 |
 |
 |
| |
NS5A Mutations/Resistance
|
| |
| |
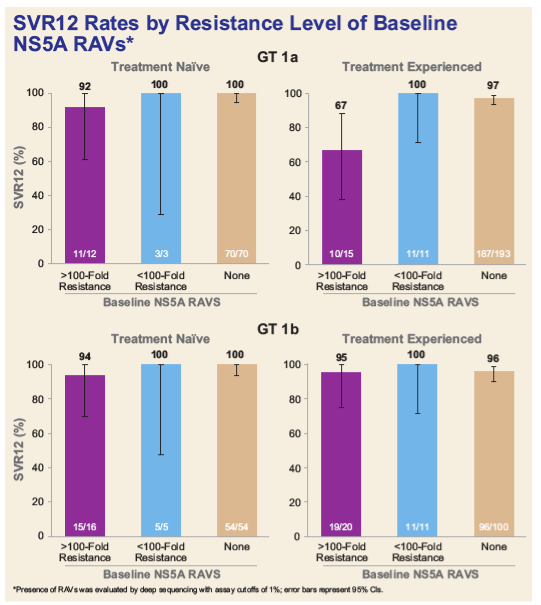
from Jules: despite the presence of NS5A mutations at baseline before treatment apparently very few patients fail due to the mutations, it appears >1000 fold NS5A resistance shows the greatest signal for failure, 100-1000 fold may signal a risk depending on the duration of NS5A therapy, and apparently <100 fold is not a risk look below from a study at EASL you will see high SVR rates despite >100 fold resistance. There is a Monogram NS5A resistance test available. Adding a 3rd drug like a protease is being studied by Gilead, adding protease GS9857 to 2 other HCV drugs, could be SOF+LDV or SOF+GS5816, and 2nd generation NS5A inhibitors are in development that may also suppress NS5A resistance: ACH3102, MK8408, ABT530, GS5816. EASL: New HCV Drugs new nukes-NS5A/Protease Inhibitors - (05/08/15) or if insurance would cover it you could add a currently available protease inhibitor like Simeprevir to Harvoni, but this has not been studied.
EASL: Resistance Analysis of Virologic Failures in Hepatitis C Genotype 1-Infected Patients Treated With Grazoprevir + Elbasvir ± Ribavirin: the C-WORTHY Study - (05/04/15)
EASL: Effect of Baseline Factors on Response to the Fixed-Dose Combination Of Daclatasvir, Asunaprevir and Beclabuvir in Non-Cirrhotic Patients with HCV Genotype 1 Infection - (05/05/15)
EASL: LONG-TERM FOLLOW-UP OF TREATMENT-EMERGENT RESISTANCE-ASSOCIATED VARIANTS IN NS3, NS5A AND NS5B WITH PARITAPREVIR/r-, OMBITASVIR- AND DASABUVIR-BASED REGIMENS - (04/24/15)
EASL: Long-Term Persistence of HCV NS5A Variants After Treatment With NS5A Inhibitor Ledipasvir - (04/29/15)
EASL: Prevalence of Pretreatment NS5A and NS5B Resistance-Associated Variants and Genetic Variation Within HCV Subtypes Across Different Countries/Gilead-Study - (04/30/15)
EASL: Long-Term Persistence of HCV NS5A Variants After Treatment With NS5A Inhibitor Ledipasvir - (04/29/15)
EASL: Retreatment of Patients Who Failed 8 or 12 Weeks of Ledipasvir/Sofosbuvir-Based Regimens With Ledipasvir/Sofosbuvir for 24 Weeks - (04/24/15)
EASL: The Prevalence and the Effect of HCV NS5A Resistance-Associated Variants in Patients With Compensated Cirrhosis Treated With Ledipasvir/Sofosbuvir ± RBV - (04/29/15)
EASL - The International Liver Congress 2015 50th Annual Meeting of the European
Association for the Study of the Liver Vienna, Austria April 22-26
|
| |
|
 |
 |
|
|