 |
 |
 |
| |
A Comparison of Daily and Non-daily
Pre-exposure Prophylaxis Dosing in Thai Men Who Have Sex With Men, Bangkok - HPTN 067 / ADAPT Study
|
| |
| |
Reported by Jules Levin
IAS 2015 Vancouver July 19-13
Timothy H. Holtz, Anupong Chitwarakorn, Marcel E. Curlin, James Hughes, K. Rivet Amico, Craig Hendrix, Bonnie J. Dye, Peter L. Anderson, Maoji Li, Vanessa Elharrar, Susan H. Eshleman, Michael Stirratt, Robert M. Grant and the Bangkok HPTN 067/ADAPT Study Team
WEBCAST:
https://www.youtube.com/watch?v=3x1vaOTEUKA
In summary, large proportions of Thai MSM took PrEP consistently when given access in a clinical setting, with coverage and adherence highest in the daily arm.
However, at our site, time-driven dosing among MSM and TGW provided coverage of sexual exposures that was comparable to daily dosing, with only slightly less adherence, and spared a considerable number of tablets required.
In our indicator of long term drug coverage, namely PBMCs, tenofovir drug detection was high in all three arms at weeks 10 and 30, indicating the feasibility of non-daily regimens in this population.
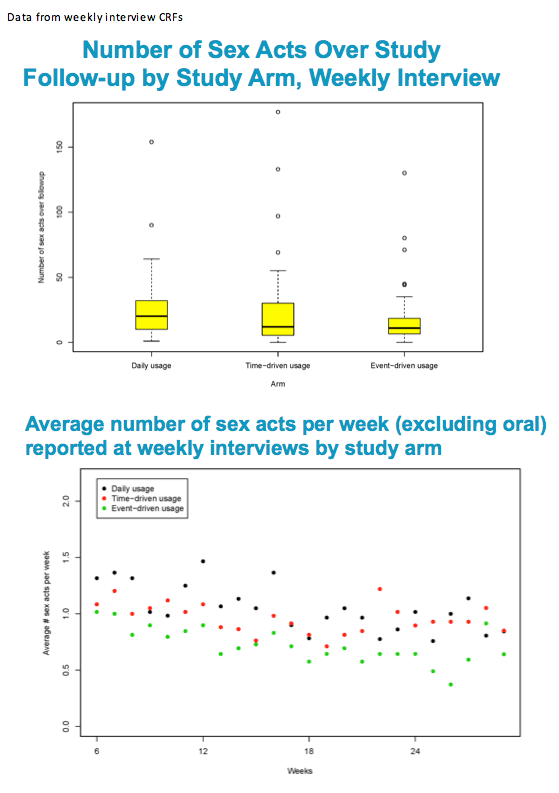
And for those of you with Ipergay in mind, and trying to compare the populations, in data that was not shown, I can tell you that non-oral sex events in this population occurred on average of once per week.

Per CDC Clinical Practice Guidelines, and US Public Health Service Guidelines, daily dosing has proven effective at reducing the risk of HIV infection in multiple clinical trials, and is the FDA-approved regimen. It provides the highest level of protection, the most forgiveness for missed doses, and fosters daily habit formation.
Among those with few exposures, non-daily dosing of PrEP could result in fewer tablets required to achieve comparable coverage. However, one would lose that margin of forgiveness for missed doses [[as we found that drug concentrations were lower]]
We note that coverage as defined in this study is not yet known to be effective.
We have shown non-daily regimens to be feasible, and if proven effective in future research, could provide an alternative for those who are able to adhere.

