 |
 |
 |
| |
Ipergay Analysis Suggests Both Pre- and Postsex TDF/FTC PrEP Doses Needed for Full Protection
|
| |
| |
IAS 2015, July 19-22, 2015, Vancouver
Mark Mascolini
Further analysis of the Ipergay trial of pre- and postsex tenofovir/emtricitabine (TDF/FTC) for preexposure prophylaxis (PrEP) suggests that the before and after doses are both needed for full protection against HIV acquisition [1].
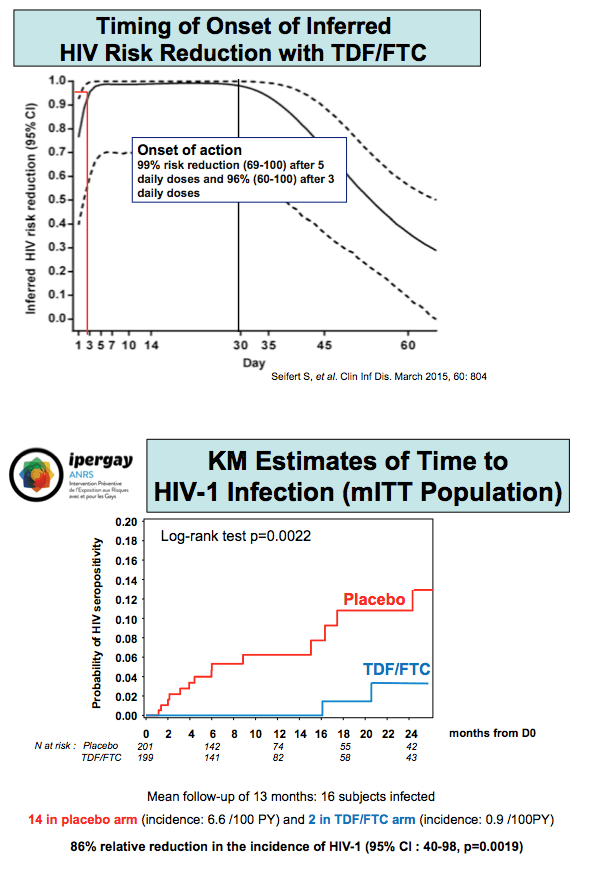
Like the British PROUD study, the 400-man double-blind, placebo-controlled Ipergay trial demonstrated 86% protection from HIV acquisition with TDF/FTC PrEP [2]. Unlike other PrEP trials in gay men, Ipergay asked participants to take two TDF/FTC pills before sex and two after sex. Principal investigator Jean-Michel Molina (University of Paris) updated findings at IAS 2015.
Researchers instructed sexually active HIV-negative gay and bisexual men to take two TDF/FTC tablets 2 to 24 hours before sex, one tablet 24 hours after sex, and one tablet 24 hours after that. Thus men would take four tablets over 3 days to cover one sex act. All men reported condom-free anal sex with at least 2 partners in the past 6 months. They had a median of 10 sex acts in the past 4 weeks and a median of 8 partners in the past 2 months.
Through an average 13 months of follow-up, 16 men became infected, 14 in the placebo arm (incidence 6.6 per 100 person-years) and 2 in the TDF/FTC arm (incidence 0.9 per 100 person-years). Those rates translated into an 86% relative reduction in HIV incidence (95% confidence interval 40% to 98%, P = 0.0019). The 2 infected men in the TDF/FTC arm had not taken their PrEP pills for months while continuing to have sex.
