 |
 |
 |
| |
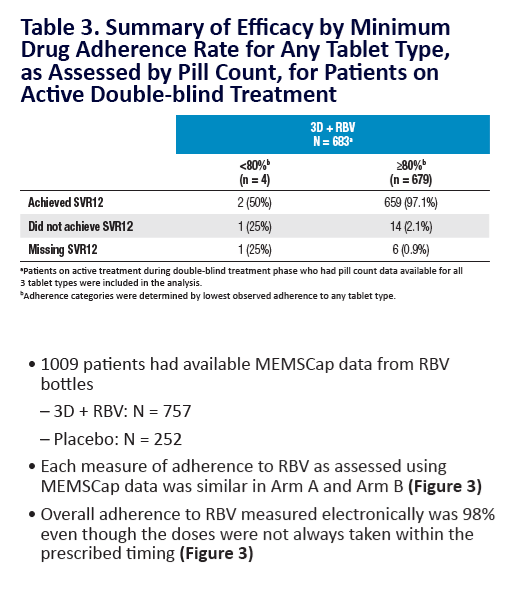
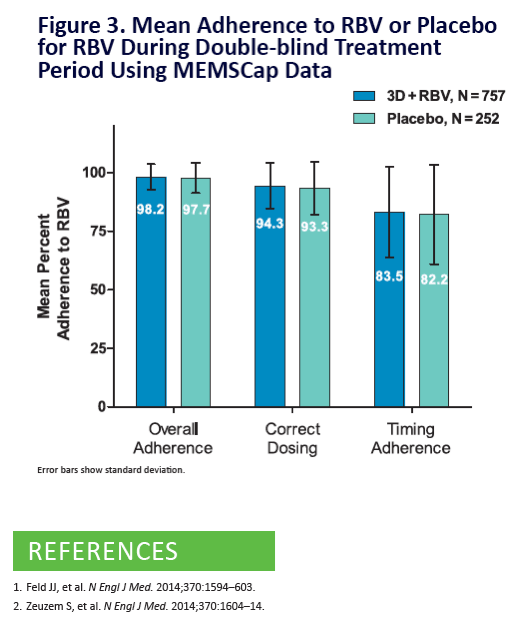
Adherence to Ombitasvir/Paritaprevir/r, Dasabuvir, and
Ribavirin is >98% in the SAPPHIRE-I and SAPPHIRE-II Trials
|
| |
| |
Reported by Jules Levin
EASL 2015 April 22-26 Vienna Austria
Tarek Hassanein1, Stuart Roberts2, Stephen D Shafran3, Mihály Makara4, Michael Bennett 5, Beat Müllhaupt6, Fred Poordad7, Stephen Ryder8, Adrian M Di Bisceglie9, Eoin Coakley10, Bo Fu10, Marisol Marti nez-Tristani10, Graham R Foster 11
1Southern California Liver Centers and Southern California Research Center, Coronado, California, United States; 2The Alfred Hospital, Melbourne, Victoria, Australia;
3University of Alberta Hospital, Edmonton, Alberta, Canada; 4Saint Laszlo Hospital, Budapest, Hungary; 5Medical Associates Research Group, San Diego, California, United States; 6University Hospital, Zurich, Switzerland; 7The Texas Liver Insti tute/University of Texas Health Science Center, San Antonio, Texas, United States; 8Notti ngham Digesti ve Diseases Centre and Biomedical Research Unit, Notti ngham, United Kingdom; 9Saint Louis University Liver Center, Saint Louis University, St. Louis, Missouri, United States; 10AbbVie Inc., North Chicago, Illinois, United States; 11Queen Marys University of London, Barts Health, London, United Kingdom
EASL: Abbvie at EASL 2015 - Healthcare Economics, Clinical Trials - (05/11/15)



|
| |
|
 |
 |
|
|