 |
 |
 |
| |
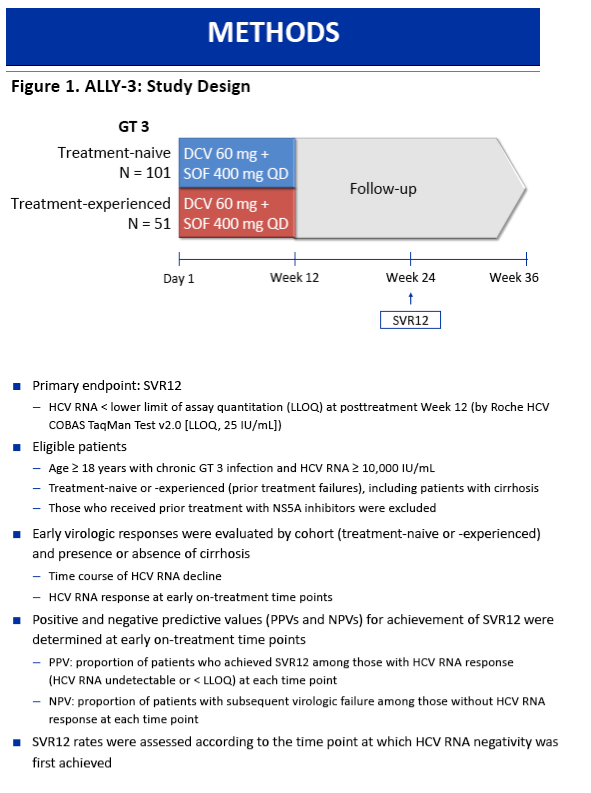
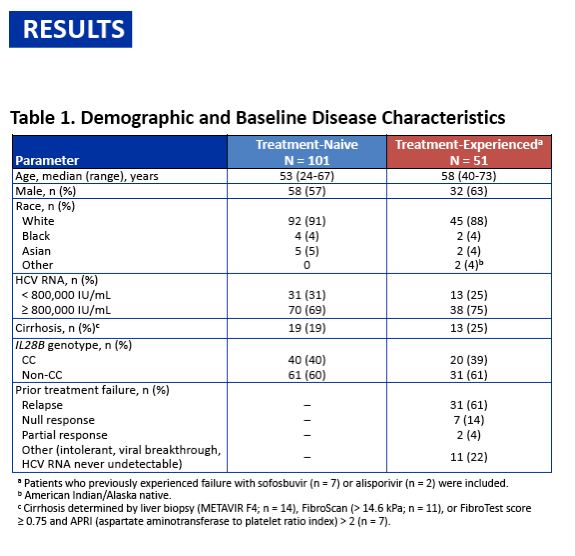
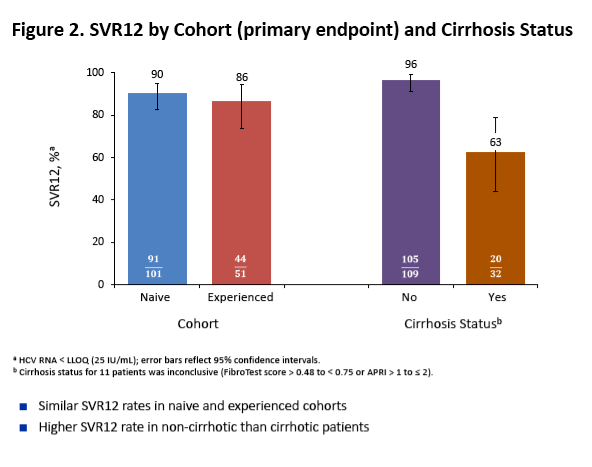
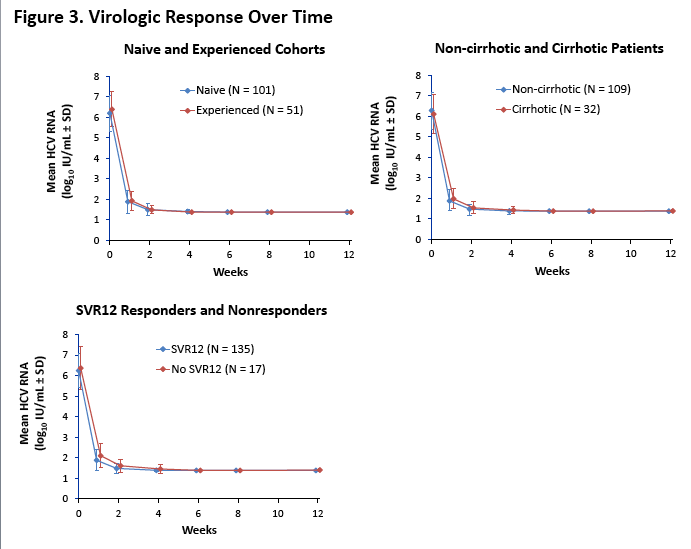
On-Treatment HCV RNA as a Predictor of Sustained Virologic Response in HCV Genotype 3-Infected Patients Treated With Daclatasvir and Sofosbuvir: A Subanalysis of the Phase 3 ALLY-3 Study
|
| |
| |
Reported by Jules Levin
EASL 2015 April 22-26 Vienna Austria
Kowdley K,1 Nelson D,2 Lalezari J,3 Box T,4 Gitlin N,5 Poleynard G,6 Rabinovitz M,7 Ravendhran N,8 Sheikh A,9 Siddique A,10 Bhore R,11 Yin PD,12 Noviello S,11 Rana K12
1Swedish Medical Center, Seattle, WA, USA; 2University of Florida, Gainesville, FL, USA; 3Quest Clinical Research, San Francisco, CA, USA; 4Clinical Research Centers of America, LLC, Murray, UT, USA; 5Atlanta Gastroenterology Associates, Atlanta, GA, USA; 6Digestive Health Specialists, Winston-Salem, NC, USA; 7University