 |
 |
 |
| |
Standard Daclatasvir Dose OK With Darunavir/RTV or Lopinavir/RTV
|
| |
| |
slides below.......
16th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, May 26-28, 2015, Washington, DC
Mark Mascolini
Unlike atazanavir/ritonavir (ATV/r), neither darunavir/ritonavir (DRV/r) nor lopinavir/ritonavir (LPV/r) raised levels of the direct-acting antiviral daclatasvir enough to warrant daclatasvir dose reduction in a study of healthy volunteers [1]. And daclatasvir did not affect pharmacokinetics of the protease inhibitors (PIs) in people with HIV.
Daclatasvir, an HCV NS5A replication complex inhibitor, is licensed in Europe and Japan and under regulatory review in the United States. It is a substrate of the CYP3A4 enzyme, which ritonavir strongly inhibits. In a previous study in healthy volunteers, taking daclatasvir with ATV/r resulted in a 2.1-fold increase in daclatasvir area under the concentration-time curve (AUC) [2]. As a result the recommended daclatasvir dose with ATV/r is 30 mg daily instead of the standard 60 mg daily.
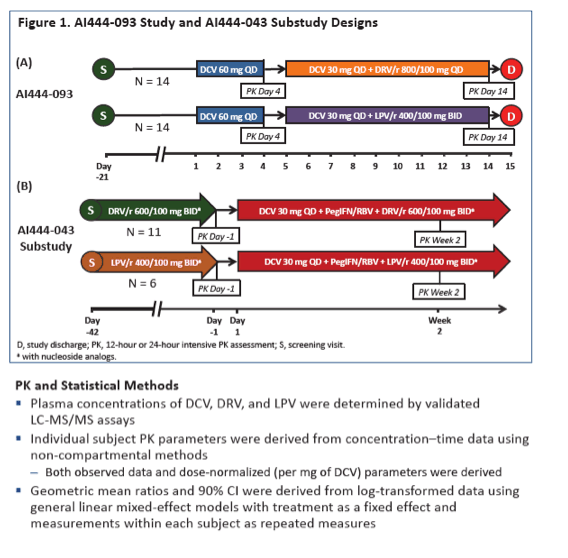
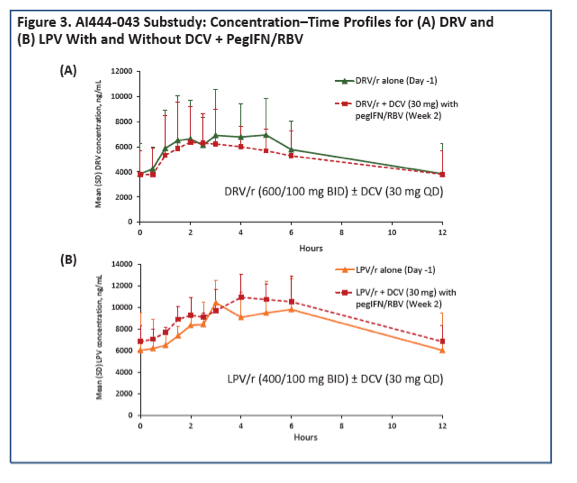
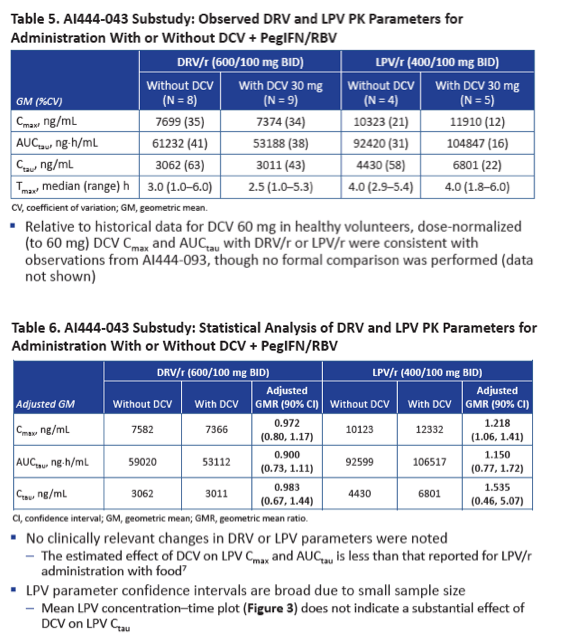
