 |
 |
 |
| |
Summary from EASL 2015 for Hepatitis C
All oral HCV DAA therapy on its way to optimization: still much to learn.
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Introduction
The introduction of all oral DAA based HCV therapy has dramatically changed HCV treatment paradigms and promises HCV cure in more than 95% of patients. Nevertheless special challenging patient populations remain which until now have only scarcely been addressed such as patients with renal impairment or on hemodialysis. At this year EASL results from larger treatment trials in exact that patient population were presented for the first time. Other more challenging patient groups addressed included patients with decompensated cirrhosis and patients post-transplant. Considering that not all patients with decompensated cirrhosis benefit from HCV therapy despite becoming HCV-RNA negative, the question arises whether there may be a point of no return for very advanced liver disease stages. Also for patients on the transplant list the question remains whether decreases in MELD score following HCV cure may impact the position on the organ waiting list, delay transplantation considerably and thereby increase risk for HCC development while on the waiting list.
Furthermore, new data on treatment of HCV genotype 3 were eagerly awaited as there are still many questions about what the most effective treatment strategy may be in this patient group, particularly in the treatment-experienced cirrhotics. Obviously this goes hand in hand with the quest for developing new HCV pan-genotypic agents, which would also simplify diagnostic work-up prior to HCV treatment initiation. Finally, large interest was generated around feedback from all oral DAA treatment in compassionate use programs and real-life cohorts in order to better understand the risk of virological failure in real-life clinical settings. Also the question remains which impact emergence of DAA resistance associated variants may have after failing all oral DAA therapy and whether there is a need for genotypic resistance testing at least after failure of DAA therapy. Finally the new set of EASL guidelines for management of hepatitis C was presented at the conference publically for the first time. The following review aims at summarizing the findings from the main HCV studies presented at EASL. However, due to the very high number of papers being presented this overview is restricted to the personally conceived highlights.
New EASL guidelines presented: what is important?
The goal of HCV therapy is to cure HCV infection in order to prevent the complications of HCV-related liver and extra-hepatic diseases, including hepatic necro-inflammation, fibrosis, cirrhosis, hepatic decompensation, hepatocellular carcinoma (HCC), severe extrahepatic manifestations and death (1). The endpoint of therapy is defined as an undetectable HCV RNA in a sensitive assay (<15 IU/mL) 12 weeks (SVR12) and 24 weeks (SVR24) after the end of treatment. All treatment-naïve and treatment-experienced patients with compensated or decompensated chronic liver disease related to HCV, who are willing to be treated and who have no contraindications to treatment, should be considered for therapy. In clear appreciation of faster liver fibrosis progression as well as higher clinical disease burden in more advanced fibrosis stages EASL has provided a helpful table which lists patient groups which can be considered for treatment prioritization (see Table 1).
Table 1: Treatment prioritization within the EASL guidelines (1)
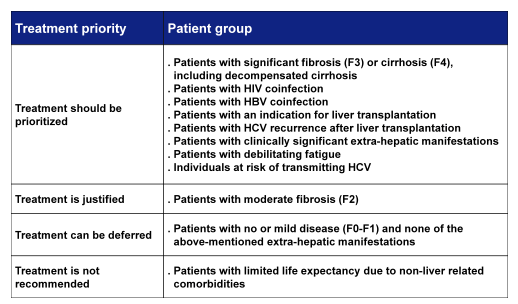
Interestingly, the need for prioritizing patients with HIV is newly listed here which is well in line with the current AASLD/IDSA guidelines which also sees a higher risk for hepatic decompensation and faster fibrosis progression in this particular patient group. It appears however, that if ideally all HIV/HCV coinfected patients would receive HIV therapy early (latest when CD4 drops below 500/μl) this more unfavorable course of liver disease could be delayed or even reverted to the same course as in HCV mono-infected subjects. The other important message with regard to coinfection is that HCV treatment in HCV/HIV co-infected persons should be identical to those in patients with HCV mono-infection. However, potential drug-drug interactions with antiretroviral drugs should be taken into account.
The current recommended treatment options by genotype are shown in figure 1.
Figure 1: Treatment options 2015 in the EASL recommendations
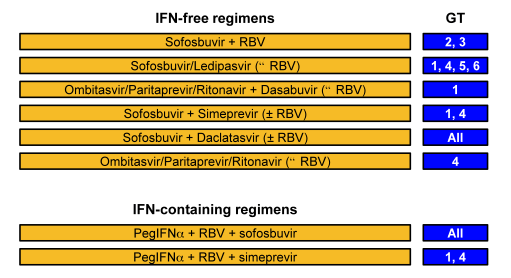
Depending on availability mostly IFN-free all oral DAA combinations are recommended but few IFN-based regimens remain in case other HCV treatments are not yet readily available. PEG-IFN/RBV + sofosbuvir may still play a role in treatment of genotype 3 as IFN-free therapy of this genotype remains challenging (see also section on HCV therapy in genotype 3 patients on page 18).
EASL: EASL Recommendations on Treatment of Hepatitis C 2015 - (04/22/15)
HCV therapy in renal impairment or hemodialysis
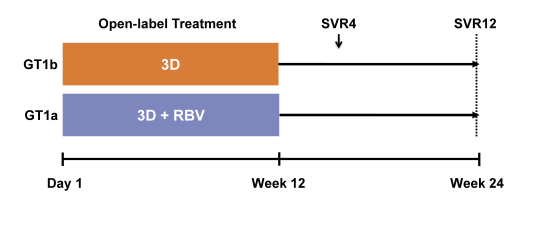
At this year EASL eagerly awaited HCV treatment data was presented in patients with severe renal impairment or already on haemodialysis. HCV infection is frequently found in this patient population but so far use of HCV drugs has been difficult, particularly if they are renally excreted. Ribavirin for example could only be administered in very low doses because of accumulation with decreased GFR and significantly enhanced toxicities. Sofosbuvir is contraindicated in patients with a GFR<30 ml/min according to the package insert. In some countries such as Canada kidney transplant in a HCV-infected haemodialysis patient is only possible after achieving SVR under HCV therapy again underlining the urgent need of developing better tolerated and more successful treatment strategies for this patient population. A first study was presented which examined safety and efficacy of the Abbvie 3-D regimen (Co-formulated ombitasvir and low dose ritonavir boosted paritaprevir (OBV/PTV/r) (25/150/100 mg QD) and dasabuvir (DSV) (250 mg BID) in non-cirrhotic HCV genotype 1 patients with chronic kidney disease with estimated glomerular filtration rate (eGFR) <30 mL/min/1.73 m2 (2). This open-label phase 3b study was performed in the US at 9 different sites. Patients with genotype 1a received additional 200mg ribavirin (in patients on hemodialysis RBV was dosed 4 hours prior to start of hemodialysis) whereas patients with HCV genotype 1b were treated without ribavirin. The study design is depicted in figure 2.
Figure 2: Study design of the RUBY trial
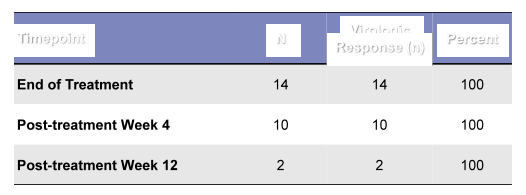
In this ongoing trial 20 patients were included, 14/20 completed 12 weeks of 3D ± RBV whereas 6/20 still remain on treatment. 85% of patients were black, 70% black, 65% were on hemodialysis, median age was 60y (range 49-69) and 20% had F3 fibrosis. The study results are shown below in figure 3. As most patients have not yet reached post-treatment week 12 conclusions on efficacy are limited. Nevertheless, to date all patients completing treatment had a good virologic response so far.
Figure 3: RUBY-1 efficacy results
EASL: SAFETY OF OMBITASVIR/PARITAPREVIR/RITONAVIR PLUS DASABUVIR FOR TREATING HCV GT1 INFECTION IN PATIENTS WITH SEVERE RENAL IMPAIRMENT OR END-STAGE RENAL DISEASE: THE RUBY-I STUDY - (04/27/15)
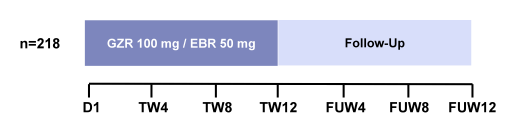
The largest study presented at EASL in the renally impaired patient population was the C-SURFER study (3). In this randomized, parallel-group, multisite, placebo-controlled trial 224 patients were randomized to immediate treatment (ITG) with grazoprevir/elbasvir (GZR/EBR) or deferred treatment (DTG) where patients received placebo for 12 weeks then open-label GZR/EBR starting at follow-up week (FW) 4. Patients were stratified by diabetes (yes/no) and hemodialysis status: hemodialysis or non-hemodialysis, 11 patients in an open-label GZR/EBR arm underwent intensive pharmacokinetic (PK) sampling. All patients on inclusion to the study had a CrCl <30 mL/min, including patients on hemodialysis. Earlier studies had already demonstrated that <1% of GZR and EBR are renally excreted and that there is no need for dose adjustments in chronic kidney disease (CKD) (4). Patients who were included all had HCV GT1 infection. The study allowed the inclusion of treatment-naive and treatment-experienced patients. The study design is depicted in figure 4, the baseline characteristics are shown in figure 5.
Figure 4: Study design of the C-SURFER study
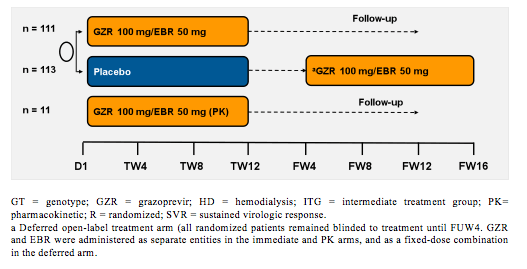
GT = genotype; GZR = grazoprevir; HD = hemodialysis; ITG = intermediate treatment group; PK= pharmacokinetic; R = randomized; SVR = sustained virologic response.
a Deferred open-label treatment arm (all randomized patients remained blinded to treatment until FUW4. GZR and EBR were administered as separate entities in the immediate and PK arms, and as a fixed-dose combination in the deferred arm.
Figure 5: Baseline characteristics of the C-SURFER trial
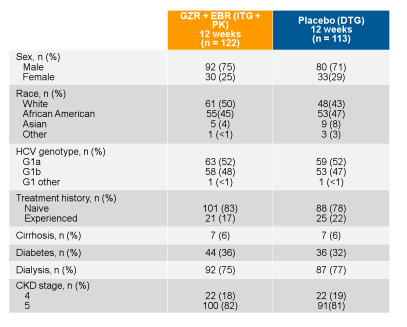
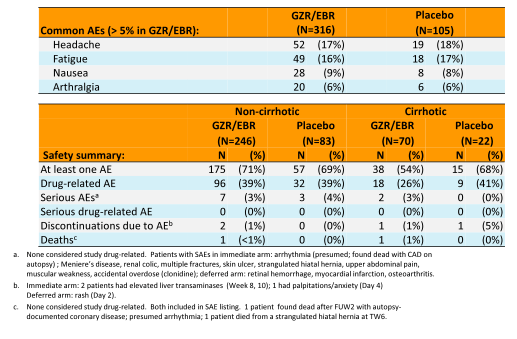
Overall, there was good racial distribution. Majority of patients had a genotype 1a infection and were treatment naïve. Only few patients were cirrhotic (6%) but many had additional comorbidities in particular diabetes. The figure 6 summarizes the main virological outcome of the trial. In the intent-to-treat analyses an impressive 94% of study participants achieved a SVR12.
Figure 6: SVR12: immediate treatment group (ITG).
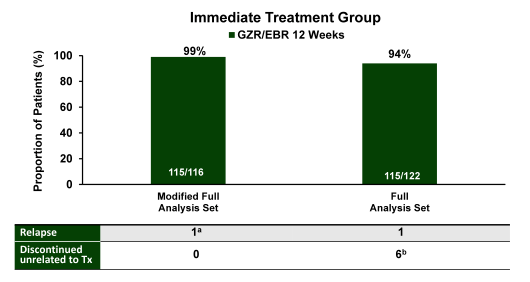
MFAS = primary efficacy analysis; Tx = treatment. FAS was a secondary analysis
aNoncirrhotic, interferon-intolerant patient with HCV GT1b infection relapsed at FW12.
bLost to follow-up (n = 2), n = 1 each for death, noncompliance, withdrawal by subject, and withdrawal by physician (owing to violent behavior).
Clearly the cure rates are excellent even if the number of cirrhotics included was low. This IFN and RBV free treatment option promises great hope for therapy in renally impaired patients where so far treatment options still have been very limited and at least opens the discussion about initiating compassionate use programs for the grazoprevir/elbasvir combination in this patient population as this would also open up chances for kidney transplantation in some countries.
The overall safety of the HCV therapy was very good with 0 discontinuations of study drug due to adverse events. Adverse events such as headache, nausea, diarrhea etc. were equally distributed between patients receiving study drug versus placebo.
EASL: C-SURFER: GRAZOPREVIR PLUS ELBASVIR IN TREATMENT-NAIVE AND TREATMENT-EXPERIENCED PATIENTS WITH HEPATITIS C VIRUS GENOTYPE 1 INFECTION AND CHRONIC KIDNEY DISEASE - (04/23/15)
AASLD/2014: The Pharmacokinetics of Ledipasvir, an HCV-Specific NS5A Inhibitor, in HCV-Uninfected Subjects With Severe Renal Impairment - (12/15/14)
AASLD/2014: Safety, Antiviral Efficacy, and Pharmacokinetics of Sofosbuvir in Patients With Severe Renal Impairment - (12/03/14)
HCV therapy in decompensated cirrhosis or post-transplant: are we improving outcome?