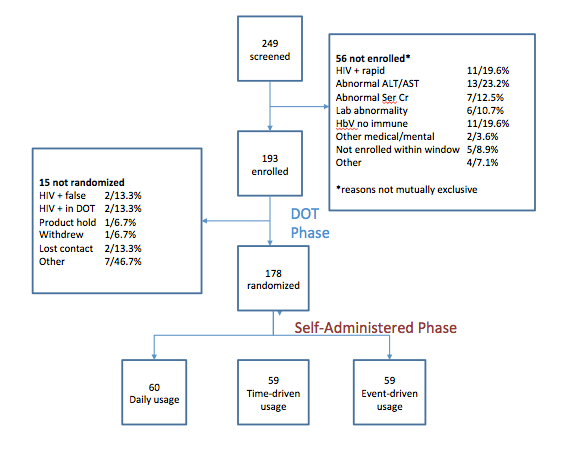
In total, we screened 249 men and transgender women for entry
56 persons were not enrolled, most commonly for abnormal liver enzymes. Among these were 11 persons who tested positive for HIV, reflecting the fact that this was a very high risk population with previously undiagnosed HIV infection
After enrolling 193 MSM and TGW, 15 were not randomized, including 2 persons who seroconverted in the DOT Phase. CHANGED
We randomized 178 participants that were evenly distributed between three arms in the self administered phase of the study.
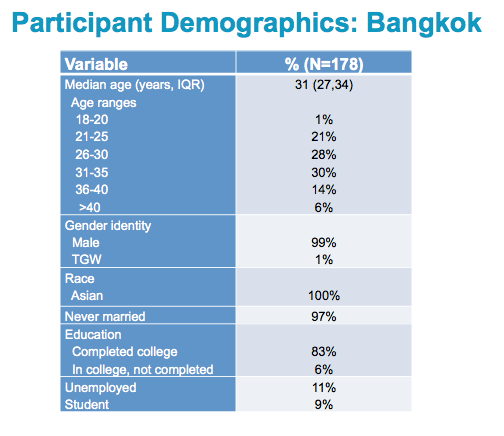
Among the 178 Thai participants, the median age was 31 years, with a min/max of 20 to 58 and an IQR of 27 and 34.
Thus, the majority of participants were in their early 30s, and nearly 90% had completed or were still in college.
There were 2 TGW participants, with the rest self identifying as Male. CHANGE
Only 11% were unemployed. Participants also included some students.
There were no statistically significant differences between the 3 arms for any of the participant demographic characteristics collected.

As Bob described, the primary outcomes in this trial were coverage of sex events, number of tablets required and taken, and self-reported side effects.
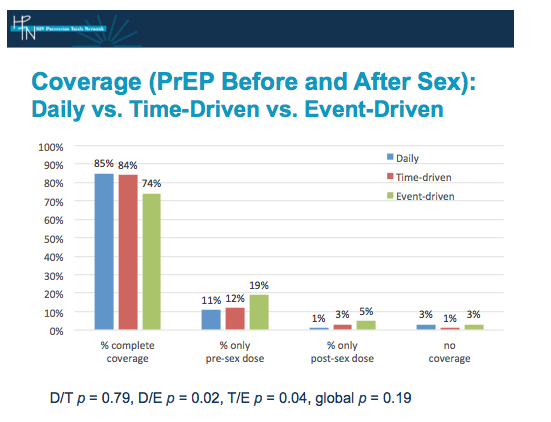
At our site, PrEP coverages were similar in arms D and T, at 85% vs 84%, with no statistically significant difference between these arms, with a p=0.79.
Both coverages were statistically significantly greater than in arm E at 74%, which is still arguably quite high for a non-daily regimen. CHANGE.
Among people randomized to non-daily dosing in all cohorts, the missed dose required for full coverage was usually the post-sex dose. CHANGED
The global p value for the 3 arms was 0.19, reflecting the nearly identical coverage of sex events between the daily arm, and the time-driven arm.