To see whether DRV/r and LPV/r have the same impact on daclatasvir, and whether daclatasvir affects levels of the PIs, Bristol-Myers Squibb (BMS) investigators conducted a two-part study. In the first part 28 healthy volunteers took daclatasvir at 60 mg once daily for 4 days. The dose dropped to 30 mg once daily on days 5 to 14 as 14 volunteers added 800/100 mg of DRV/r once daily and 14 added 400/100 mg of LPV/r twice daily. Researchers collected plasma samples on day 4 (daclatasvir alone) and day 14 (daclatasvir plus PI). The BMS team figured geometric mean ratios (GMRs) and 90% confidence intervals (CI) for daclatasvir with versus without PIs for observed and dose-normalized AUC and maximum concentration (Cmax). To determine the impact of daclatasvir on steady-state concentrations of DRV/r and LPV/r, the second part of the study focused on 11 HIV-positive people taking a stable 600/100-mg twice-daily DRV/r regimen and 5 taking a stable 400/100-mg twice-daily LPV/r regimen who started 30 mg of daclatasvir daily with peginterferon and ribavirin.
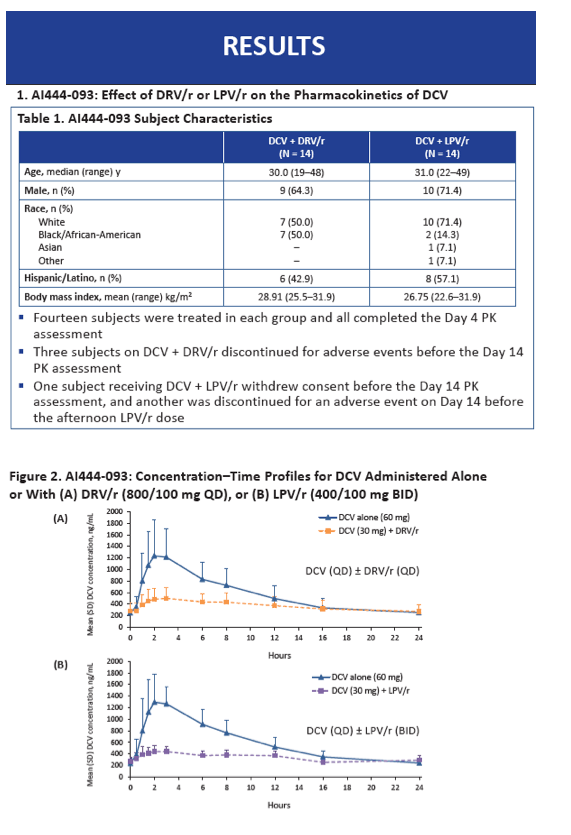
In the first part of the study, median age stood around 30 years, two thirds of volunteers were men, 61% were white, and 32% were black. All participants completed the first 4 days of study. Three people taking daclatasvir with DRV/r stopped because of adverse events before day 14, as did one taking daclatasvir with LPV/r. Another person in the LPV/r group withdrew consent before day 14.
