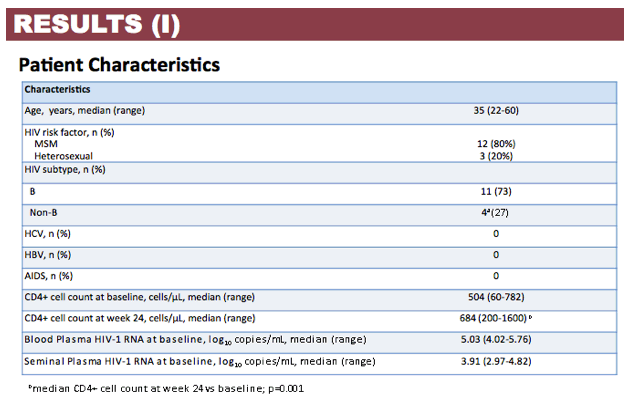
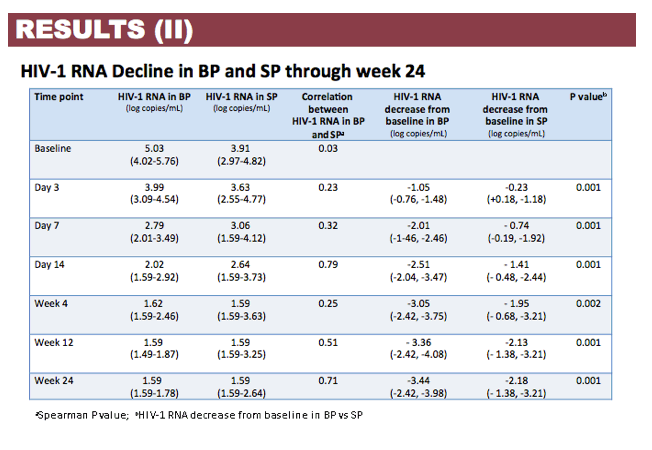
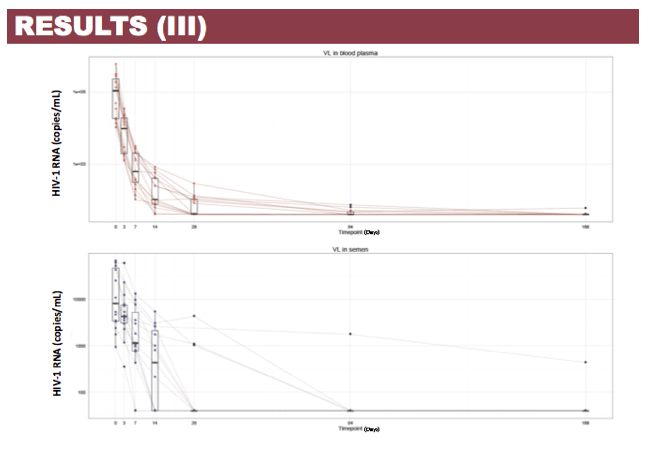
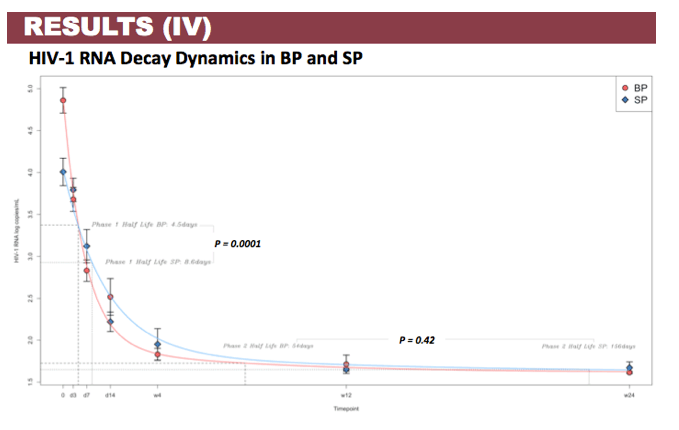
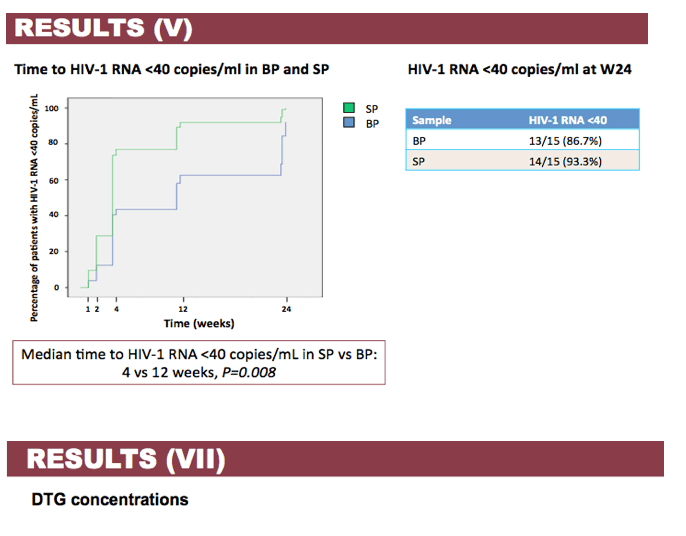
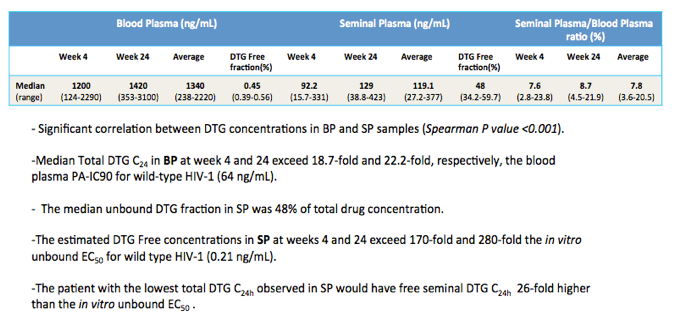
Results: Patient characteristics at baseline were: Median (range) age 35 (22-60) years; CD4+ cell count 504 (60-782) cells/µL; BP HIV-1 RNA 5.03 (4.02-5.76) log copies/mL; SP HIV-1 RNA 3.91 (2.97-4.82) log copies/mL; and HIV-1 subtype B and non-B in 11 and 4 patients, respectively, with no differences in median HIV-1 RNA in BP and SP according to B or non-B HIV-1 subtype. Viral decay was faster in BP than in SP in the first decay phase (half-life 4.5 vs 8.6 days, p=0.0001) but differences in the second decay phase were not statistically significant (54 vs 156 days, p=0.43). Median time to HIV-1 RNA < 40 copies/mL was significantly shorter in SP than BP (4 vs 12 weeks, p=0.008) with no differences when adjusted by baseline HIV-1 RNA. Overall, median DTG C24 in SP was 119.1 (27.2-377) ng/mL, which was 7.8% (3.7-21%) of the BP C24. The lowest seminal DTG concentration observed was 78-fold the in vitro IC50 (0.2 ng/mL).
Conclusion: Although the DTG concentration in SP was only 7.8% of the BP concentration, it sufficed to contribute to achieving rapid suppression of HIV-1 replication in this compartment.






|
| |
|
 |
 |
|
|