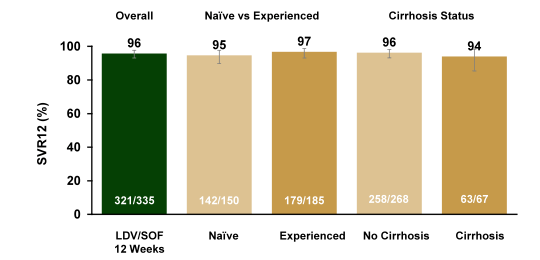
Various studies were reported at EASL which evaluated efficacy and safety of all oral DAA therapy in the so perceived more challenging patient groups namely advanced cirrhosis and post-transplant patients. Clearly the arising treatment options in this segment of patients has led to quite a range of clinically very important questions. Do we need ribavirin in these more difficult to treat patients? Are there special safety concerns with ribavirin or DAA therapy in this very sick patient group? Is there a point of no return where HCV cure has no impact on further disease outcome and can potential benefits in MELD score after successful HCV therapy impact chance of liver organ allocation? One new study presented in this patient group was the SOLAR-2 study in GT 1 or 4 treatment-naïve or -experienced patients with either decompensated liver disease or post-transplant which received 12 or 24 weeks of therapy with sofosbuvir/ledipasvir + ribavirin. Patients were randomized to receive 12 or 24 weeks of treatment stratified into 6 groups: patients without transplant and either (1) Child-Pugh-Turcotte (CPT) B cirrhosis, or (2) CPT C cirrhosis; or patients with recurrent HCV after liver transplantation and were either (3) without cirrhosis (F0 to F3), (4) CPT A cirrhosis, (5) CPT B cirrhosis, or (6) CPT C cirrhosis. In patients with Metavir F0-F3 and CPT A cirrhosis ribavirin was dosed weight-based (1000 or 1200 mg/day) whereas in CPT B and C cirrhosis patients received 600 mg/day with subsequent dose escalation. The study design is shown below in figure 7.
Figure 7: Study design of the SOLAR-2 study
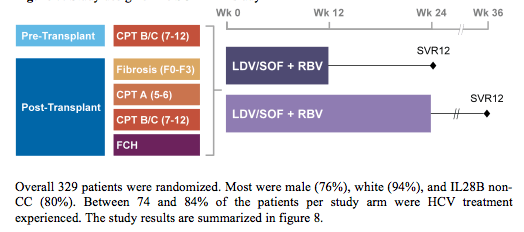
Overall 329 patients were randomized. Most were male (76%), white (94%), and IL28B non-CC (80%). Between 74 and 84% of the patients per study arm were HCV treatment experienced. The study results are summarized in figure 8.
Figure 8: SOLAR-2: SVR12 Results (GT1 and GT4)
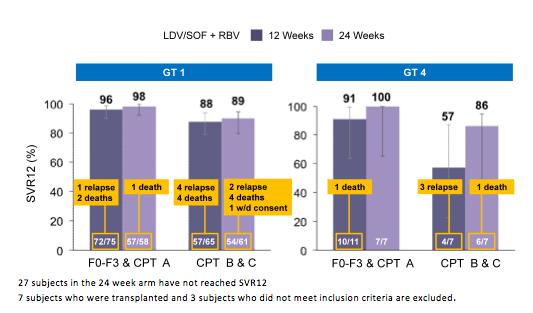
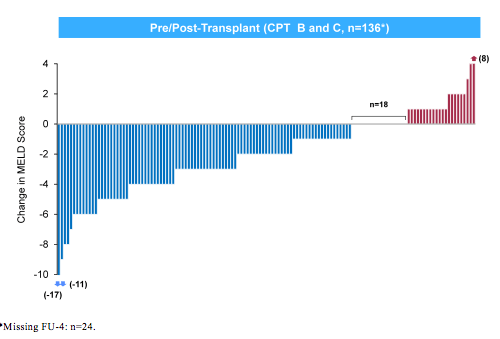
LDV/SOF + RBV resulted in high SVR12 rates in HCV patients with advanced liver disease, irrespective of transplantation status. For genotype 1 patients, no difference in SVR12 rates between 12 and 24 weeks was observed. There were too few genotype 4 patients for meaningful comparisons. The significant number of deaths observed raises the question about overall safety of DAA therapy in decompensated cirrhosis. First of all it is important to note that none of the deaths was reported as treatment related. However, it may be difficult to assess in how far deaths are attributed to the natural course of advanced liver disease or may reflect direct effects or toxicity from DAA therapy + ribavirin in this particular patient group. Clearly this underlines the need for cohort studies in the pre-transplant setting to better understand risk for death in these patients. Interestingly, as in previous studies in advanced cirrhosis most patients with CPT B or C show an improvement of MELD score under successful DAA therapy but there are again few patients who despite HCV therapy show no change or a worsening of their MELD score (see figure 9). Clearly this needs to be better understood to identify patients with potential risk of MELD worsening.
Figure 9: MELD Score Change From Baseline to Follow-up Week 4
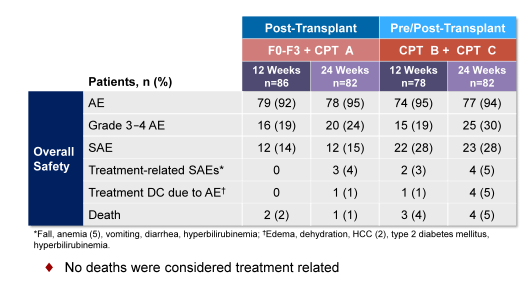
LDV/SOF + RBV for 12-24 weeks were generally safe and well tolerated in patients with advanced liver disease, pre and post liver transplantation. Only very few treatment discontinuations because of treatment related adverse events were noted. Figure 10 summarizes the main safety findings of the study.
Figure 10: Overall Safety Summary of the SOLAR-2 study
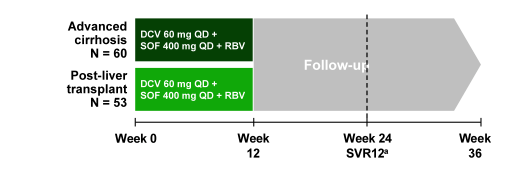
Another study in advanced cirrhosis or post-transplant was the ALLY-1 study which was an open-label study which enrolled treatment naïve or experienced adults with HCV infection of any genotype (GT) in 2 cohorts: (1) advanced cirrhosis, (2) post-liver transplant recurrence. Patients received 12 weeks of treatment with once-daily DCV 60mg + once-daily SOF 400mg and RBV (initially 600 mg/d, adjusted for hemoglobin and creatinine clearance). In the cirrhosis cohort, patients transplanted during treatment could receive 12 weeks of extended treatment immediately post-transplant regardless of treatment duration before transplant. The primary endpoint was HCVRNA < LLOQ (25 IU/mL) at post-treatment week 12 (SVR12) in patients with GT1 in each cohort. The study design is shown below in figure 11.
Figure 11: Study design of ALLY-1
The cirrhosis (N = 60) and post-transplant (N = 53) cohorts were, respectively, 40% and 42% treatment-naive and 75% and 77% GT1. The Child-Pugh class in the cirrhosis cohort was
20% A, 53% B, and 27% C. MELD score ranged from 8 to 27. No post-transplant patients had cholestatic recurrence or hepatic decompensation. The SVR12 rates are shown below in figure 12.
Figure 12: ALLY-1: SVR12 Results (by Cohort)
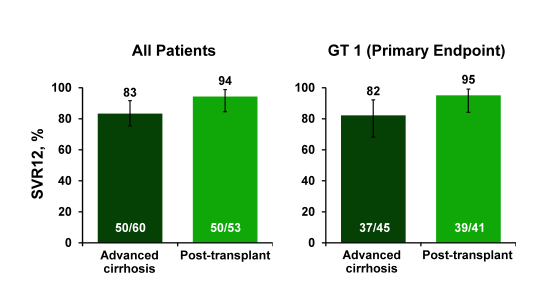
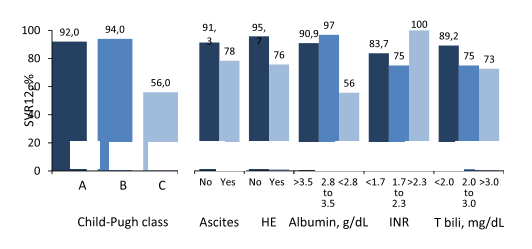
Overall, cure rates are higher in patients post-transplant than in advanced cirrhosis suggesting that HCV therapy in post-transplant patients seems no longer to be a challenging patient group. In a regression analysis, no difference by gender, age, IL28B, or HCV RNA was found in the advanced cirrhosis cohort with GT 1. Lower response rates were particularly seen for CHILD C patients and patients with low albumin levels (see figure 13).
Figure 13: SVR12 by Child-Pugh Class. Advanced cirrhosis cohort, all genotypes.
The combination of sofosbuvir and daclatasvir and ribavirin was overall well tolerated. The main safety findings are summarized in figure 14.
Figure 14: On-Treatment Safety and Tolerability
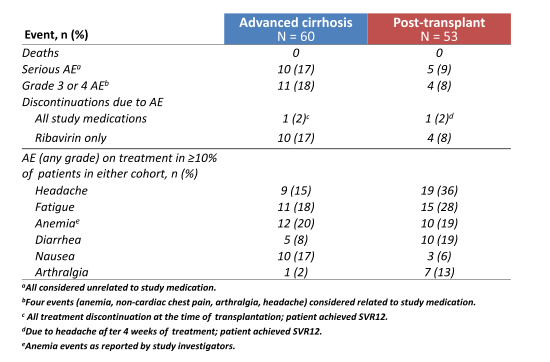
The large number of ribavirin discontinuations underlines the increased toxicity of ribavirin in the advanced fibrosis patient population.
In conclusion DCV+SOF+RBV for 12 weeks was safe and well tolerated in both cohorts. SVR12 rates were >90% in patients with Child-Pugh class A or B cirrhosis but lower in Child-Pugh class C. SVR12 was achieved by 94% of liver transplant recipients with HCV recurrence.
In addition to the randomized clinical trials data from real-life cohorts was also reported at EASL from patients with decompensated cirrhosis. Safety and efficacy of all oral HCV therapy in patients with decompensated cirrhosis participating in the ongoing multi-center HCVTARGET study was evaluated. In particular the combination of sofosbuvir+ ribavirin as well as the combination of sofosbuvir + simeprevir +/- ribavirin was analyzed. Clinical, adverse events and virologic data were collected throughout treatment and post-treatment follow-up between December 2013 and November 2014. The analyses were restricted to patients with cirrhosis and MELD score of ≥10 who had not undergone liver transplantation. The Safety Cohort consisted of patients who reached an end-treatment (EOT) time point while the Efficacy Cohort had a known virologic outcome (treatment failure or SVR4). Figure 15 summarizes the virological efficacy (SVR12) for genotype 1 patients with advanced cirrhosis.
Figure 15: HCV-TARGET: SVR12 Results - GT1
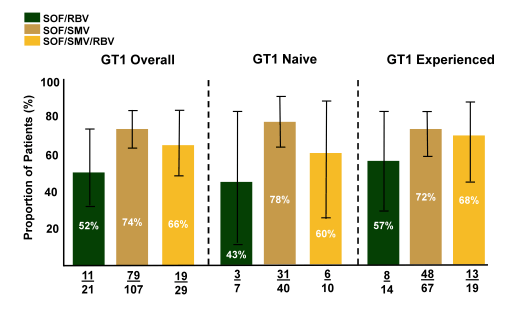
Lowest cure rates in this difficult-to-treat population were achieved with sofosbuvir + ribavirin underlining once again that dual DAA combination should be the way forward in more difficult to treat cirrhotic patients. But even combination of sofosbuvir/simeprevir was not achieving HCV cure in more than 74% of all HCV GT 1 patients emphasizing that in the real world outside of clinical trials the overall failure rate may be slightly higher than observed so far in trials. Addition of ribavirin did not seem to increase SVR rates but numbers were limited allowing no further conclusions.
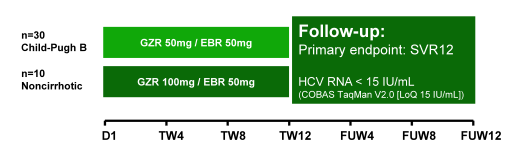
With regard to new DAA combinations data from the C-SALT study were presented (8). The purpose of the phase 2/3 C-SALT study (PN059) was to assess the efficacy, safety and pharmacokinetics (PK) of GZR plus EBR in patients with HCV G1, G4 or G6 infection and Child-Pugh (CP) class B cirrhosis. The phase 2 portion of C-SALT (part A) is described here.
CP-B patients with HCV infection received GZR (50mg QD) and EBR (50mg QD) for 12 weeks without RBV (see figure 16). Note that in this very advanced patient group a lower dose of 50 mg grazoprevir was studied. GZR and EBR were therefore administered as separate entities (FDC not used in C-SALT). The chosen dose of GZR 50 mg utilized in C-SALT, was modelled upon the exposure of GZR in non-HCV-infected CP-B patients. In C-SALT non-cirrhotic patients with HCV infection were enrolled for PK analyses and received GZR (100mg QD) plus EBR (50mg QD) for 12 weeks. Plasma samples for PK analyses were collected in a subset of CP-B and non-cirrhotic patients over 24 hours at TW4. The primary end point was SVR12 in CP-B patients (HCVRNA < LLoQ [15 IU/mL]). Overall, 30 CP-B and 10 non-cirrhotic patients were enrolled. Among the CP-B patients, 57% were male, 97% were white, 100% had G1 infection (G1a, 90%), mean MELD was 9.9 (SD, 2.5) and 63% were HCV treatment-naïve.
Figure 16: Study design of the C-SALT study
No patient had virologic breakthrough, had a treatment-related SAE, discontinued study drug, or had a grade 3 or 4 ALT elevation. Four patients had transient grade 3 total bilirubin elevations without increased ALT or AST. The main study outcome, SVR12 rates are shown below in figure 17.
