 |
 |
 |
| |
Influence of Age on Outcomes in HIV-Infected Adults Initiating Tenofovir Alafenamide vs Tenofovir Disoproxil Fumarate With Elvitegravir, Cobicistat, and Emtricitabine
|
| |
| |
Reported by Jules Levin
IDSA 2015, October 7-11, 2015, San Diego, CA
Eric S. Daar,1 Michael Saag,2 Benoit Trottier,3 Amanda Clarke,4 Edwin DeJesus,5 David Parks,6 Eugenio Teofilo,7 Cynthia Brinson,8 Hal Martin,9 Susan Guo,9 Sandra Friborg,9 Marshall Fordyce9
1Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, CA; 2University of Alabama at Birmingham, AL; 3Clinique Médicale L'Actuel, Montreal, Quebec, Canada; 4Royal Sussex County Hospital, Brighton, UK; 5Orlando Immunology Center, Orlando, FL; 6Central West Healthcare, St. Louis, MO; 7Centro Hospitalar de Lisboa Central, Lisboa, Portugal; 8Central Texas Clinical Research, Austin, TX; 9Gilead Sciences, Inc., Foster City, CA
Background: As the HIV-infected population ages and has increasing prevalence of co-morbidities, efficacy and safety of antiretrovirals in older patients is of heightened importance. In the general population, E/C/F/TAF demonstrated similar efficacy and improved bone and renal safety compared to E/C/F/TDF.
Methods: Treatment naïve HIV-infected adults were randomized 1:1 to a single tablet regimen of E/C/F/TAF or E/C/F/TDF once daily in two Phase 3 double blind studies. Pre-specified pooled week 48 analyses of efficacy, renal safety, and bone mineral density (BMD) in those ≥ 50 years are described in this analysis.
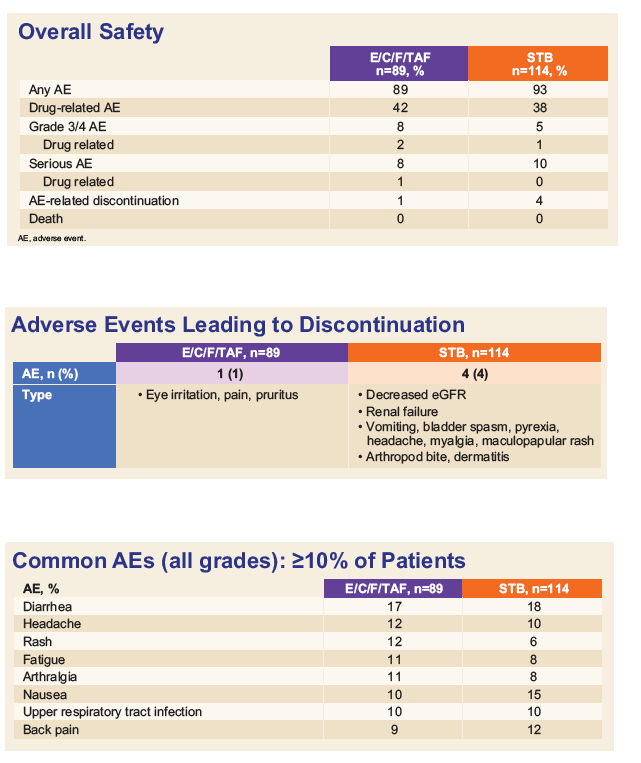
Results: Of 1,733 patients treated, 203 were ≥ 50 years of age; 89 randomized to E/C/F/TAF and 114 to E/C/F/TDF. Baseline viral load, CD4 counts, renal laboratories and BMD were similar.
Virologic success by FDA snapshot algorithm at week 48 was 94.4% for E/C/F/TAF vs. 91.2% for E/C/F/TDF, similar to efficacy in the entire study population.
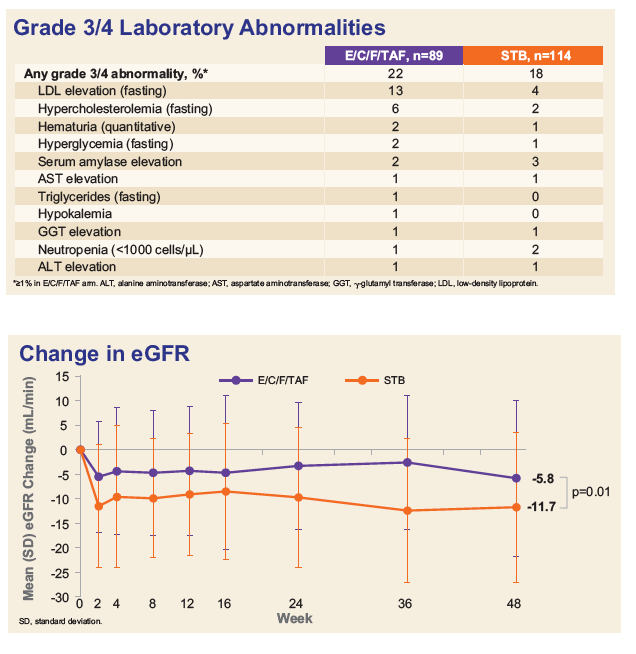
Adverse events leading to discontinuation was 1.1% for E/C/F/TAF vs. 3.5% for E/C/F/TDF. Mean change in estimated glomerular filtration rate at week 48 was -5.8 mL/min for E/C/F/TAF vs. -11.7 mL/min for E/C/F/TDF (p=0.010).