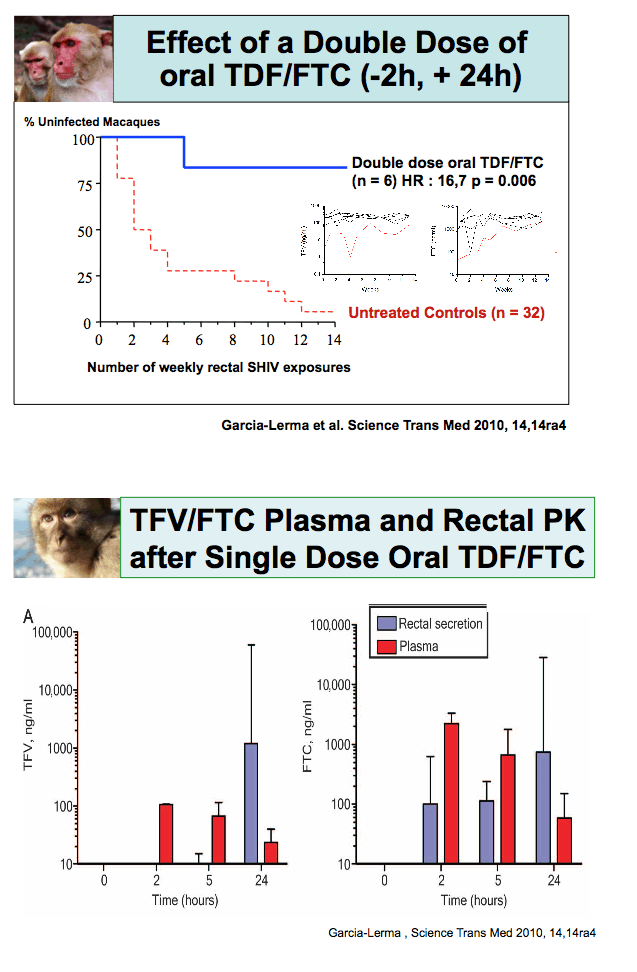
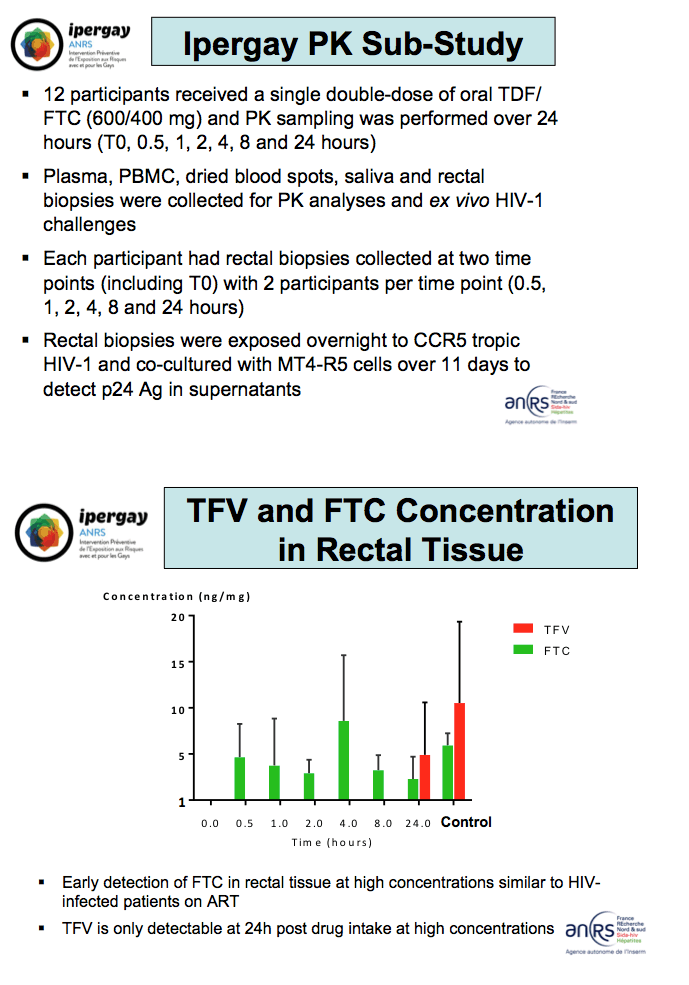
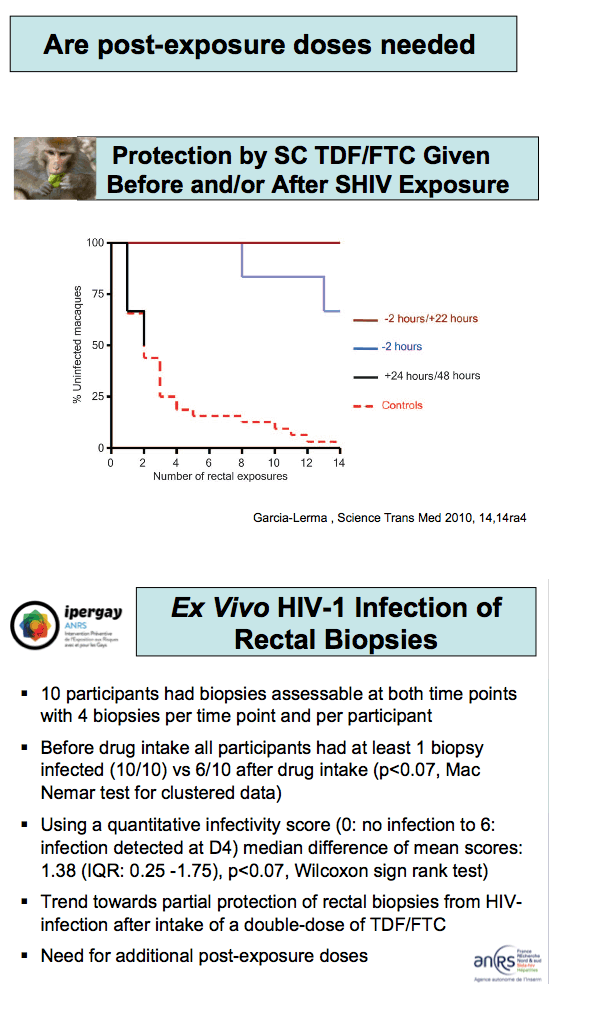
In a substudy 12 men received a single double dose of TDF/FTC and gave samples for drug level analysis seven times over 24 hours. All participants had rectal biopsies at two points, with all participants giving a time 0 sample and two participants biopsied at each later time point (0.5, 1, 2, 4, 8, and 24 hours). The investigators exposed rectal biopsies to CCR5-tropic HIV-1 cocultured with MT4-R5 cells over 11 days.