Figure 17: C-SALT Part A: SVR12 Results
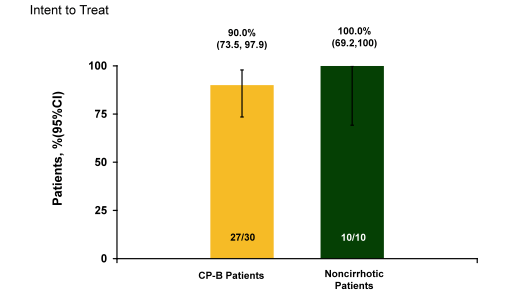
At FUW4 2 patients relapsed in the CP-B patient group and 1 CP-B patient died of progressive liver failure (CP score 7, FUW4 HCV RNA not collected). In conclusion, high rates of virologic response were observed in CP-B patients receiving a combination of GZR (50 mg) plus EBR (50 mg). The regimen was well tolerated with no evidence of hepatotoxicity. Plasma GZR exposure was slightly higher in CP-B patients receiving 50 mg compared to non-cirrhotic patients receiving 100 mg. EBR exposure was similar in both CP-B and non-cirrhotic groups.
EASL: Daclatasvir, Sofosbuvir, and Ribavirin Combination for HCV Patients with Advanced Cirrhosis or Post-transplant Recurrence: ALLY-1 Phase 3 Study - (04/28/15)
EASL: Ledipasvir/Sofosbuvir With Ribavirin is Safe and Efficacious in Decompensated and Post-Liver Transplantation Patients With HCV Infection: Preliminary Results of the SOLAR-2 Trial - (04/24/15)
EASL: Ledipasvir/Sofosbuvir With Ribavirin Is Safe in >600 Decompensated and Post-Liver Transplantation Patients With HCV Infection: An Integrated Safety Analysis of the SOLAR-1 and SOLAR-2 Trials - (04/29/15)
EASL: Harvoni Post-Transplant - (05/11/15)
EASL: All Oral HCV Therapy is Safe and Effective in Patients with Decompensated Cirrhosis: Report from HCV-TARGET - (04/23/15)
EASL: Simeprevir plus daclatasvir and sofosbuvir in treatment-naïve and treatment-experienced patients with chronic hepatitis C virus genotype 1 or 4 infection and decompensated liver disease: interim results from the Phase II IMPACT study - (04/24/15)
Special populations
Anything new for genotype 3?
The success rates for treatment of HCV genotype 3 so far have been limited particularly in treatment-experienced patients with cirrhosis. The all oral combination of sofosbuvir/ daclatasvir was shown to work well in treatment-naïve non-cirrhotic patients within the recent ALLY-3 study but to decrease considerably in presence of cirrhosis (9). The question whether additional ribavirin may help to prevent relapse remained unanswered as ribavirin was not studied in this particular study. Results from an ongoing ALLY-3+ study which also examines the role of ribavirin will most likely be presented at AASLD later this year. That there remains a need for better treatment options in this patient group was underlined by the results from the GT3 subanalysis with the TARGET real-life cohort (10). Of 202 patients with GT 3 the majority were treated with SOF/ribavirin (RBV). The corresponding SVR rates for the sofosbuvir + ribavirin combination (SOF/R) are shown below in figure 18 and clearly demonstrate that in the setting of cirrhosis and previous treatment failure SVR12 rate drops as low as 44%. Predictive factors are in particular low serum albumin and low platelets both markers of more advanced cirrhosis stages.
Figuren18: Real life SVR rates for GT3 from the TARGET cohort
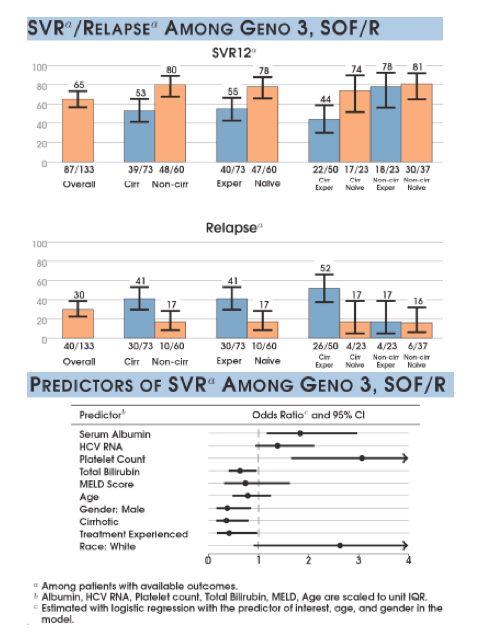
Similar findings were also reported from the TRIO observational cohort. Again SVR rates for sofosbuvir and ribavirin in GT3 patients was lowest in cirrhotic treatment experienced patients with 57% achieving SVR12 (11). The other interesting information from cohort studies is obviously the discontinuation rate in real life studies. Discontinuations were recorded overall in 9% of patients (4% noncompliant; 3% due to intolerance; 2% unrelated to treatment). No treatment-related SAEs were observed; 2% died but the deaths were unrelated to HCV therapy.
Interesting data on the NHS England Early Access Program (EAP) was presented at EASL which provided 12 weeks of therapy with sofosbuvir, with or without ribavirin and an NS5A inhibitor (provided by Gilead or BMS) to a cohort of ~500 patients with decompensated cirrhosis (12). In this first analysis data from 467 HCV patients (235 with GT1 infection, 189 with GT3 infection) was presented. Mean age of the total study population was 55.6 years (range 29-81), 72.2% were male, 73.9% white, 47.1% were treatment experienced, 10.1% were liver transplanted and 5.7% were HIV coinfected. With regard to liver disease 94.4% either had present or history of decompensated cirrhosis. 66.2% were classified as CPT B and 9.9% as C, 38.1% had active ascites and 17.1% active encephalopathy. So clearly this was a very advanced patient population. The SVR12 rates obtained so far for this ongoing study are depicted below in figure 19.
Figure 19: SVR12 by Genotype and Regime from the English EAP study
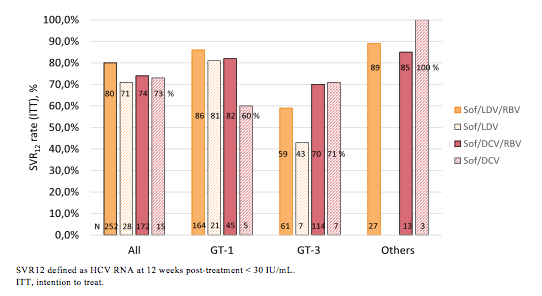
SVR12 defined as HCV RNA at 12 weeks post-treatment < 30 IU/mL.
ITT, intention to treat.
In decompensated cirrhosis 12 weeks therapy with sofosbuvir plus an NS5A inhibitor is effective with most patients achieving SVR12. Patients infected with Genotype 1 HCV respond well with >80% achieving SVR12, response rates were slightly reduced in patients with G3. Although numbers are small it appears as if more patients achieve SVR12 with the combination sofosbuvir + daclatasvir rather than ledipasvir/sofosbuvir, respectively. This would make sense as the in vitro data suggests more activity against GT3 for daclatasvir than ledipasvir. With a maximum of 71% responding to GT3 HCV therapy obviously the question becomes whether extending duration to 24 weeks could further improve SVR rates in this challenging patient population with very advanced cirrhosis stages.
Some first data on longer treatment durations with sofosbuvir and daclatasvir derive from the French ATU program (13). Overall, 601 HCV genotype 3 patients with severe fibrosis (F3)
or cirrhosis (F4), or HCV extra-hepatic manifestations or post-liver transplant HCV recurrence or indication for liver or kidney transplantation enrolled in the program. Most patients were male (75%), HCV mono-infected (83%), cirrhotic (75%), and treatment experienced (73%). This trial will provide valuable information once all patients have finished HCV therapy and follow-up. At the EASL presentation only few patents had completed therapy and/or follow-up which only allow a first look in a limited number of patients. The first SVR4 rates obtained are shown below in figure 20.
Figure 20: SVR4 rates with DCV + SOF ± RBV by treatment duration
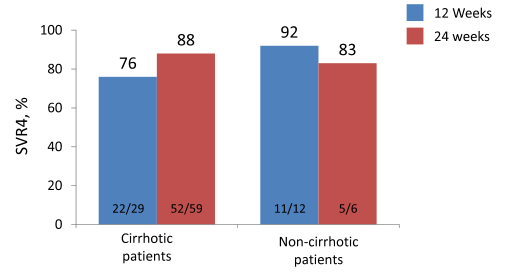
Clearly, longer follow-up and more patients need to be awaited before final conclusions can be drawn. First impression however is, that potentially treatment extension to 24 weeks may help to increase SVR rates in cirrhotic GT 3 patients receiving sofosbuvir and daclatasvir +/-ribavirin therapy.
As even dual DAA therapy seems still to struggle with cirrhotic GT3 patients one obvious question is whether this indeed may be a treatment situation were PEG-IFN could still play a role. Obviously, this would only be feasible in compensated cirrhosis as IFN should not be administered in decompensated cirrhosis because of the enhanced toxicity and increased risk for life-threatening events. In this context first results from the BOSON trial were presented (see figure 21)(14). The phase 3 BOSON study evaluated the safety and efficacy of SOF+PEG/RBV for 12 weeks vs SOF+RBV for 16 or 24 weeks in treatment-experienced genotype 2 (GT2) HCV-infected patients with cirrhosis, and in treatment-naïve and -experienced genotype 3 (GT3) HCV-infected patients with and without cirrhosis. Patients were randomized 1:1:1 to receive either SOF+RBV for 16 or 24 weeks or SOF+PEG/RBV for 12 weeks and stratified by HCV genotype and cirrhosis status. All patients received SOF 400mg daily and RBV 1000-1200mg in a divided daily dose. PEG was administered as 180 mg weekly injection. The primary end point was sustained virologic response 12 weeks after treatment (SVR12).
Figure 21: Study design of the BOSON trial
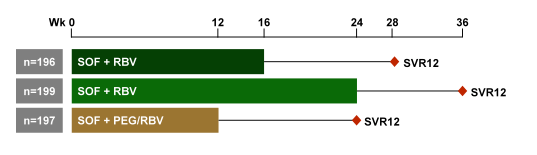
Of 592 patients randomized and treated, 92% had GT3 HCV, 67% were male, 84% white, 3% treatment experienced, 62% had non-CC IL28B genotypes, and 37% had cirrhosis. Mean platelets were 198 cell/mm3 (range 54-537). The SVR12 rates are summarized below in figure 22.
Figure 22: SVR12 in GT 2 vs GT 3 in the BOSON trial
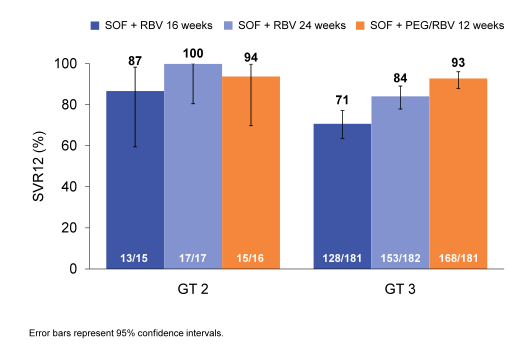
GT2 treatment experienced patients with cirrhosis had high SVR12 rates in all treatment groups: 87% of those receiving SOF+RBV for 16 weeks, 100% of those receiving SOF+RBV for 24 weeks, and 94% of those receiving SOF+PEG/RBV for 12 weeks. Among GT3 patients, SVR12 rates were highest in those receiving SOF+PEG/RBV for 12 weeks (93%) as compared to SOF+RBV for 24 (84%, p 0.008) or 16 weeks (71%, p < 0.001). The most common adverse events in all arms were fatigue, headache, insomnia, and nausea. Overall, 6 (1%) patients discontinued treatment due to adverse events; one of them was in the interferon study arm. In conclusion, GT2 treatment-experienced patients with cirrhosis had high SVR12 rates in all treatment arms. In GT3 patients, including a large proportion of treatment-experienced patients with cirrhosis, SOF+PEG/RBV for 12 weeks resulted in the highest
SVR12 rates observed to date in a Phase 3 study. Overall and in all subgroups, GT3 patients receiving 24 weeks of SOF+RBV had higher SVR12 rates than those receiving 16 weeks of treatment, confirming that 24 weeks is the optimal duration for this combination in GT3
patients. SOF+PEG/RBV for 12 weeks was well tolerated with a high rate of treatment completion. These data suggest SOF+PEG/RBV treatment should still be considered for IFN-eligible GT3 patients, particularly for those with cirrhosis and/or prior treatment failure. Please be aware of contraindications for IFN based therapy in decompensated cirrhosis. Nevertheless, this study clearly lets interferon reappear as a possible partner in HCV therapy for more difficult GT3 patients with prior treatment failure and more advanced fibrosis stages.
EASL: Sofosbuvir Plus Peg-IFN/RBV for 12 Weeks vs Sofosbuvir/RBV for 16 or 24 Weeks in Genotype 3 HCV-Infected Patients and Treatment-Experienced Cirrhotic Patients With Genotype 2 HCV: The BOSON Study - (04/27/15)
EASL: Treatment of decompensated HCV cirrhosis in patients with diverse genotypes: 12 weeks sofosbuvir and NS5A inhibitors with/without ribavirin is effective in HCV Genotypes 1 and 3 - (04/24/15) English EAP
APASL: All-Oral 12-Week Combination Treatment With Daclatasvir and Sofosbuvir in Patients Infected With HCV Genotype 3: ALLY-3 Phase 3 Study - (03/18/15)
EASL: Efficacy evaluation of 24 week SOF + RBV in a heterogeneous, real-world population of Genotype 3 HCV; data from the TRIO network - (04/30/15)
EASL: DACLATASVIR PLUS SOFOSBUVIR WITH OR WITHOUT RIBAVIRIN IN PATIENTS WITH HCV GENOTYPE 3 INFECTION: INTERIM ANALYSIS OF A FRENCH MULTICENTER COMPASSIONATE USE PROGRAM - (04/24/15)
Any news for HIV/HCV coinfected individuals?
At this year EASL repeat presentations of the BMS trial ALLY-2 in HIV/HCV coinfected patients with an SVR12 rate of 96% (please see figure 23) after 12 weeks of sofosbuvir/daclatasvir and the ION-4 study with an SVR12 rate of also 96% (please see figure 24) after 12 weeks of sofosbuvir/ledipasvir were presented (same presentations were shown at CROI 2015, please see NATAP CROI report on hepatitis) (15-18).