As Bob described, the primary outcomes in this trial were coverage of sex events, number of tablets required and taken, and self-reported side effects.
At our site, PrEP coverages were similar in arms D and T, at 85% vs 84%, with no statistically significant difference between these arms, with a p=0.79.
Both coverages were statistically significantly greater than in arm E at 74%, which is still arguably quite high for a non-daily regimen. CHANGE.
Among people randomized to non-daily dosing in all cohorts, the missed dose required for full coverage was usually the post-sex dose. CHANGED
The global p value for the 3 arms was 0.19, reflecting the nearly identical coverage of sex events between the daily arm, and the time-driven arm.
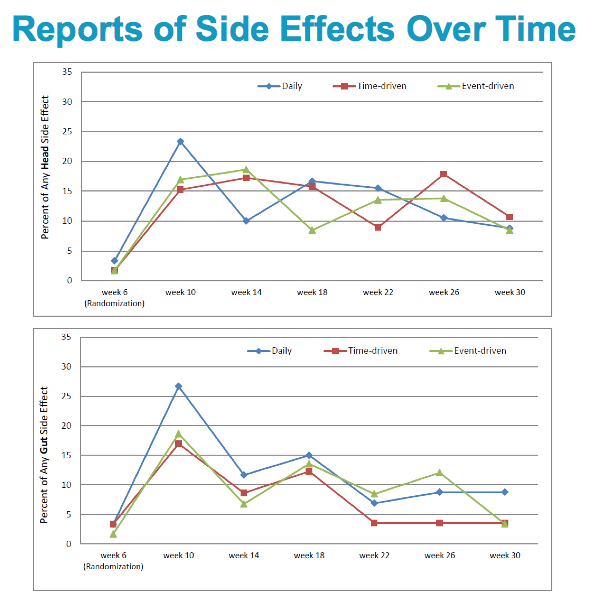
The neurologic and GI side effect proportions shown here are EVER occurring during the follow up period. Reported side effects were not uncommon, but were generally mild, and not substantially different in the daily and non-daily arms.
Over time, all side effect reports diminished greatly, and the proportion that were ascribed to the tablets was small.
CHANGE:
Self reported GI: (such as nausea, vomiting, diarrhea, gas, bloating and abdominal pain)
Self reported Neuro (such as headache, dizziness, lightheadedness)
As described previously, we had 2 seroconversions during the 6-week pre-randomization weekly DOT study phase. Both occurred at week 4.
Both had low levels of FTC detected and no detectable TVF in plasma. Both had low detectable levels of both FTC and TVF in PBMCs.
These 2 of 193 participants contributed 22.9 person-years; with an incidence 8.7% (1.1%-31.5%) during DOT phase.
This is comparable to the published incidence data from our MSM cohort and VCT services, and reflects the high risk population we enrolled.
0 of 178 participants over 94.8 person-years; incidence 0.0% (0.0%-3.9%) during self-administered (SA) phase
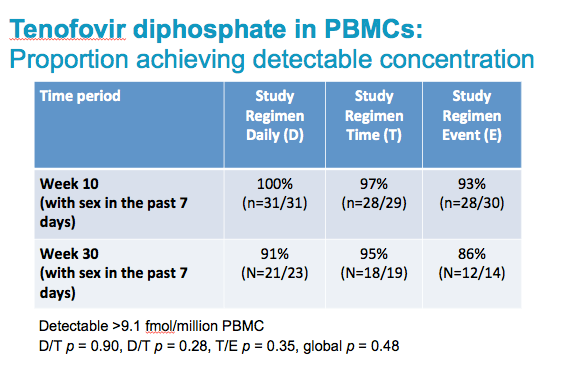
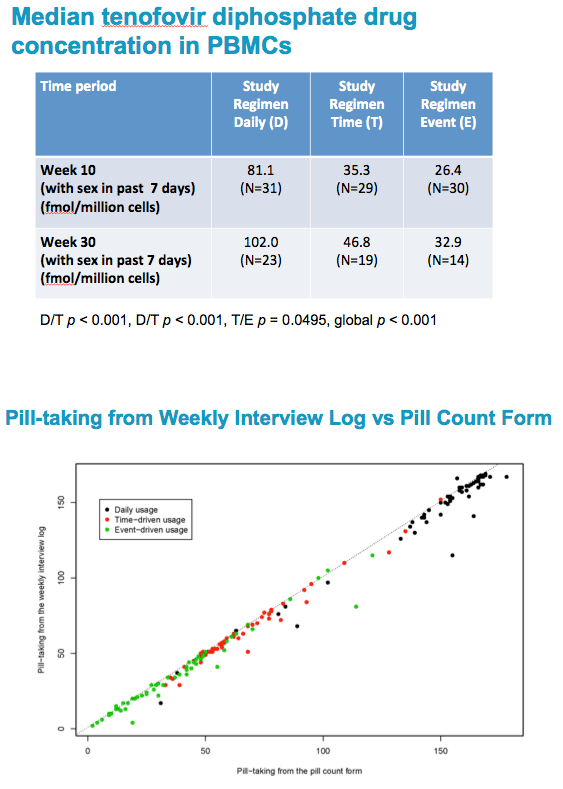
Among those reporting sex in the past 7 days, the proportion of MSM and TGW in BKK who had detectable drug levels in PBMCs associated with pre- and post sex dosing was equivalent in all three arms.
There was no statistically significant difference between any of the three daily or non-daily arms in our study, at either 10 or 30 weeks. With one exception, in each arm at both time periods, we found over 90% of participants with detectable concentration of tenofovir.
All participants in daily and time-driven arm were expected to have ≥9.1fmol/106 per regimen recommendations;
Only sexually active in past 7 days event-driven arm participants expected to have ≥9.1fmol/106 per regimen recommendations.
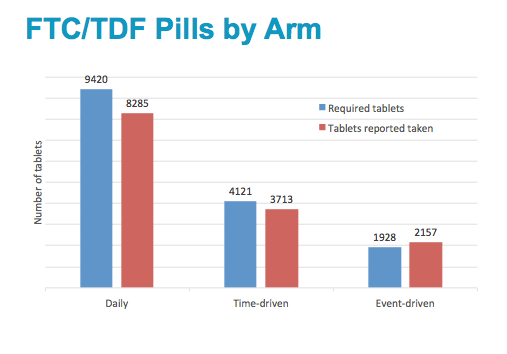
The histogram shows the number of required tablets per arm, and the tablets reported taken, either required or not required. The daily arm had the highest number of tablets required.
The number of required tablets was estimated for each arm based on the sex event history.
Side effects over time demonstrated a clear start up effect, with gradual diminished reports of neurologic and GI side effect reports with each FU visit.
Data from weekly interview CRFs