Early detection of FTC at high concentrations in rectal tissue reflected levels measured in HIV-infected people taking an FTC-containing regimen. Tenofovir became detectable at high concentrations only 24 hours after dosing.
Ten men all had four assessable biopsies per time point. Before TDF/FTC dosing 10 of 10 participants had at least one biopsy that became infected with HIV, compared with 6 of 10 biopsies that became infected after dosing (P < 0.07). A quantitative infectivity score with 0 indicating no infection and 6 indicating infection at day 4 showed that the median difference between mean before and after scores stood at 1.38 (interquartile range 0.25 to 1.57, P < 0.07). Molina suggested this trend may indicate a need for additional postsex doses.
Molina advanced the following conclusions:
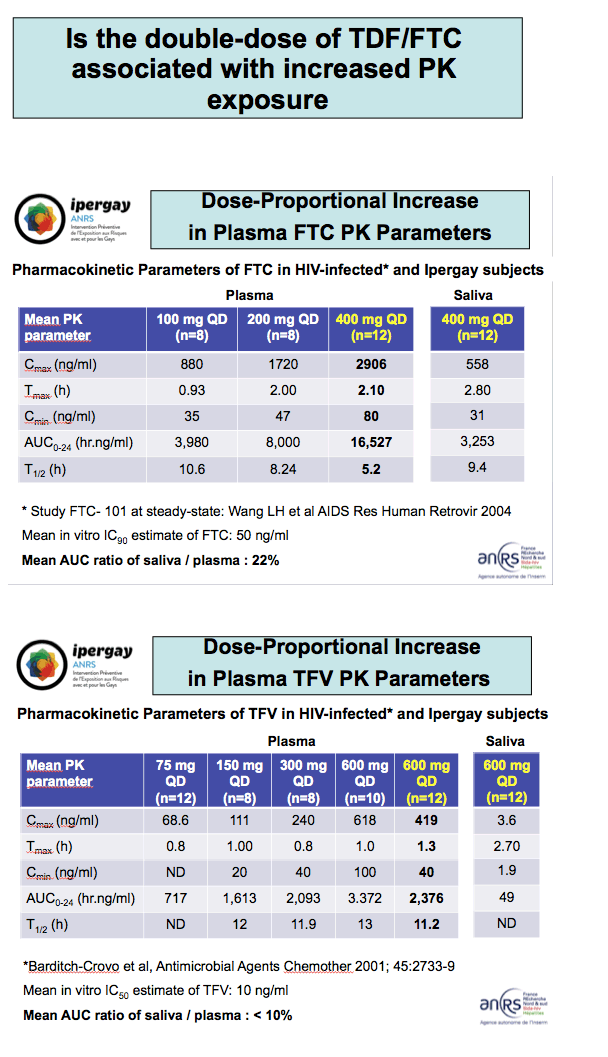
-- A double dose of TDF/FTC leads to rapid and high concentrations of FTC and tenofovir in plasma.
-- FTC can achieve rapid and high concentrations in rectal tissue and saliva.
-- Pre- and postsex doses "both appear to be critical for providing full protection against HIV acquisition."
He cautioned that the effectiveness of as-needed (coitally dependent) PrEP in people who have sex less often than Ipergay men remains to be demonstrated.
References
1. Molina JM. Coitally dependent TDF/FTC in MSM: Updates on PrEP efficacy in Ipergay. IAS 2015. 8th Conference on HIV Pathogenesis, Treatment and Prevention. July 19-22, 2015. Vancouver. Abstract MOSY0102.
2. Molina JM, Capitant C, Spire B, et al. On demand PrEP with oral TDF-FTC in MSM: results of the ANRS Ipergay trial. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 23. http://www.natap.org/2015/CROI/croi_07.htm
----------------
Coitally-Dependent TDF/FTC in MSM
Updates on PrEP Efficacy in IPERGAY
Jean-Michel Molina
and the ANRS Ipergay Study Group
Hospital Saint-Louis and University of Paris 7, Inserm U941, Paris, France


|
| |
|
 |
 |
|
|