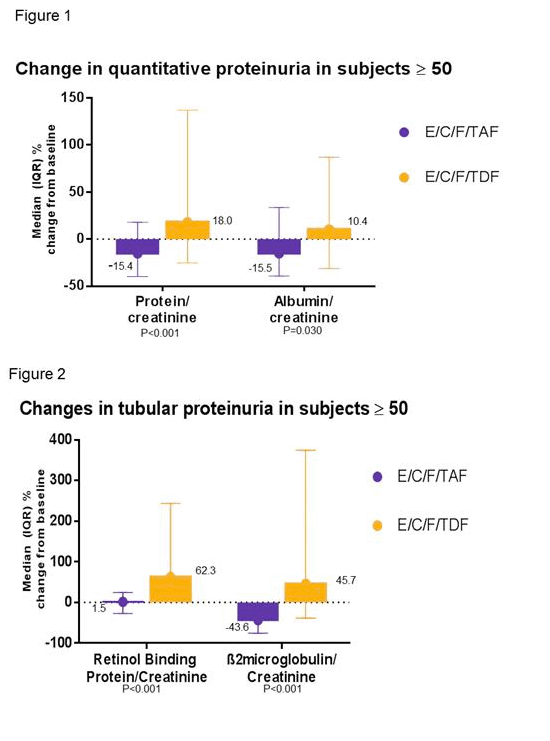
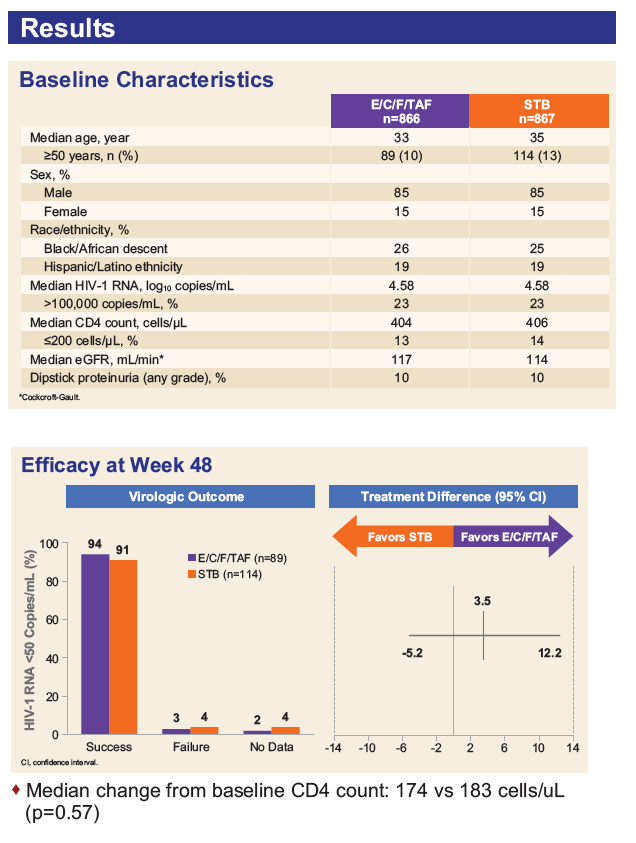
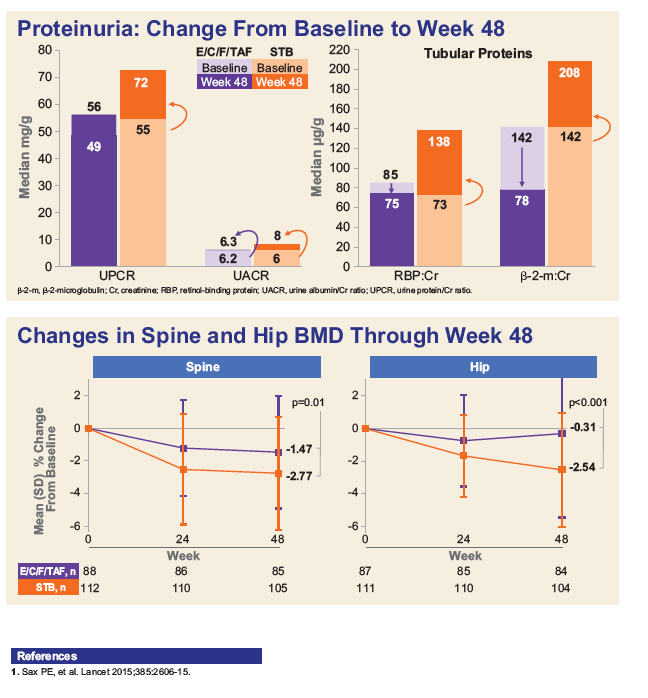
As shown in Figures 1 & 2, subjects ≥ 50 years had significantly improved renal tubular profiles with E/C/F/TAF vs. E/C/F/TDF.
Mean decrease in BMD was significantly less in the E/C/F/TAF vs. E/C/F/TDF arm; spine (-1.47% vs. -2.77%, p=0.011); hip (-0.31% vs. -2.52%, p<0.001).
Conclusion: Among subjects ≥ 50 years of age, those receiving 48 wks of E/C/F/TAF had comparable efficacy and improved bone and renal safety compared to those on E/C/F/TDF, as previously demonstrated in the overall study population. These findings demonstrate an important safety improvement of TAF relative to TDF in patients 50 years and older, which is of particular importance as the population living with HIV ages and experiences more non-AIDS related comorbidities.


|
| |
|
 |
 |
|
|