 |
 |
 |
| |
No Evidence of Pharmacokinetic Drug-Drug Interaction in Healthy Subjects Between Coadministered Grazoprevir (MK-5172)/Elbasvir (MK-8742) and Sofosbuvir
|
| |
| |
Reported by Jules Levin
EASL 2015 April 22-26 Vienna Austria
William L. Marshall*1; Wendy W. Yeh1; Crystal Bethel-Brown2; Daria Stypinski2; Patrice Auger2; Christine Brandquist2; Dana Gill2; John Brejda2; Luzelena Caro1; Patricia Jumes1; Xiaobi Huang1; Zifang Guo1; Monika Martinho1; H-P Feng1; Danielle Armas2; Marian Iwamoto1; Joan R. Butterton1
1Merck & Co., Inc., Kenilworth, NJ, USA; 2Celerion, Tempe, AZ, USA
EASL 2015 was Merck's debutant ball for their phase 3 study results and FDA approval is expected later this year. Merck acquired nuke polymerase from Idenix, similar class as sofosvuir (Sovaldi), MK3682 is one of them:
Merck Phase 3 clinical trials evaluating grazoprevir/elbasvir, C-EDGE program, - (04/28/15)
A Phase 1 Study to Evaluate the Interaction of HCV NS5B Inhibitor MK-3682 With HCV NS3/4A Protease Inhibitor MK-5172 and HCV NS5A Inhibitor MK-8408 in Healthy Subjects - (04/23/15)


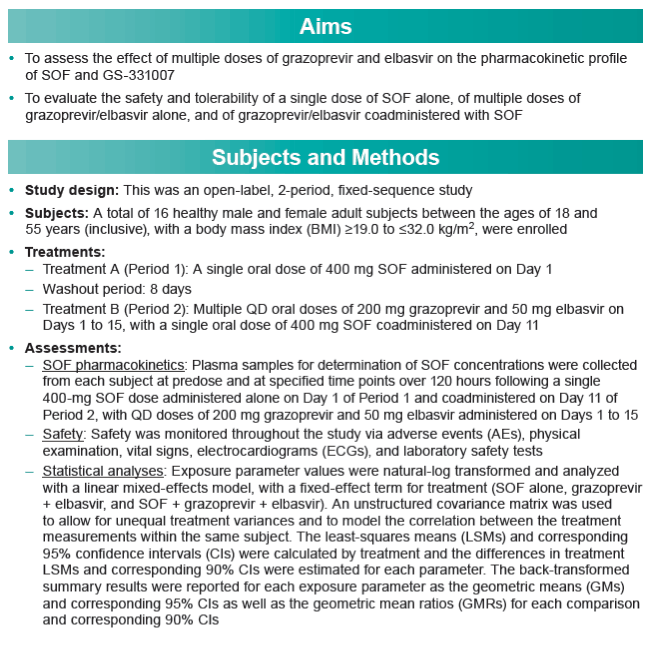

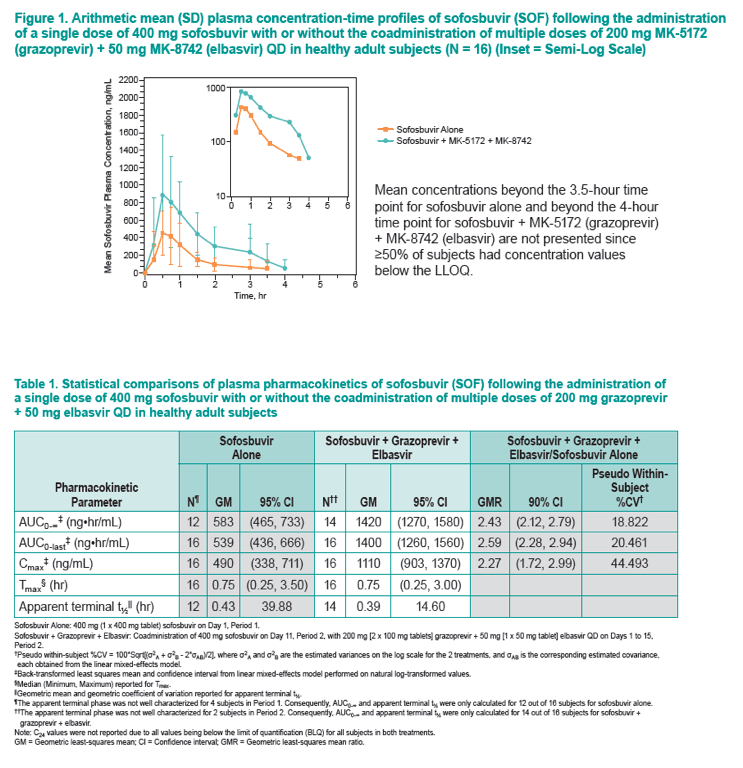
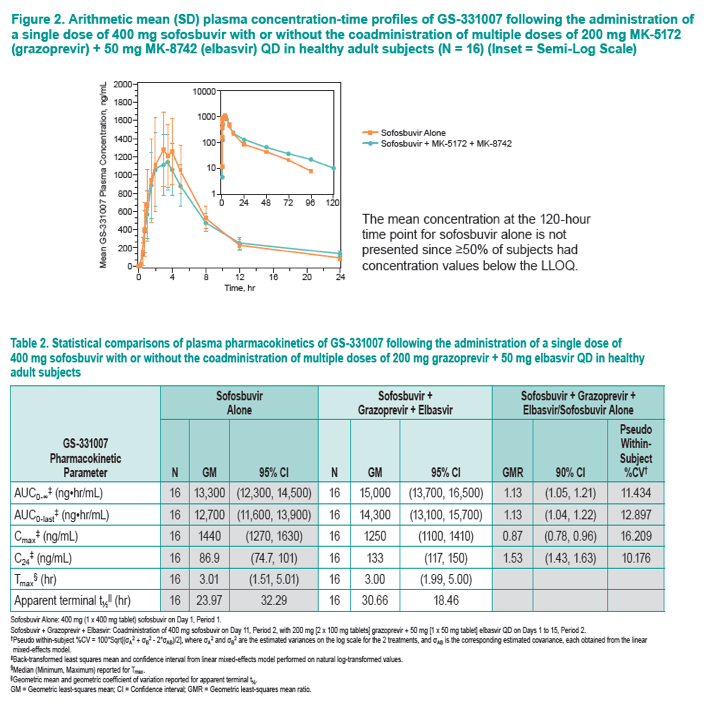
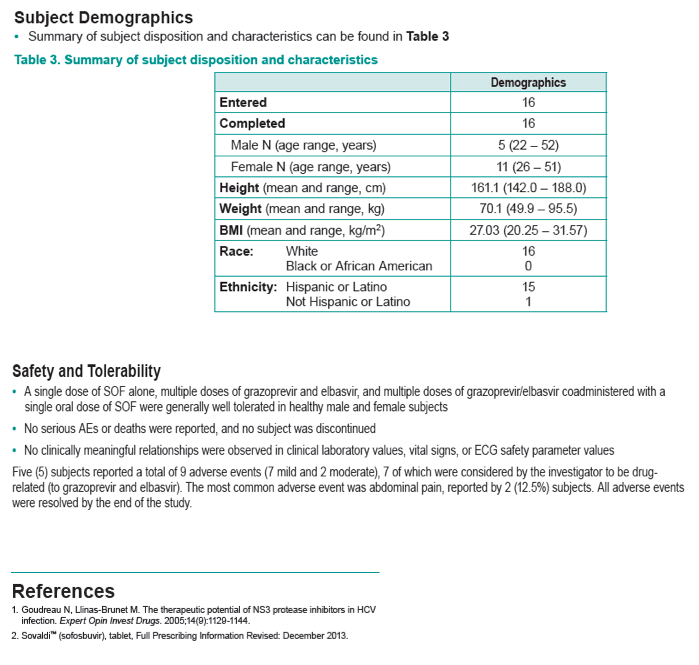
|
| |
|
 |
 |
|
|