Figure 23: ALLY-2: DCV+SOF in HIV/HCV Coinfection
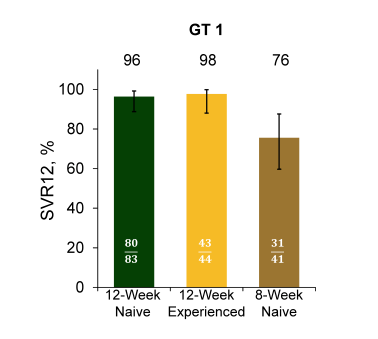
Figure 24: ION-4: LDV/SOF for 12 Weeks in HIV/HCV Coinfection
In addition for the first time full results of the C-EDGE coinfection study were presented which examined virological efficacy and safety of the fix-dose combination of grazoprevir/elbasvir for 12 weeks in 218 treatment-naive patients with HCV GT1, 4 or 6 infection with or without cirrhosis (19). Patients were co-infected with HIV-1 and either naive to ART with CD4+ >500 cells/mm3 and HIV RNA <50,000 copies/mL or on stable on ART for ≥8 weeks and CD4+ >200 cells/mm3 and undetectable HIV RNA. Stable antiretroviral therapy (ART) included tenofovir or abacavir, and either emtricitabine or lamivudine plus raltegravir, dolutegravir, or rilpivirine. The study design is shown in figure 25.
Figure 25: Study design of the C-EDGE coinfection study
Overall, 83% of study participants were male, 17.4% were black or African-American, and 66.1% had a GT1a, 20.7% GT1b /other, 12.8% GT4 and 0.5% GT6 HCV infection. 16.1% were cirrhotic, 35.3% had an IL28B CC genotype and 59.6% had a baseline HCV RNA >800,000 IU/mL. 96.8% were receiving ART with undetectable HIV RNA. The median (1st quartile - 3rd quartile) CD4 count was 568 cells/μl (424-766). 21.6% of patients were on an abacavir containing ART regimen, 75.2% on a tenofovir containing regimen; 51.8% received raltegravir as third agent, 27.1% dolutegravir and 17.4% rilpivirine. The results of the primary study endpoint (SVR12 = HCV RNA <15 IU/mL) is depicted below in figure 26.
Figure 26: SVR12 - C-EDGE Co-infection (Full Analysis set)
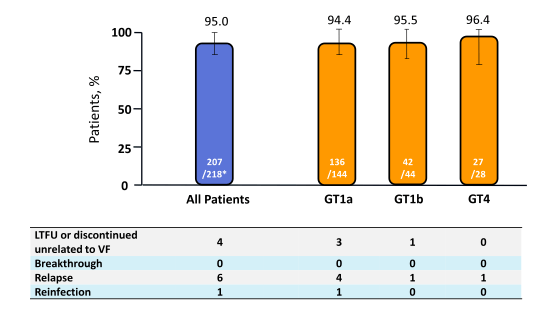
*1 patient with GT6 infection and 1 patient with GT1 not-otherwise subtyped were also included; both patients achieved SVR12.
GT = genotype; LTFU = long-term follow-up
Further sub group analyses showed no difference in SVR rate with regard to age, race, gender, cirrhosis, baseline HCV viral load, IL28B genotype or antiretroviral regimen. Overall tolerability overall was very good with 0% discontinuations because of adverse events. 2 patients developed late ALT or AST >5.0 x ULN. Clearly, the overall SVR rates once again are impressive and confirm that in the all oral DAA era SVR rates in HIV/HCV coinfected patients are superimposable to cure rates obtained in HCV mono-infection. The fact that one reinfection was observed in one patient between week 4 and 12 of follow-up underlines that DAA treatment alone will not control the ongoing epidemic of acute HCV in HIV infected subjects but that educational efforts including explanation of transmission pathways as well as counseling on sexual risk behavior and perhaps even behavioral interventions will be needed to minimize reinfections in the HIV patient population.
EASL: C-EDGE COINFECTION: PHASE 3 STUDY OF GRAZOPREVIR/ELBASVIR IN PATIENTS WITH HCV/HIV - (04/23/15)
CROI/2015: Daclatasvir in Combination With Sofosbuvir for HIV/HCV Coinfection: ALLY-2 Study - (02/27/15)
EASL: Daclatasvir Plus Sofosbuvir for Treatment of HCV Genotypes 1-4 in HIV-HCV Coinfection: The ALLY-2 Study - (04/28/15)
EASL: BMS at EASL - (05/11/15)
CROI/2015: Ledipasvir/sofosbuvir for 12 Weeks in Patients Coinfected With HCV and hiv-1: ION-4 - (02/27/15)
EASL: A Single-Tablet Regimen of Ledipasvir/Sofosbuvir for 12 Weeks in HCV Genotype 1- or 4-Infected Patients With HIV-1 Coinfection: the Phase 3 ION-4 Study - (04/28/15)
Anything new for treatment of HCV genotype 1?
Various interesting presentations were made with regard to HCV genotype 1 therapy. One poster which received considerable attention was a poster on the real-world effectiveness of ledipasvir/sofosbuvir 8 weeks for treatment of chronic HCV. Obviously, 8 weeks of this combination is currently possible in treatment naïve GT1 patients in case of HCV viral loads below 6 Mill IU. As this includes considerable cost savings this was of great interest. The aim of the present analysis was two-fold: Characterize the population receiving 8w LDV/SOF in Germany in clinical practice and secondly to describe outcomes. The first cohort of CHC patients who initiated treatment with 8w LDV/SOF, between 24/11/2014 to 22/01/2015, in a single center in Germany, were included into the analysis. To reflect clinical practice, no restriction was a priori imposed with regards to GT, treatment experience or fibrosis stage. The intra-center, non-strict criteria for 8 week treatment initiation included no previous treatment history, HCV RNA < 6 million IU/ml and Metavir stage < F3. The baseline characteristic and per protocol interim analysis of SVR4 rates are shown below in figure 27.
Figure 27: Real-world Effectiveness of LDV/SOF for 8 Weeks from a German single center
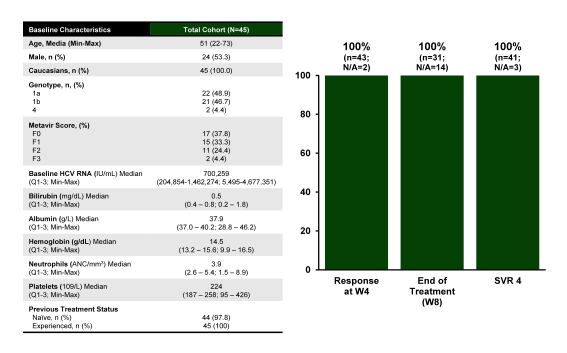
Results from this first real-world analysis on 8w LDV/SOF treatment indicate that this RBV-free regimen was prescribed, in the vast majority of patients, according to the SmPC (Summary of product characteristics) for treatment-naïve non-cirrhotic chronic hepatitis C patients with HCV RNA <6 million IU/mL at baseline. Moreover, in this population, LDV/SOF was a highly effective, safe and well tolerated treatment option with no major adverse drug reactions or any discontinuation reported.
EASL: Real-world effectiveness of Ledipasvir/Sofosbuvir 8 weeks chronic HCV treatment - (04/28/15)
Another much awaited presentations were from the OPTIMIST study-1 and 2 (21,22). Until now the combination of sofosbuvir and simeprevir was used mostly on the basis of the phase II study results (COSMOS trial). Due to the high need for all oral DAA combinations these drugs had been licensed and used before the phase 3 results were available. The phase 3, randomized, open-label study OPTIMIST-1 (NCT02114177) evaluated efficacy and safety of 12 and 8 weeks of SMV+SOF, in treatment-naïve or -experienced HCV genotype (GT)1-infected pts without cirrhosis (21). At baseline randomization (1:1; stratified by HCV GT subtype ±Q80K, IL28B GT, treatment history) to 12 or 8 weeks of SMV 150mg QD + SOF 400mg QD was undertaken. Superiority of SMV+SOF for 12 and 8 weeks, each vs a historical control (HC; derived from published data), was tested. If both were superior, non-inferiority of 8 vs 12 weeks of SMV+SOF was to be assessed. Primary efficacy endpoint was SVR 12 weeks after end of treatment (EOT; SVR12).The main baseline characteristics and virological efficacy outcome are depicted below in figure 28.
Figure 28: OPTIMIST 1: SMV+SOF for 8 or 12 Weeks in GT 1 Without Cirrhosis
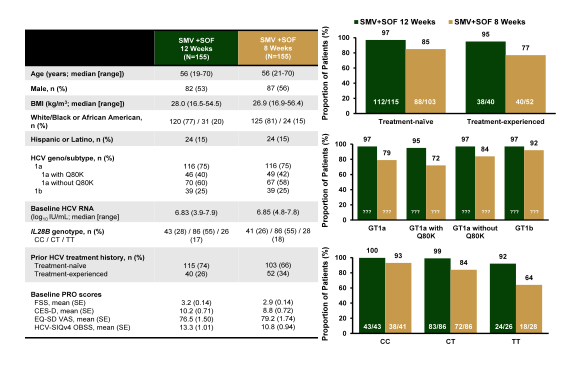
SMV+SOF for 12 weeks was superior to historic control whereas SMV+SOF for 8 weeks did not achieve superiority versus historic control in treatment-naïve and -experienced HCV GT1-infected pts without cirrhosis. The therapy of the 2 oral DAAs was well tolerated. Clearly these results help to strengthen the data for this DAA combination and also propose 12 weeks as the in general optimal treatment duration. As seen from other data sets the presence of the Q80K mutation at baseline seems to lose its impact in DAA combination therapy. The striking lower SVR rate in patients in the 8 week treatment arm with a TT IL28B genotype prompts the question whether factors can be reliably identified which would help to determine in which patient population shorter treatment durations may be applicable.
EASL: A Phase 3, randomised, open-label study to evaluate the efficacy and safety of 12 and 8 weeks of simeprevir (SMV) plus sofosbuvir (SOF) in treatment-naïve and -experienced patients with chronic HCV genotype 1 infection without cirrhosis: OPTIMIST-1 - (04/28/15)
The OPTIMIST-2 (NCT02114151) study aimed to demonstrate superiority of 12 weeks of SMV+SOF, in treatment-naïve or -experienced (including interferon (IFN)-intolerant) HCV
genotype (GT)1-infected pts with cirrhosis compared with a historical control (composite of the SVR12 rates of approved direct acting antiviral/IFN + ribavirin regimens) (22). Treatment-naïve or -experienced pts with chronic HCV GT1 infection and documented presence of cirrhosis received SMV 150mg QD+SOF 400mg QD for 12 wks. The primary efficacy endpoint was SVR12 in the overall population. Safety and patient-reported outcomes were assessed. 103 pts received treatment (male, 81%; median age, 58 years; Black/African American, 18%; IL28B CC, 28%; GT1a/1b, 70/30%; treatment-naïve [n = 50, 49%]; treatment-experienced [n = 53, 52%]). SVR12 with SMV+SOF (84% [95% confidence interval: 76, 91]) met the primary endpoint of superiority to the historical control (70%). The main baseline characteristics and virological outcomes are shown below in figure 29.
Figure 29: OPTIMST 2: SMV+SOF for 12 weeks in GT 1 with cirrhosis
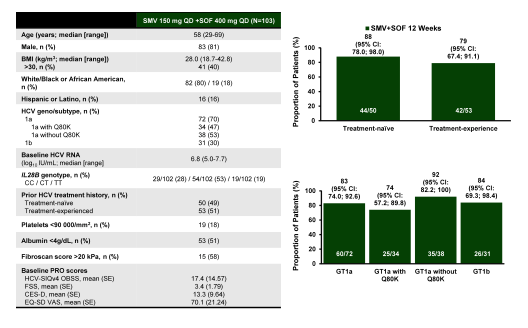
SMV+SOF for 12 weeks achieved superiority in SVR12 rates vs the historical control in treatment-naïve and -experienced HCV GT1-infected pts with cirrhosis and was generally well tolerated. In comparison to the treatment naïve and experienced patients without cirrhosis however, lower cure rates were observed which were as low as 74% in cirrhotics with GT1a infection with baseline Q80K mutation.
In addition, interesting data was presented from the comparison of telaprevir + PEG-IFN and ribavirin versus the Abbvie 3-D ± ribavirin regimen in treatment-naive HCV GT1-infected patients without cirrhosis. (23). Patients with HCV GT1a infection were randomized 2:1 to receive 3D+RBV or TPV+pegIFN/RBV. Patients with GT1b infection were randomized 2:2:1 to 3D+RBV, 3D, and TPV+pegIFN/RBV. The primary endpoint was SVR12. The main baseline characteristics of the study participants and the virological outcome data is depicted below in figure 30.
Figure 30: Malachite-I: 3D ± RBV or Telaprevir + pegIFN/RBV in HCV GT 1
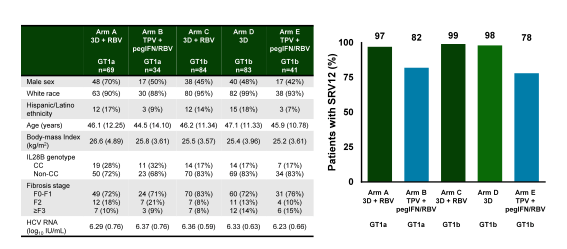
For patients receiving 3D+RBV or 3D, SVR4 and SVR12 rates were 97.1-98.8%; 3 patients had on-treatment failure and 1 patient had post-treatment relapse. In contrast in the control arm with telaprevir + PEG-IFN + ribavirin only 78% achieved cure of HCV. Rates of common AEs (including anemia, nausea, and pruritus), AEs leading to drug discontinuation or RBV dose modification, and serious AEs were significantly lower in patients receiving 3D±RBV compared to patients receiving TPV+pegIFN/RBV). The frequency of RBV-associated AEs, such as anemia, was significantly lower in the 3D±RBV arms compared to the TPV+pegIFN/RBV arm. This study is important as it allows to assess how the tolerability of all oral DAA-based HCV regimens differs from first generation DAA + IFN + RBV HCV regimens. Clearly getting rid of interferon improves tolerability significantly. It is also reassuring to see that on top of improved tolerability these all oral combinations also lead to much higher SVR rates. These results help to underline why triple therapy with telaprevir + PEG-IFN + RBV is no longer recommended by guidelines but rather oral DAA combinations have become the new standard of HCV therapy.