of Pittsburgh, Pittsburgh, PA, USA; 8Digestive Disease Associates, Baltimore, MD, USA; 9Gastrointestinal Specialists of Georgia, Marietta, GA, USA; 10Virginia Mason Medical Center, Seattle, WA, USA; 11Bristol-Myers Squibb Research and Development, Princeton, NJ, USA; 12Bristol-Myers Squibb Research and Development, Wallingford, CT, USA
EASL: Daclatasvir Plus Sofosbuvir for Treatment of HCV Genotypes 1-4 in HIV-HCV Coinfection: The ALLY-2 Study - (04/23/15)
All-oral 12-week treatment with daclatasvir plus sofosbuvir in patients with hepatitis C virus genotype 3 infection: ALLY-3 phase III study - (04/15/15)
Hepatology 2015
APASL: All-Oral 12-Week Combination Treatment With Daclatasvir and Sofosbuvir in Patients Infected With HCV Genotype 3: ALLY-3 Phase 3 Study - (03/18/15)


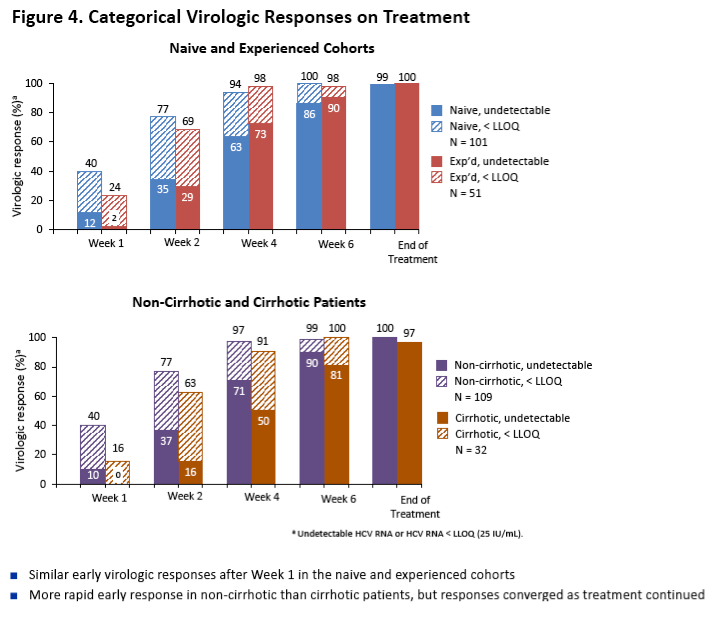
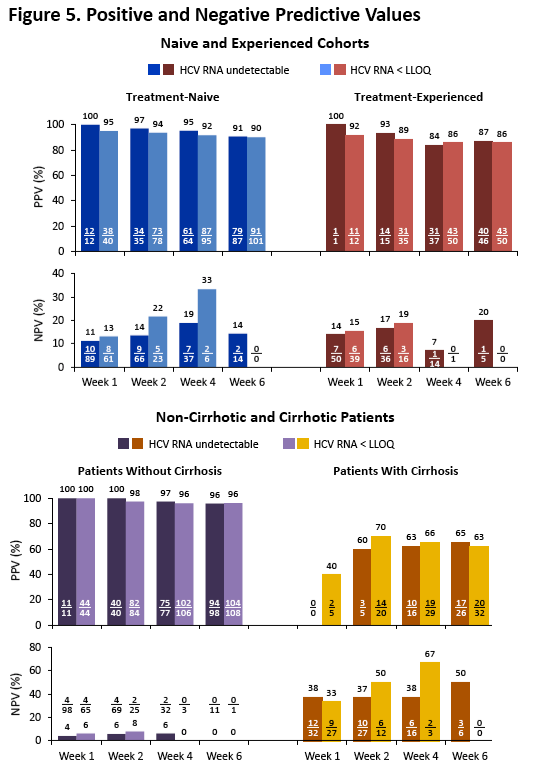
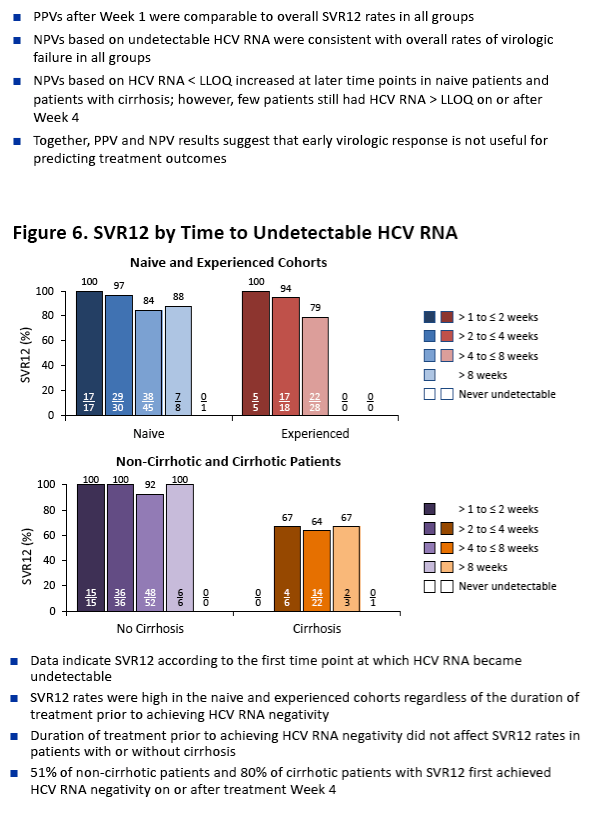

|
| |
|
 |
 |
|
|