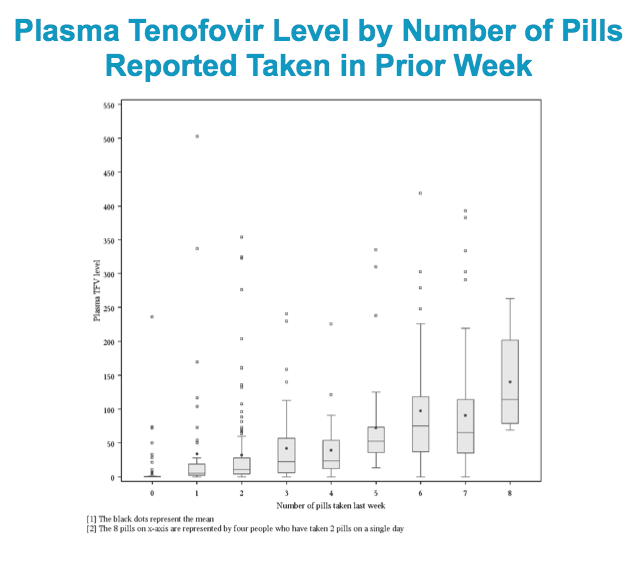
When we looked at TFV drug concentrations in PBMCs at Weeks 10 and 30, participants in the daily arm had the highest concentration of drug in the blood.
Higher concentrations of drug provide greater forgiveness for occasional missed doses, which is therefore greater in the daily arm.
The T and E arms had statistically significantly lower drug levels in PBMCs than did the D arm.
The median value for 4 doses per week is 18.8 (14.5-20.4) fmol/million cells, based on HPTN 066 (Anderson)
Femtomole = 10-15

Screening, 6, 18, 30
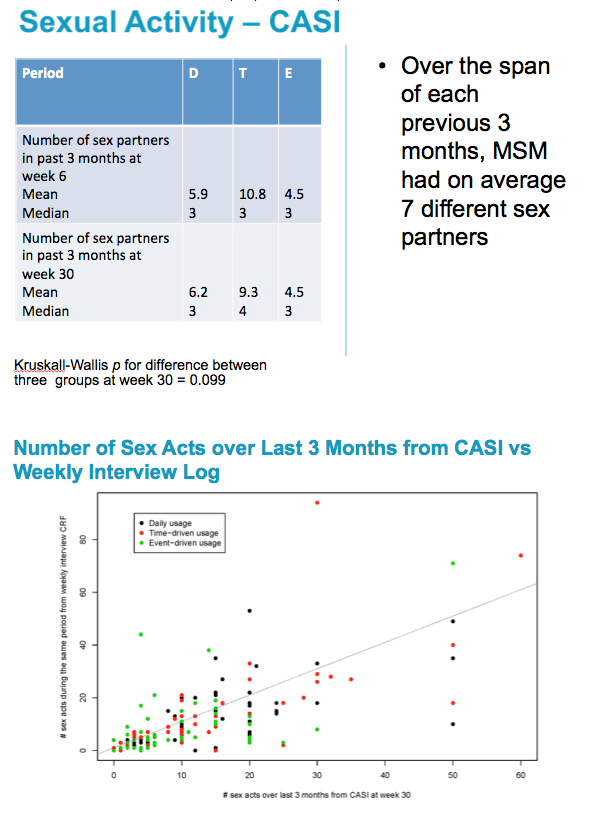
There is a significant difference between arms in the number of times they had sex in the three months prior to week 30 as reported in the CASI?
Difference is only between E and T/D
Hypothesis being tested is for a difference in the outcome (number of times had sex in past 3 months) between arms at week 30.
The test used was a Kruskall-Wallis test (nonparametric test)
There is no significant difference between the arms in the number of partners in the three months prior to week 30 as reported in the CASI
Hypothesis being tested is for a difference in the outcome (number of partners in the past 3 months) between arms at week 30.
The test used was a Kruskall-Wallis test (nonparametric test)


|
| |
|
 |
 |
|
|