EASL: A Phase 3, open-label, single-arm study to evaluate the efficacy and safety of 12 weeks of simeprevir (SMV) plus sofosbuvir (SOF) in treatment-naïve or -experienced patients with chronic HCV genotype 1 infection and cirrhosis: OPTIMIST-2 - (04/29/15)
EASL: J&J at EASL - (05/11/15)
EASL: Malachite-I Phase 3b Trial of Ombitasvir/Paritaprevir/r and Dasabuvir ± Ribavirin or Telaprevir + Peginterferon/Ribavirin in Treatment-Naïve Adults With Hc v Genotype 1 - (04/27/15) and Malachite-II
Anything new in treatment of genotype 4, 5 and 6?
At EASL some new data on treatment of genotypes 4-6 was presented. The new ongoing
trial presented will be the first to evaluate ombitasvir/paritaprevir/r with ribavirin in an exclusively Egyptian population of patients with and without cirrhosis (23). The study design of trial is depicted below in figure 31. Up to now this phase 3, open-label, multicenter study has completed enrollment of the targeted number of 160 treatment-naïve and prior pegIFN/RBV-experienced subjects with HCV GT4 infection at 5 investigative sites. More data from this study will be presented shortly. The background for this study was the PEARL-1 study which has just been published online in the Lancet. In PEARL-1 the efficacy and safety of ombitasvir (OBV), a NS5A inhibitor and paritaprevir co-dosed with ritonavir, PTV/r) with or without ribavirin (RBV) was studied in 135 patients with HCV GT4 infection without cirrhosis. SVR12 was 100% in both treatment naïve and prior interferon (IFN) and RBV experienced patients receiving OBV plus PTV/r with RBV for 12 weeks (see also figure 32).
Figure 31: Study design
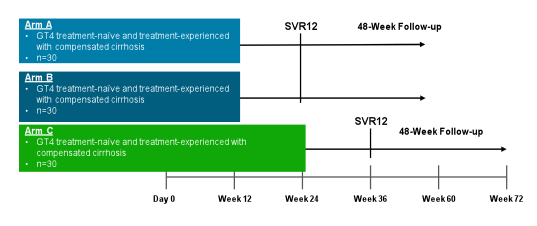
Figure 32: PEARL-I: Efficacy of OBV/PTV/r + RBV in Treatment-Naïve and Treatment-Experienced Non-cirrhotic Subjects with GT4 Infection for the PEARL-1 study
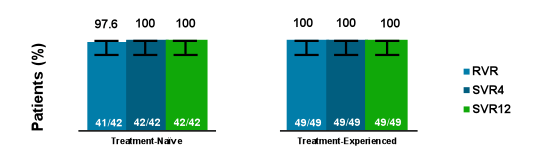
Clearly these are great cure rates which emphasize why it really makes sense to further study this particularly in Egypt which has the highest rate of GT4 infections globally. Also including patients with cirrhosis will allow to better understand the efficacy even in more challenging to treat patients with GT4 infections.
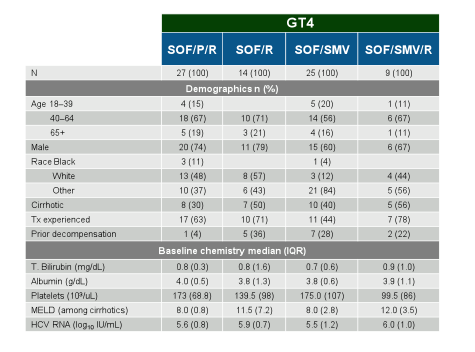
An additional presentation looked at safety and effectiveness of sofosbuvir based regimens for GT4 (also addressed GT3) (26). The aim of this study was to evaluate the safety and efficacy of sofosbuvir (SOF) containing regimens for the treatment of patients with GT 3 and
4 in HCV-TARGET (HCVT), a multicentre, prospective, observational cohort study. The baseline charcateristics are summarized in table 2.
EASL: An Open-Label Study to Evaluate the Efficacy and Safety of Co-Formulated Ombitasvir/Paritaprevir/Ritonavir With Ribavirin in Adults with Chronic HCV Genotype 4 Infection in Egypt (AGATE-II) - (04/24/15)
EASL: A Randomized, Open-Label Study to Evaluate Efficacy and Safety of Ombitasvir/Paritaprevir/Ritonavir Co-Administered With Ribavirin in Adults With Genotype 4 Chronic Hepatitis C Infection and Cirrhosis (AGATE-I) - (04/23/15)
EASL: Abbvie at EASL 2015 - Healthcare Economics, Clinical Trials - (05/11/15)
Table 2: Baseline characteristics of the GT4 patient population in TARGET
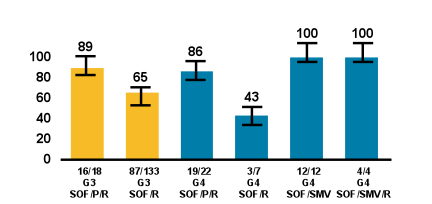
Patients with GT 4 received peginterferon (PEG) plus SOF/RBV (n=27), SOF/RBV (n=14) or simeprevir (SMV)/SOF (n=25) or SMV/SOF/RBV (n=9). Interim SVR data for those patients which already finished follow-up are shown below in figure 33 for the different regimens used. It becomes clear despite low numbers in some groups that dual DAA therapy seems to be the way forward for GT4 therapy.
Figure 33: SVR rates for GT3 and 4 patients overall in the TARGET cohort
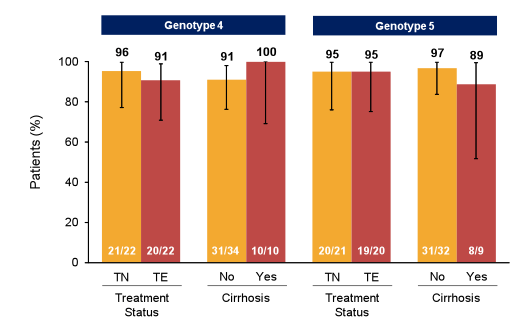
Finally, in an oral presentation the safety, tolerability and efficacy of ledipasvir/sofosbuvir (LDV/SOF) in patients with chronic GT4 or GT5 HCV infection was discussed. Treatment-naïve and treatment-experienced patients with chronic GT4 or GT5 HCV infection were enrolled at 5 sites in France to receive 12 weeks of LDV/SOF (90 mg/400mg daily). Overall 22 treatment-naïve as well as treatment experienced patients with HCV GT4 infection were enrolled as well as 21 and 20 treatment-naïve and treatment experienced HCV GT5 patients, respectively. The SVR12 rates by treatment history and cirrhosis status are shown in figure 34.
Figure 34: SVR12 by Treatment History and Cirrhosis Status
EASL: Ledipasvir/Sofosbuvir for the Treatment of Patients With Chronic Genotype 4 or 5 HCV Infection - (04/29/15)
Anything about new drugs?
At this year EASL clearly most new data on new drugs or combinations came from the Merck development program. Studies with grazoprevir (GZR) and elbasvir (EBR) have already been presented for HIV coinfection, Child-Pugh (CP) class B cirrhosis and renally impaired patients in the corresponding sections above. Important other studies were in particular the C-EDGE studies in treatment naïve (TN) and treatment experienced (TE) genotype 1, 4 or 6 patients.
The C-EDGE TN study (P060) investigated the safety and efficacy of a once daily regimen of GZR (NS3/4A protease inhibitor) and EBR (NS5A inhibitor) for 12 weeks (weeks) in TN patients (pts) with GT 1, 4, or 6 infection (28,29). C-EDGE TN was an international, randomized, blinded, placebo-controlled, parallel-group trial of an oral fixed-dosed combination of GZR 100 mg/EBR 50mg once daily in TN, HCV GT 1-, 4-, or 6-infected pts. Cirrhotic patients were eligible. Exclusion criteria included decompensated liver disease, HCC, HIV or HBV co-infection, platelets <50×103/mL, or albumin <3.0 g/dL. Patients were assigned in a 3:1 ratio to receive either immediate or deferred therapy after stratification by GT and fibrosis stage assessed by biopsy or noninvasive means. HCVRNA levels were measured by the COBAS TaqMan v2.0 assay. The primary efficacy endpoint was pre-specified as the proportion of treated pts in the immediate GZR/EBR arm with unquantifiable RNA levels (<15 IU/mL) 12 weeks after the end of study treatment (SVR12). The study design is shown below in figure 35.
Figure 35: Study design of the C-EDGE TN study
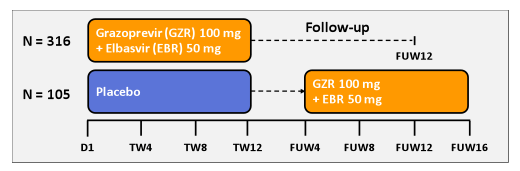
Overall, 421 (90%) of 469 screened pts were enrolled, randomized, and treated with ≥1 dose of study drug: 194 (46%) women; 157 (37%) non-white; 385 (91%) GT1 and 36 (9%) GT4/6
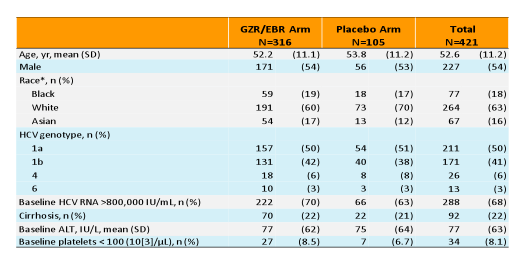
infections. Among the 92 (22%) pts with cirrhosis, the diagnosis was biopsy-proven in 26 (28%); the median platelet count [IQR] and albumin level [IQR] in cirrhotics were 123.5 [91.5-157.5] ×103/mL and 4.1 [3.8-4.4] g/dL, respectively. The baseline demographics are summarized below in table 3.
Table 3: Demographics of the C-EDGE TN trial
The SVR12 rates are shown below in figure 36.
Figure 36: SVR12 rates in the C-EDGE TN study
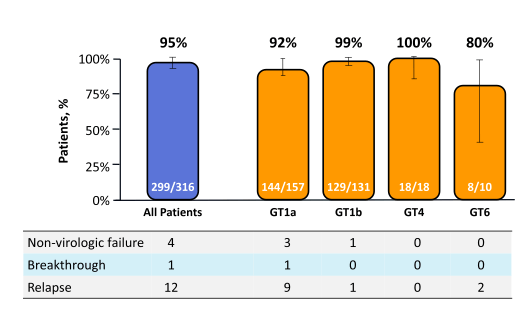
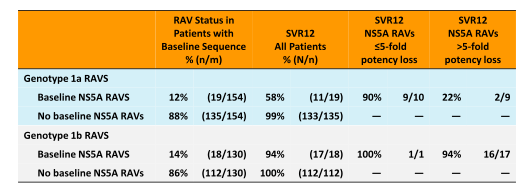
Overall, cure rate with 95% was very high for this IFN and RBV free fixed-dose combination HCV therapy. Responses were slightly higher in GT1b and 4 than in 1a. Lowest response rates were observed in GT6 patients, but numbers were quite small in this group. Looking at the higher relapse rate for GT1a patients the question arises whether certain baseline factors may impact risk for relapse in particular resistance associated variants (RAVS) at baseline. Neither race, presence of cirrhosis, gender, age or baseline IL28B genotype had any influence. Low baseline HCV viral load (defined as < 800.000 IU/ml) however, was associated with a higher chance of obtaining SVR. Indeed all patients with virologic failure had baseline HCV RNA of > 800,000 IU/mL. RAVS against NS3/4A were found in 57% of GT1a patients and 19% of GT1b patients respectively. None of the NS3/4A RAVS had any impact on overall SVR rate. Whereas genotype 1a RAVS against NS5A in particular those with >5-fold potency loss were associated with significantly reduced SVR12 rates suggesting that at least for GT 1a patients presence of baseline NS5A RAVS with >5-fold potency loss will negatively impact probability of achieving HCV cure. The corresponding findings are summarized in Table 4.
Table 4: Resistance Associated Variants: NS5A(Resistance analysis population)
The overall tolerability was very good with very few drug discontinuations because of adverse events (<1%). The most common adverse events and safety data is summarized in table 5.
Table 5: Grazoprevir / Elbasvir: Adverse events
In summary, the fix dose combination of grazeprovir/elbasvir achieved SVR by 95% of patients with high efficacy across GT1, GT4, and GT6 infection and also high efficacy in cirrhotics (SVR12 = 97.1%). Overall virologic failure rate was 4%; lower efficacy observed only among patients with high viral load and primarily with baseline GT1a RAVs that cause >5-fold potency reduction to elbasvir. The new DAA combination was generally well-tolerated, with a similar safety profile in cirrhotic and non-cirrhotic patients.
EASL: THE PHASE 3 C-EDGE TREATMENT-NAIVE (TN) STUDY OF A 12-WEEK ORAL REGIMEN OF GRAZOPREVIR (GZR, MK-5172)/ELBASVIR (EBR, MK-8742) IN PATIENTS WITH CHRONIC HCV GENOTYPE (GT) 1, 4, OR 6 INFECTION - (04/24/15)
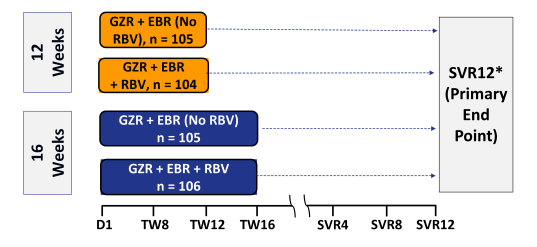
The next important study with the grazoprevir/elbasvir combination was C-EDGE TE (treatment experienced) (30). The aim of C-EDGE TE was to evaluate the efficacy and safety of the once-daily grazoprevir (GZR) + elbasvir (EBR) fixed-dose combination +/- RBV for 12 or 16 weeks in patients who have failed prior treatment with Peg-IFN + RBV. The study design is shown below in figure 37.