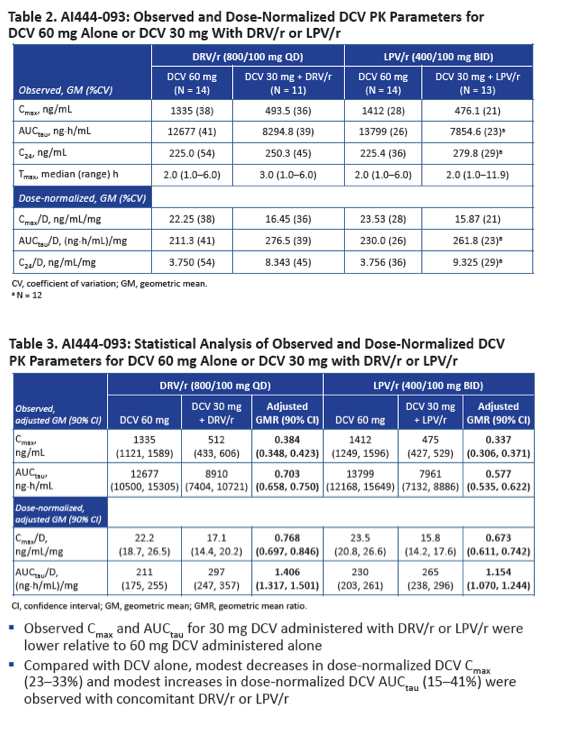
In this part of the study, observed AUC and Cmax for 30 mg of daclatasvir with DRV/r or LPV/r were lower than observed AUC and Cmax for 60 mg of daclatasvir alone. Dose-normalized daclatasvir AUC was moderately higher with DRV/r than with daclatasvir alone (GMR 1.406, 90% CI 1.317 to 1.501) and with LPV/r than with daclatasvir alone (GMR 1.154, 90% CI 1.070 to 1.244). Dose-normalized daclatasvir Cmax was lower with DRV/r than with daclatasvir alone (GMR 0.768, 90% CI 0.697 to 0.846) and with LPV/r than with daclatasvir alone (GMR 0.673, 90% CI 0.611 to 0.742).
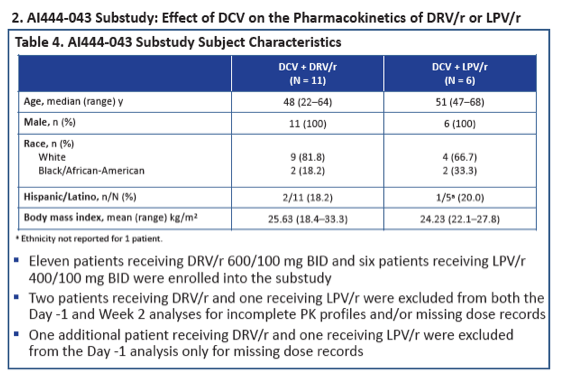
In the analysis of daclatasvir impact on DRV/r and LPV/r pharmacokinetics, the 17 study participants had a median age around 50, all were men, 76% were white, and 24% were black. GMRs determining DRV/r and LPV/r concentrations with versus without daclatasvir showed a negligible impact (for example, AUC GMR 0.900 for DRV/r and 1.150 for LPV/r).
The BMS team concluded that DRV/r and LPV/r do not boost daclatasvir concentrations to the degree seen with ATV/r and therefore that the standard 60-mg once-daily daclatasvir dose should be used with DRV/r or LPV/r. Daclatasvir dose should be cut to 30 mg once daily with ATV/r. Also, daclatasvir has little impact on pharmacokinetics and DRV/r or LPV/r, which can be given at standard doses when coadministered with daclatasvir.
References
1. Gandhi Y, Adamczyk R, Wang R, et al. Assessment of drug-drug interactions between daclatasvir and darunavir/ritonavir or lopinavir/ritonavir. 16th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. May 26-28, 2015. Washington, DC. Abstract 80.
2. Bifano M, Hwang C, Oosterhuis B, et al. Assessment of pharmacokinetic interactions of the HCV NS5A replication complex inhibitor daclatasvir with antiretroviral agents: ritonavir-boosted atazanavir, efavirenz and tenofovir. Antivir Ther. 2013;18:931-940.
--------------------------
Assessment of Drug-Drug Interactions Between Daclatasvir and Darunavir/Ritonavir or Lopinavir/Ritonavir
Reported by Jules Levin
16th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. Washington DC, May 26-28, 2015
Gandhi Y, Adamczyk R, Wang R, Stonier M, Kandoussi H, Hesney M, Liu Z, Ackerman P, Garimella T, Eley T
Bristol-Myers Squibb Research & Development, Princeton, NJ

EASL: Daclatasvir Plus Sofosbuvir for Treatment of HCV Genotypes 1-4 in HIV-HCV Coinfection: The ALLY-2 Study - (04/28/15)
Daclatasvir: Overview of Drug-Drug Interactions With Antiretroviral Agents and Other Common Concomitant Drugs........[Eley T et al. HIV DART 2014] ........http://www.natap.org/2014/HCV/121714_01.htm



|
| |
|
 |
 |
|
|