Figure 37: Study design of the C-EDGE TE study
Patients were randomized to 12 or 16 weeks ± RBV and stratified according to cirrhosis status and prior treatment response to peg-interferon + ribavirin. The primary end point of the study was SVR12 (HCV RNA < 15 IU/mL at 12 weeks after end of treatment; COBAS TaqMan v2.0). The baseline demographics are shown below in figure 38.
Figure 38: Baseline demographics in the C-EDGE TE study
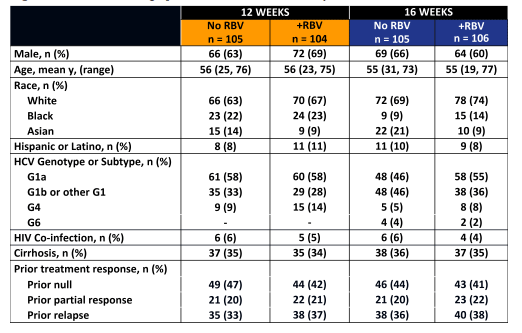
The high rate of previous null-responders, cirrhotics and GT1a patients highlights that this is indeed quite a challenging patient group to treat. The main virological outcome is shown in figure 39.
Figure 39: Primary efficacy results (SVR12), Intent To Treat Analysis
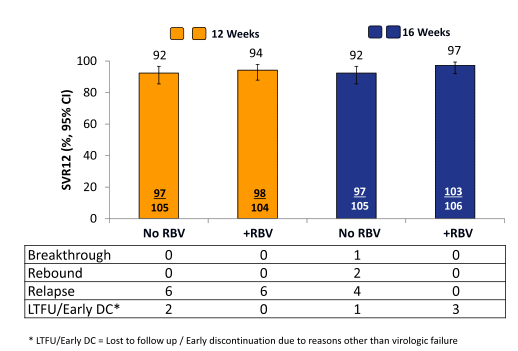
Clearly, the overall cure rate with 92% is high for this difficult-to-treat population. Whereas in the 12 week arm the addition of ribavirin seemed to associated with no difference in SVR rate (both 12 week arms had 6 relapses) no relapse was observed in the 16 week + ribavirin arm suggesting that extending treatment duration to 16 weeks plus the addition of ribavirin may actually be helpful in preventing relapse in those more difficult to treat patients. This becomes even more evident when one looks at the SVR rate by cirrhosis status and prior treatment response (see figure 40).
Figure 40: HCV Genotype 1a: Efficacy by prior treatment response and cirrhosis status
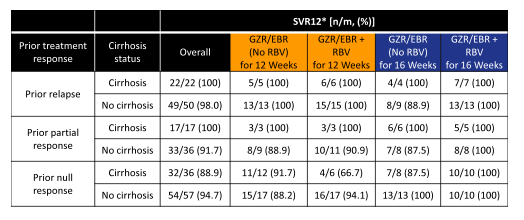
So even in cirrhotic, previous null-responders with GT1a infection, all patients were cured with 16 weeks and the addition of ribavirin. Other subgroup analyses revealed that 12 weeks without RBV achieved SVR12 in 100% of patients with history of relapse to prior PR as well as 12 weeks without RBV achieved SVR12 in 100% of G1b infected patients. As in the treatment naïve trial NS5A Variants with >5-Fold Shift to EBR were associated with decreased SVR12 rates in GT1a patients.
EASL: EFFICACY AND SAFETY OF GRAZOPREVIR/ELBASVIR +/- RBV FOR 12 OR 16 WEEKS IN PATIENTS WITH HCV G1, G4 OR G6 INFECTION WHO PREVIOUSLY FAILED PEGINTERFERON/RBV: C-EDGE TREATMENT-EXPERIENCED - (04/23/15)
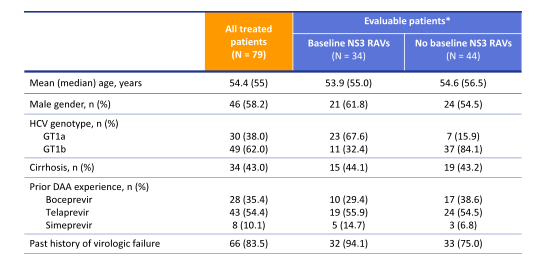
Even more challenging may be the treatment of patients who have already failed a DAA based triple therapy. C-SALVAGE is an international open-label study of GZR 100mg QD, EBV 50mg QD, and weight-based RBV BID for 12 weeks in patients with chronic HCV GT1 infection who had failed ≥4 weeks of peginterferon and RBV combined with boceprevir, telaprevir, simeprevir, or sofosbuvir (31,32). Per protocol, ~80% of the enrolled subjects were to have experienced virologic failure. Exclusion criteria included decompensated liver disease, hepatocellular carcinoma, HIV or HBV co-infection, thrombocytopenia <50×103/mL, or hypoalbuminemia <3.0 g/dL. HCVRNA levels were measured by COBAS TaqMan v2.0 assay. Resistance-associated variants (RAVs) were identified at baseline by population sequencing. The primary efficacy outcome was a HCVRNA level below the limit of quantification (15 IU/mL) 12 weeks after the end of treatment (SVR12). Overall, 79 patients were treated in this trial. The baseline characteristics are shown below in figure 41.
Figure 41: Baseline characteristics in the C-SALVAGE study
In this trial an even higher percentage of patients with cirrhosis was included and patients had also mostly already failed DAA-based therapy. The SVR rates are shown below in figure 42.
Figure 42: Efficacy endpoints over time
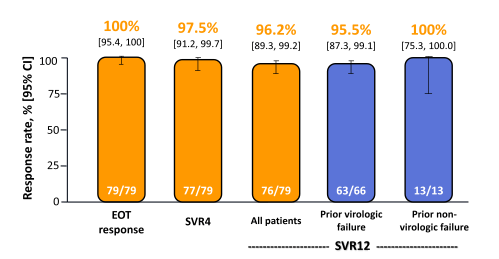
GZR + EBR + RBV x 12 weeks resulted in an impressive SVR12 rate of 96.2% in patients who had failed previous PR plus an earlier-generation PI. SVR12 was attained in 63/66 (95.5%) patients with a history of virological failure and SVR12 was attained in 31/34 (91.2%) patients harboring baseline NS3 RAVs conferring decreased susceptibility to 1st-generation PIs. In the 3 patients with relapse NS5A RAVS were found after relapse. At baseline one of those 3 patients had no NS5A RAVS and one had the L31M and one a H58D.
EASL: C-SALVAGE: GRAZOPREVIR (GZR; MK-5172), ELBASVIR (EBR; MK-8742) AND RIBAVIRIN (RBV) FOR CHRONIC HCV-GENOTYPE 1 (GT1) INFECTION AFTER FAILURE OF DIRECT-ACTING ANTIVIRAL (DAA) THERAPY - (04/23/15)
Another exciting study which combined new with already licensed DAAs was the C-SWIFT study (33). Shorter durations of therapy using novel combinations of DAAs may offer benefits such as increased compliance, lower cost and broader usage and acceptance by treating providers. Also in particular for GT3 improved treatment options are still eagerly awaited. The aim of this study was to assess the efficacy and safety of grazoprevir (GZR;NS3/4A protease inhibitor) and elbasvir (EBR; NS5A inhibitor) fixed-dose combination (FDC) + sofosbuvir (SOF; nucleotide NS5B polymerase inhibitor) in treatment-naive patients with and without cirrhosis with hepatitis C virus (HCV) genotype 1 (G1) infection for shortened durations of 4, 6 or 8 weeks and G3 infection for durations of 8 or 12 weeks. The study design is depicted below in figure 43.
Figure 43: Study design of the C-SWIFT study
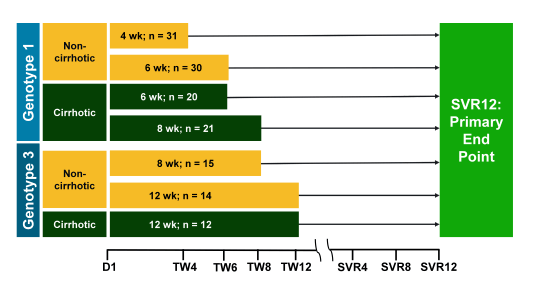
C-SWIFT enrolled 102 G1 and 41 G3 treatment-naïve patients with and without cirrhosis. G1 noncirrhotic (n = 61) patients were randomized to 4 or 6 week durations; G1 cirrhotic (n = 41) patients were randomized to 6 or 8 weeks of therapy. G3 noncirrhotic patients (n = 29) were randomized to 8 or 12 weeks and G3 cirrhotic patients (n = 12) were assigned to 12 weeks of therapy. All patients received the same regimen: GZR/EBR (100 mg/50 mg) FDC + SOF (400 mg). HCVRNA was assessed using COBAS TaqMan v2.0 (LoQ 15 IU/mL). The primary end point is the proportion of patients achieving HCVRNA <15 IU/mL 12 weeks after end of
treatment (SVR12). A total of 143 patients were enrolled; 66% male, 98% white, and 45% Hispanic. Of the 102 G1 patients, 82% were G1a. Mean viral load at baseline was 6.65 log10 IU/mL. The main virological outcomes are shown in figure 44.
Figure 44: HCV G1: Primary efficacy results SVR12; Modified Intent To Treat Analysis*
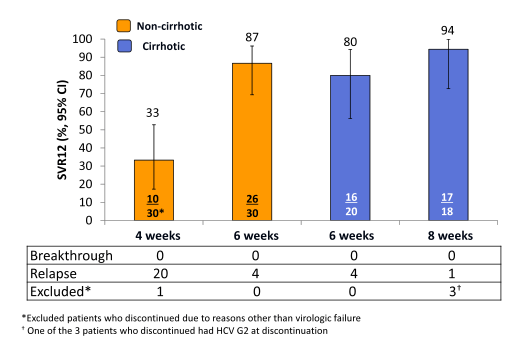
Overall, highest SVR rates for the GT1 patients were achieved with the longer treatment duration of 8 weeks suggesting that maybe there is a minimum duration of HCV therapy. Results for the GT arms are shown below (Figure 45).
Figure 45: C-SWIFT: SVR Results - HCV 3
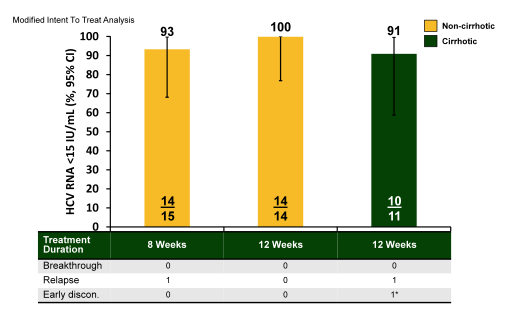
Excellent results were also obtained for the genotype 3 patients where in the two 12 week's arms only one relapser was observed. Now clearly patient numbers are small but the good results encourage further GT3 studies. Of note is however, that the combination of grazoprevir and elbasvir plus ribavirin for treatment of GT3 infection without sofosbuvir obtained much lower SVR rates of 45.0% and 57.1% for 12 and 18 weeks treatment duration, respectively (34). This underlines the activity of sofosbuvir in this combination and also suggests that the grazoprevir/elbasvir combination has reduced GT3 activity. [from Jules: keep in mind Merck is developing their own nuke MK3682.]
EASL: C-SWIFT: GRAZOPREVIR/ELBASVIR + SOFOSBUVIR IN CIRRHOTIC AND NONCIRRHOTIC, TREATMENT-NAIVE PATIENTS WITH HEPATITIS C VIRUS GENOTYPE 1 INFECTION, FOR DURATIONS OF 4, 6 OR 8 WEEKS AND GENOTYPE 3 INFECTION FOR DURATIONS OF 8 OR 12 WEEKS - (04/23/15)
Merck at EASL 2015 - (05/22/15)
Another novel and exciting study on new DAA combinations with sofosbuvir was presented by Ed Gane (35). Previously it had been demonstrated that GS-9857, a potent pan-genotypic HCV NS3/4A protease inhibitor (PI) with an enhanced resistance profile compared to first generation HCV PIs, demonstrated >3 log reductions in HCVRNA after 3 days of monotherapy in a Phase 1b study in patients infected with HCV genotypes 1-4. In the new study presented at EASL the safety and efficacy of GS-9857 with SOF/GS-5816, a fixed dose combination of pan-genotypic NS5B and NS5A inhibitors, in a regimen with 3 different mechanisms of action for 4 or 6 weeks in treatment-naïve and direct-acting antiviral (DAA)-experienced patients with HCV genotype 1 with and without cirrhosis was reported. Treatment-naïve genotype 1 HCV-infected patients without cirrhosis were treated for 4 or 6 weeks with GS-9857 with SOF/GS-5816. Treatment-naïve genotype 1 HCV-infected patients with cirrhosis and those who had failed ≥2 DAAs were enrolled in separate cohorts to also receive 6 weeks of GS-9857 with SOF/GS-5816. 75 patients with HCV genotype 1 were enrolled. The majority were male (51, 68%), had subtype 1a infection (59, 79%), and had the IL28B non-CC genotype (51, 68%). Median HCVRNA in each group ranged from 6.31 to 6.46 log10 IU/mL. All patients completed treatment and 97% achieved rapid viral suppression to < LLOQ by Week 4. The SVR12 rates are shown below in figure 46.
Figure 46: SOF/GS-5816 + GS-9857: SVR12 in GT 1 patients
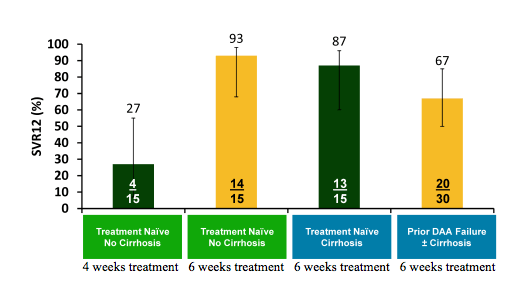
GS-9857 with SOF/GS-5816 was well tolerated for 4 or 6 weeks. There were no Grade 3 or 4 adverse events, serious adverse events, or deaths. The most frequent adverse events were
nausea (19, 25%), headache (18, 24%), and fatigue (12, 16%). Overall, treatment for 6 weeks was highly efficacious in GT1 treatment-naïve patients without cirrhosis. Treatment-experienced patients or those with cirrhosis may benefit from longer durations of therapy. Ongoing Phase 2 studies are evaluating GS-9857 with SOF/GS-5816 for durations of 6, 8 and 12 weeks in genotype 1-6 patients.
EASL: SAFETY AND EFFICACY OF SHORT-DURATION TREATMENT WITH GS-9857 COMBINED WITH SOFOSBUVIR/GS-5816 IN TREATMENT-NAIVE AND DAA-EXPERIENCED GENOTYPE 1 PATIENTS WITH AND WITHOUT CIRRHOSIS - (04/23/15)
EASL: Gilead at EASL 2015 - (05/12/15)
A further study also addressed shorter treatment durations with new DAAs from Achillion. ACH-3102 is a 2nd generation NS5A inhibitor in Phase 2 development for the treatment of chronic hepatitis C viral (HCV) infection. ACH-3422, a uridine nucleotide HCV NS5B polymerase inhibitor in Phase 1, is planned for use in combination with ACH-3102. This Phase 2, open-label, randomized, controlled, partial crossover study investigates the efficacy and safety of ACH-3102 plus sofosbuvir (used as a proxy for ACH-3422) (36). Study population was a treatment naïve, genotype 1, non-cirrhotic patient group. Overall, 6 and 8 weeks with each 12 patients were studied. SVR12 was achieved in 100% of all patients. Therefore, this becomes the first study to report 100% SVR12 in patients using a two-drug combination for 6 weeks. Clearly these results are very promising and results from larger trials are awaited eagerly. Additional genotypic and phenotypic analyses performed on baseline samples showed that ACH-3102 had greater potency against multiple GT-1a and -1b NS5A mutants compared with ledipasvir and daclatasvir (37). This holds promise for retreatment in some of the failures which are currently taking place. [from Jules: after EASL just last week a J&J/Achillion deal was announced, J&J will be developing Achillion drugs.]
EASL: Achillion at EASL 2015 - (05/15/15)
Does resistance matter after all?
The last but not least topic to tackle is resistance against DAAs. Whereas previously it was often stated that resistance was less likely to play a role in the context of DAA therapy and that there was no need for genotypic resistance testing other than potentially identifying the Q80K mutation in simeprevir based regimens with interferon, this may change with new results from various presentations here at the EASL conference. Whereas it has been shown that resistance associated variants (RAVs) against NS3/4A which appear after virological failure under HCV protease inhibitor based therapy tend to disappear over time (faster for GT1b than 1a) and even retreatment with HCV PIs has been possible there was much less data on what happens with NS5A RAVs. Interesting data on this issue was presented by Krishnan and colleagues which assessed the persistence of treatment-emergent RAVs in HCV NS3, NS5A and NS5B in patients who did not achieve SVR in Phase 2 and 3 clinical trials with paritaprevir/r, ombitasvir and dasabuvir, with available data through at least PTW24, by population and clonal sequencing (38). In patients who experienced virologic failure in the Phase 2 and 3 clinical trials, the predominant RAVs in GT1a were R155K and D168V in NS3, M28T and Q30R in NS5A, and S556G in NS5B. For paritaprevir, RAVs (any position) in NS3 persisted at detectable levels through at least PTW24 in 45% (31/69) of the patients, and through PTW48 in 7% (4/55) of patients. NS3 R155K remained detectable in 66% (8/12) and 25% (2/8) of the patients through PTW24 and 48, respectively; and D168 variants remained detectable in 37% (21/56) and 2% (1/53) of the patients through PTW24 and 48, respectively. For ombitasvir, RAVs in NS5A including M28T and Q30R persisted at detectable levels through PTW24 in 98% (67/68) of the patients, and through PTW48 in 96% (50/52) of the patients. For dasabuvir, RAVs in NS5B persisted at detectable levels through at least PTW24 in 75% (30/40) of the patients, and through PTW48 in 61% (22/36) of the patients. NS5B S556G persisted in 89% (24/27) and 79% (19/24) of patients through PTW24 and 48, respectively. Due to very high SVR rates in GT1b, trends in persistence of treatment-emergent RAVs could not be evaluated. The resistance findings are summarized in figure 47.
Figure 47: Persistence of RAVs following Ombitasvir/Paritaprevir/r + Dasabuvir
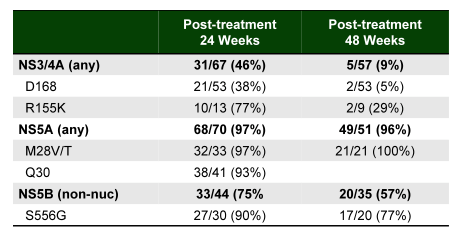
It appears that indeed NS5A variants may stay around much longer than anticipated and potentially could affect subsequent treatment attempts, which would include a different NS5A inhibitor.
EASL: LONG-TERM FOLLOW-UP OF TREATMENT-EMERGENT RESISTANCE-ASSOCIATED VARIANTS IN NS3, NS5A AND NS5B WITH PARITAPREVIR/r-, OMBITASVIR- AND DASABUVIR-BASED REGIMENS - (04/24/15)
Additional interesting data on the persistence of NS5A RAVs was presented from former ledipasvir failures (39). The aim of this study was to evaluate the persistence of resistance-associated variants (RAVs) in subjects who did not achieve SVR in previous Gilead-sponsored HCV clinical studies after receiving LDV without SOF who were then enrolled in a 3-year follow-up registry study. Specimens from subjects treated with LDV in Gilead studies GS-US-248-0120, GS-US-248-0121, GS-US-248-0131, GS-US-248-0132, GS-US-256-0124 and GS-US-256-0148, were analyzed to determine their NS5A RAVs at baseline (BL) in the parent study (n = 84), post-BL in the parent study (n = 78), BL in the registry study (n = 80), and follow-up (FU) visits weeks 12 (n = 66), 24 (n = 66), 36 (n = 19), 48 (n = 80) and 96 (n = 65) in the registry study. Population sequencing was used for majority of samples at BL and Post-BL of the parent study, and deep sequencing for all samples in the registry study. If RAVs were observed, samples from subsequent time-points were deep sequenced until RAVs were no longer detected (1% detection threshold). Figure 48 shows the distribution of RAVs at baseline and at virological failure.
Figure 48: NS5A RAVs before treatment and after virological failure with LDV without sofosbuvir therapy
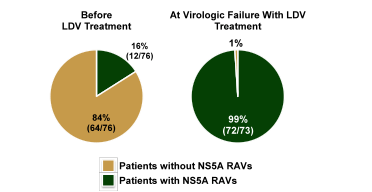
It appears that almost all patients developed NS5A RAVs at treatment failure. The course of RAVs over time is shown in figure 49.
Figure 49: Majority of RAVs Detected After 96 Weeks (> 1% of Population)
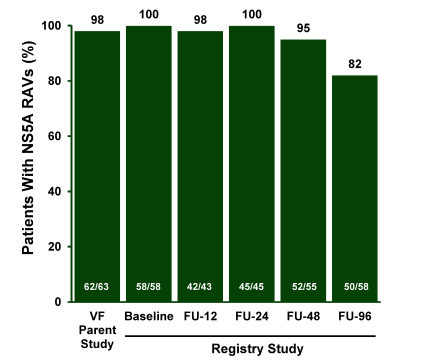
In conclusion, NS5A variants can persist for >96 weeks post-treatment in subjects who relapse to regimens containing NS5A inhibitor suggesting high fitness of the NS5A RAVs.
Clinically the most important question is if for example NS5A RAVs persist could they impact subsequent DAA therapies which contain a NS5A inhibitor and could genotypic resistance testing help to guide treatment choices after DAA failure. In this context a very interesting presentation was made by Eric Lawitz and colleagues (40). This study evaluated
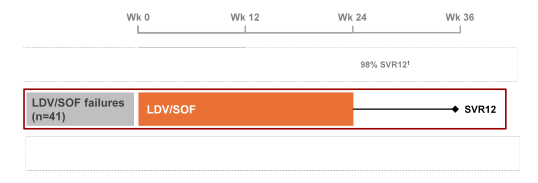
the efficacy of LDV/SOF for 24 weeks as retreatment for the few patients which failed in the phase 3 program (2%; n=35) and other patients that have failed similar regimens in LDV/SOF Phase 2 studies. This open-label study enrolled genotype 1 HCV infected patients who relapsed following 8 or 12 weeks of LDV/SOF ± ribavirin (RBV). NS5A and NS5B resistance associated variants were evaluated by deep sequencing. The primary endpoint was SVR12. The study design of this retreatment trial is shown below in figure 50.
Figure 50: Study design of a retreatment trial with ledipasvir/sofosbuvir in previous relapsers in the phase 2/3 program
41 patients were enrolled and completed treatment. The mean age was 58 years, 83% male, 24% African-American, 46% had cirrhosis, 93% IL28B CT/TT genotype, 83% GT1a, and mean baseline HCVRNA was 6.2 log10 IU/mL. The main baseline characteristics are summarized below (see figure 51).
Figure 51: Demographics and Baseline Characteristics (GT 1 Retreatment)
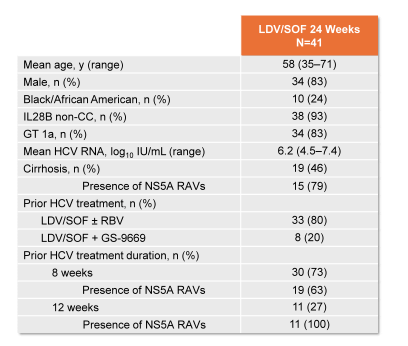
It is noteworthy that the presence of NS5A RAVS was lower in patients who had been exposed for only 8 weeks of ledipasvir/sofosbuvir therapy versus 12 weeks of therapy with 63 and 100% respectively. The main virological outcome depending on cirrhosis state, prior treatment duration and baseline RAVs is depicted below in figure 52.
Figure 52: SVR12 by Subgroup
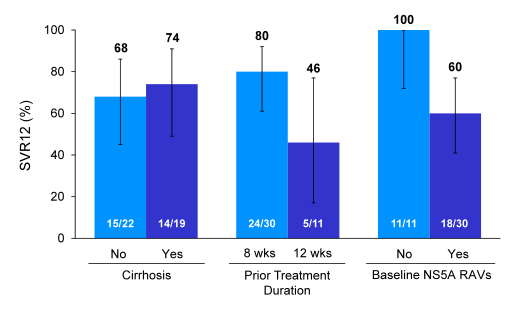
Overall, presence of cirrhosis had no impact on achieving SVR12, whereas prior longer treatment duration and presence of baseline RAVs had a clear impact on chance to achieve SVR12. Obviously longer prior treatment duration was associated with higher risk for developing RAVs upon relapse so these two events are somewhat linked. As in treatment-naïve patients with low HCV RNA levels shorter treatment duration of 8 weeks can be selected this would at least imply that in case of virological failure retreatment may be easier as risk of developing RAVs is lower. However, it also becomes apparent that once RAVs are there, future retreatment will be difficult, particularly as NS5A RAVs tend to persist (see studies above). Clinically the next important question then becomes are all RAVs the same or are there some which are more impactful in affecting SVR12 probability? Figure 53 shows SVR12 rates depending on number and type of RAVs from this study. [from Jules: another important question is what will be the effectiveness against this NS5A resistance by new 2nd generation NS5A inhibitors: Merck's MK8408, Abbvie's (ABT-530 and Achillion's ACH-3102 (now belongs to J&J); and for that matter what affect the little NS3 resistance we see will have if any.]
Figure 53: SVR12 by Baseline NS5A RAVs
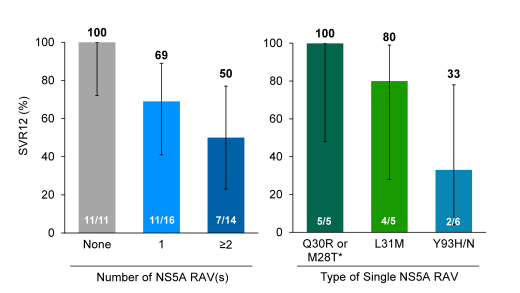
Clearly, what we can learn from these analyses is that number of RAVs matter and that in particular detection of the L31M and/or Y93H/N is associated with a higher risk for developing virological failure. This implies that after all oral DAA therapy including a NS5A inhibitor genotypic resistance testing indeed can be helpful to select the most active and promising retreatment therapy.
EASL: Long-Term Persistence of HCV NS5A Variants After Treatment With NS5A Inhibitor Ledipasvir - (04/29/15)
EASL: Retreatment of Patients Who Failed 8 or 12 Weeks of Ledipasvir/Sofosbuvir-Based Regimens With Ledipasvir/Sofosbuvir for 24 Weeks - (04/24/15)
EASL: The Prevalence and the Effect of HCV NS5A Resistance-Associated Variants in Patients With Compensated Cirrhosis Treated With Ledipasvir/Sofosbuvir ± RBV - (04/29/15)
EASL: Prevalence of Pretreatment NS5A and NS5B Resistance-Associated Variants and Genetic Variation Within HCV Subtypes Across Different Countries/Gilead-Study - (04/30/15)
Summary
· New EASL guidelines have been presented which underline that all treatment-naïve and treatment-experienced patients with compensated or decompensated chronic liver disease related to HCV, who are willing to be treated and who have no contraindications to treatment, should be considered for therapy. Updated priorization criteria for patients with immediate treatment need are provided as well as HCV therapy selection based on genotype, fibrosis stage and treatment experience.
· The Abbvie 3D HCV regimen has been well tolerated in GT1 patients with advanced renal disease, including those on hemodialysis, with or without ribavirin. All patients which achieved post-treatment week 4 show SVR4
· Once-daily GZR/EBR for 12 weeks was highly effective for treatment of HCV GT1 infection among patients with chronic kidney disease stage 4/5 (n=224); efficacy was consistent across different subpopulations: GT1a and 1b, diabetes, and hemodialysis.
· LDV/SOF + RBV resulted in high SVR12 rates in HCV patients with advanced liver disease, irrespective of transplantation status. The majority of patients with decompensated cirrhosis achieved improvements of their MELD score. However, some patients showed no improvement or progressed suggesting we need to better understand how HCV cure impacts individual liver disease stage. Changes in MELD score may also impact chance to receive a liver transplant which needs to enter the discussion of when to best treat an HCV infected individual on the transplant list.
· Overall, cure rates are higher in patients post-transplant than in advanced cirrhosis suggesting that HCV therapy in post-transplant patients seems no longer to be a challenging patient group. In the ALLY-1 study (sofosbuvir + daclatasvir + ribavirin) lower response rates were particularly seen for CHILD C patients and patients with low albumin levels.
· 24 weeks SOF + RBV HCV therapy in heterogeneous real-world populations with genotype 3 are associated with low SVR12 rates in the presence of cirrhosis and prior HCV treatment failure highlighting the need for more efficacious treatment options for GT3 patients.
· In decompensated cirrhosis 12 weeks therapy with sofosbuvir plus an NS5A inhibitor +/- ribavirin is effective with most patients achieving SVR12. Patients infected with Genotype 1 HCV respond well with >80% achieving SVR4, response rates were slightly reduced in patients with G3 with daclatasvir + sofosbuvir achieving higher SVR rates than the combination of ledipasvir/sofosbuvir (English NHS EAP).
· In GT3 patients, including a large proportion of treatment-experienced patients with cirrhosis, SOF+PEG/RBV for 12 weeks resulted in the highest SVR12 rates observed to date in a Phase 3 study. Overall and in all subgroups, GT3 patients receiving 24 weeks of SOF+RBV had higher SVR12 rates than those receiving 16 weeks of treatment, confirming that 24 weeks is the optimal duration for this combination in GT3 patients. SOF+PEG/RBV for 12 weeks was well tolerated with a high rate of treatment completion. These data suggest SOF+PEG/RBV treatment should still be considered for IFN-eligible GT3 patients.
· High rates of SVR (95%) were achieved in patients with HCV GT1, 4 and 6 and HIV coinfection receiving the all-oral, fixed-dose combination of GZR/EBR for 12 weeks. Similar high SVR rates (96%) respectively were observed for 12 weeks HCV therapy with ledipasvir/sofosbuvir (ION-4 study) and sofosbuvir + daclatasvir (ALLY-2) also in HIV coinfection.
· 8 weeks real-life single-center experience with treating HCV with ledipasvir/sofosbuvir confirms that shorter treatment durations may be well feasible even outside clinical trials in patients with a HCV viral load < 6 Mill IU/ml and no cirrhosis.
· The fixed-dose combination of grazeprovir/elbasvir achieved SVR12 by 95% of treatment-naïve patients with high efficacy across GT1, GT4, and GT6 infection and also high efficacy in cirrhotics (SVR12 = 97.1%). Overall virologic failure rate was 4%; lower efficacy observed only among patients with high viral load and primarily with baseline GT1a RAVs that cause >5-fold potency reduction to elbasvir. The new DAA combination was generally well-tolerated, with a similar safety profile in cirrhotic and non-cirrhotic patients.
· GZR + EBR + RBV x 12 weeks resulted in an impressive SVR12 rate of 96.2% in patients who had failed previous PR plus an earlier-generation PI.
· GS-9857 with SOF/GS-5816 was well tolerated for 4 or 6 weeks. Overall, treatment for 6 weeks was highly efficacious in GT1 treatment-naïve patients without cirrhosis. Treatment-experienced patients or those with cirrhosis may benefit from longer durations of therapy.
· ACH-3102 plus sofosbuvir (used as a proxy for ACH-3422) achieved SVR12 in 100% of all HCV GT1 non-cirrhotic patients treated in a pilot trial. Therefore, this becomes the first study to report 100% SVR12 in patients using a two-drug combination for 6 weeks.
· In GT1a patients who have developed virological failure under DAA therapy the proportion of subjects with RAVs in NS3 declined over time, while most subjects with RAVs in NS5A and NS5B had these variants detectable through Post-Treatment Week 48.
· NS5A variants can persist for >96 weeks post-treatment in subjects who relapse to regimens containing NS5A inhibitor suggesting high fitness of the NS5A RAVs.
· Number of RAVs after failure to a NS5A inhibitor based therapy matter. In particular detection of the L31M and/or Y93H/N is associated with a higher risk for developing virological failure if retreatment is done with ledipasvir/sofosbuvir. This implies that after all oral DAA therapy including a NS5A inhibitor genotypic resistance testing indeed can be helpful to select the most active and promising retreatment therapy.
References
1. EASL Recommendations on Treatment of Hepatitis C 2015. European Association for the Study of the Liver. J Hepatol 2015
2. Pockros P, et al. Safety of ombitasvir/paritaprevir/ritonavir plus dasabuvir for treating HCV GT1 infection in patients with severe renal impairment or end-stage renal disease: The RUBY-I Study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract L01
3. Roth D et al.: C-SURFER: Grazoprevir plus elbasvir in treatment-naïve and treatment experienced patients with hepatitis C virus genotype 1 infection and chronic kidney disease. 50th International Liver Congress (ILC), April 22-26, 2015, Vienna, Austria; abstract LP02
4. Yeh WW, et al. Hepatology. 2014;60(suppl 4):1940.
5. Manns M, et al.: Ledipasvir/Sofosbuvir with ribavirin is safe and efficacious in decompensated and post liver transplantation patients with HCV infection: Preliminary results of the prospective SOLAR 2 trial. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract G02
6. Poordad F, et al.: Daclatasvir, sofosbuvir, and ribavirin combination for HCV patients with advanced cirrhosis or posttransplant recurrence: Phase 3 ALLY-1 study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract L08
7. Reddy R, et al.: All oral HCV therapy is safe and effective in patients with decompensated cirrhosis: Interim report from the HCV-TARGET real world experience. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O008
8. Jacobson IM. Et al.: Effiacacy and safety of grazoprevir and elbasvir in hepatitis C genotype 1-infected patients with CHILD-PUGH class B cirrhosis (C-SALT part A). 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O008
9. Nelson D et al.: All-oral 12-week Combination Treatment With Daclatasvir (DCV) and Sofosbuvir (SOF) in Patients Infected with HCV Genotype (GT) 3: ALLY-3 Phase 3 Study. 65th Annual Meeting of the American Association for the Study of Liver diseases, November 7-11, 2014, Boston, USA; abstract LB-3
10. Alqahtani S, et al.: Safety and effectiveness of sofosbuvir-based regimens for the treatment of hepatitis C genotype 3 and 4 infections: internal analysis of a prospective observational study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract PO840
11. Kowdley K, et al.: Efficacy evaluation of 24 week SOF + RBV in a heterogeneous, real-world population of genotype 3HCV patients; data from the TRIO network. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract PO867
12. Foster G, et al.: Treatment of decompensated HCV cirrhosis in patients with diverse genotypes: 12 weeks of sofosbuvir and NS5A inhibitors with/without ribavirin is effective in HCV genotypes 1 and 3. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O002
13. Hezode C, et al.: Daclatasvir plus sofosbuvir with or without ribavirin in patients with HCV genotype 3 infection: interim analysis of a French multicentre compassionate use program. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP05
14. Foster G, et al.: Sofosbuvir + PEG-Interferon/ribavirin for 12 weeks vs sofosbuvir + ribavirin 16 or 24 weeks in genotype 3 HCV infected patients and treatment-experienced cirrhotic patients with genotype 2 HCV: The BOSON study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract PO5
15. Naggie S, et al.: Ledipasvir/Sofosbuvir for 12 Weeks in Patients Coinfected With HCV and HIV-1. 22nd Conference on Retroviruses and Opportunistic Infections, February 23-26, 2015; abstract 152LB
16. Wyles D, et al.: Daclatasvir in Combination with Sofosbuvir for HIV/HCV Coinfection: ALLY-2 Study. 22nd Conference on Retroviruses and Opportunistic Infections, February 23-26, 2015; abstract 151LB
17. Wyles DL, et al.: Daclatasvir plus sofosbuvir for treatment of HCV genotypes 1-4 in HIV-HCV coinfection: The ALLY-2 study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP01
18. Cooper C, et al. A single tablet regimen of ledipasvir/sofosbuvir for 12 weeks in HCV genotype 1 or 4 infected patients with HIV-1 coinfection: The phase 3 ION-4 study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P1353
19. Rockstroh JK, et al. C-EDGE coinfected phase 3 study of grazoprevir/elbasvir in patients with HCV/HIV. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P0887
20. Buggisch P, et al.: Real-world effectiveness of Ledipasvir/Sofosbuvir 8 weeks chronic HCV treatment. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP32
21. Kwo P, et al.: A Pphase 3, randomized, open-label study to evaluate the efficacy and safety of 8 and 12 weeks of simeprevir (SMV) plus sofosbuvir (SOF) in treatment-naïve and experienced patients with chronic HCV genotype 1 infection without cirrhosis: OPTIMIST-1. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP14
22. Lawitz E. et al.: A phase 3, open-label, single-arm study to evaluate the efficacy and safety of 12 weeks of simeprevir (SMV) plus sofosbuvir (SOF) in treatment-naïve or -experienced patients with chronic HCV genotype 1 infection and cirrhosis: OPTIMIST-2. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP04
23. Conway B, et al.: MALACHITE-I: phase 3b trial of ombitasvir/paritaprevir/r and dasabuvir +/- ribavirin or telaprevir +PEG-Interferon/ribavirin in treatment-naïve adults with HCV genotype 1. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract PO842
24. Doss W, et al., An open-label study to evaluate the efficacy and safety of co-formulated ombitasvir/paritaprevir/ritonavir with ribavirin in adults with chronic HCV genotype 4 infection in Egypt. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P1351
25. Hezode C, et al., Ombitasvir plus paritaprevir plus ritonavir with or without ribavirin in treatment-naive and treatment-experienced patients with genotype 4 chronic hepatitis C virus infection (PEARL-I): a randomised, open-label trial. Lancet. 2015 Mar 30. pii: S0140-6736(15)60159-3.
26. Alqahtani, S., et al.: Safety and effectiveness of sofosbuvir-based regimens for the treatment of hepatitis C genotype 3 and 4 infections: interim analysis of a prospective observational study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P0840
27. Abergel A, et al.: Ledipasvir/Sofosbuvir treatment results in high SVR rates in patients with chronic genotype 4 and 5 HCV infection. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O056
28. Zeuzem S, et al.: The phase 3 C-EDGE treatment-naive (TN) study of a 12-week oral regimen of grazoprevir (GZR, MK-5172)/elbasvir (EBR, MK-8742) in patients with chronic HCV genotype (GT) 1, 4, or 6 infection. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract G07
29. Zeuzem S, et al: Grazoprevir-Elbasvir Combination Therapy for Treatment-Naive Cirrhotic and Noncirrhotic Patients With Chronic HCV Genotype 1, 4, or 6 Infection: A Randomized Trial. Ann Intern Med. 2015 Apr 24. doi: 10.7326/M15-0785. [Epub ahead of print]
30. Kwo P, et al.: Efficacy and safety of grazpoprevir/elbasvir +/- RBV for 12 weeks in patients with HCV G1 or G4 infection who previously failed PEG-interferon/RBV: C-EDGE treatment experienced trial. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P0886
31. Forns X, et al.: Grazoprevir (GZR)/elbasvir (EBR) plus ribavirin (RBV) for chronic hepatitis C virus genotype-1 infection after failure of combination therapy containing a direct-acting antiviral agent. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P0886
32. Forns X, et al.: Grazoprevir/Elbasvir plus ribavirin for chronic HCV genotype-1 infection after failure of combination therapy containing a direct-acting antiviral Agent. J Hepatol. 2015 Apr 17. pii: S0168-8278(15)00291-3. doi: 10.1016/j.jhep.2015.04.009. [Epub ahead of print]
33. Poordad F, et al.: C-SWIFT: Grazoprevir/elbasvir + sofosbuvir in cirrhotic and noncirrhotic, treatmen-naïve patients with hepatitis C genotype 1 infection, for durations of 4,6 or 8 weeks and genotype 3 inection for durations of 8 or 12 weeks. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O006
34. Gane E et al.: Efficacy of 12 or 18 weeks of grazoprevir plus elbasvir with ribavirin in treatment-naïve, non-cirrhotic HCV genoype 3 infected patents. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract P0776
35. Gane E, et al.: Safety and efficacy of short-duration treatment with GS-9857 combined with Sofosbuvir/GS-5816 in treatment-naïve and DAA-experienced genotype 1 patients with and without cirrhosis. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP03
36. Gane E, et al.: Sustained virological response ater AFTER ACH-3102 and sofosbuvir treatment for 8 or 6 weeks: A phase 2 "PROXY" study. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract LP06
37. Patel D, et al.: Achievement of SVR12 despite the presence of HCV variants resistant to first generation NS5A inhibitors in genotype-1 hepatitis C patients after 8-week therapy of ACH-3102 in combination with sofosbuvir. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract PO885
38. Krishnan P et al.: Long-term follow-up of treatment-emergent resistance-associated variants in NS3, NS5A and NS5B with paritaprevir/r-, ombitasvir- and dasabuvir-based regimens. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O057
39. Wyles D et al.: Long-term persistence of HCV variants after treatment with NS5A inhibitor ledipasvir. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O057
40. Lawitz E et al.: Retreatment of patients who failed 8 or 12 weeks of ledipasvir/sofosbuvir-based regimens with ledipasvir/sofosbuvir for 24 weeks. 50th International Liver Congress (ILC), April 22-26, Vienna, Austria; abstract O005
|
| |
|
 |
 |
|
|