 |
 |
 |
| |
(HepDart) Ledipasvir/Sofosbuvir (LDV/SOF) for 8 Weeks in Genotype 1 (GT1) Treatment-Naïve (TN) Non-Cirrhotic (NC) Patients with HCV Viral load (VL) <6 million IU/ml (6M); A Comparative Analysis of the Phase-3 ION-3 Efficacy Data to Real World Effectiveness (RWE)
|
| |
| |
Reported by Jules Levin
HEP DART 2015, December 6-10, 2015, Wailea, HI
N Tsai1*, M Curry2, P Buggisch3, Y Lee4, M Natha5, E Eggleton5, B Kreter5, DM Brainard5, K Kowdley61Queens Medical Center, UH, Honolulu, Hawaii; 2Beth Israel, Boston, MA; 3Hamburg, Germany; 4TRIO Health Analytics; 5Gilead Sciences, Inc., Foster City, CA; 6Swedish Medical Center, Seattle, WA
AASLD: Failure with All-oral DAA Regimens: Real-world experience from the TRIO Network. Academic and community treatment of a real-world, heterogeneous population - (12/03/15)
AASLD: (HCV-TARGET) - Treatment Outcomes With 8, 12 and 24 Week Regimens of Ledipasvir/Sofosbuvir for the Treatment of Hepatitis C Infection: Analysis of a Multicenter Prospective, Observational Study - (12/03/15)
EACS: Sofosbuvir plus ledipasvir for 8 weeks in HCV-mono- and HIV-HCV-coinfected patients - Results from the German hepatitis C cohort (GECCO) (10/27/15)



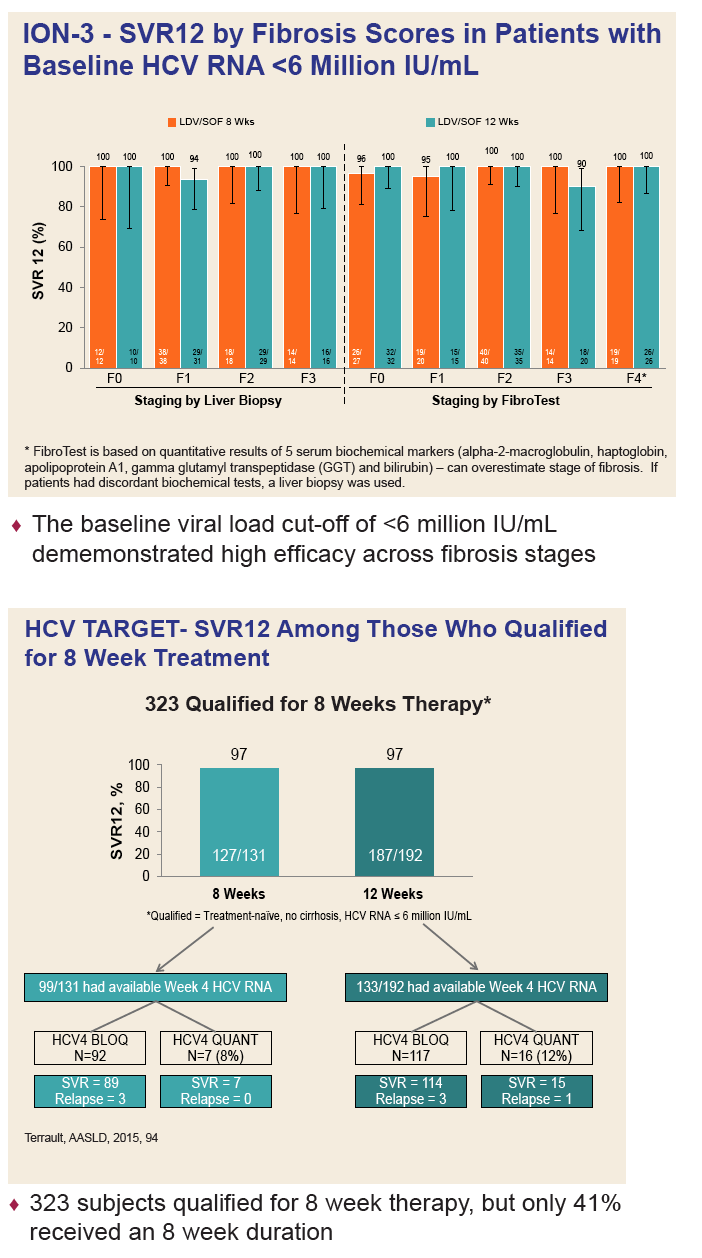
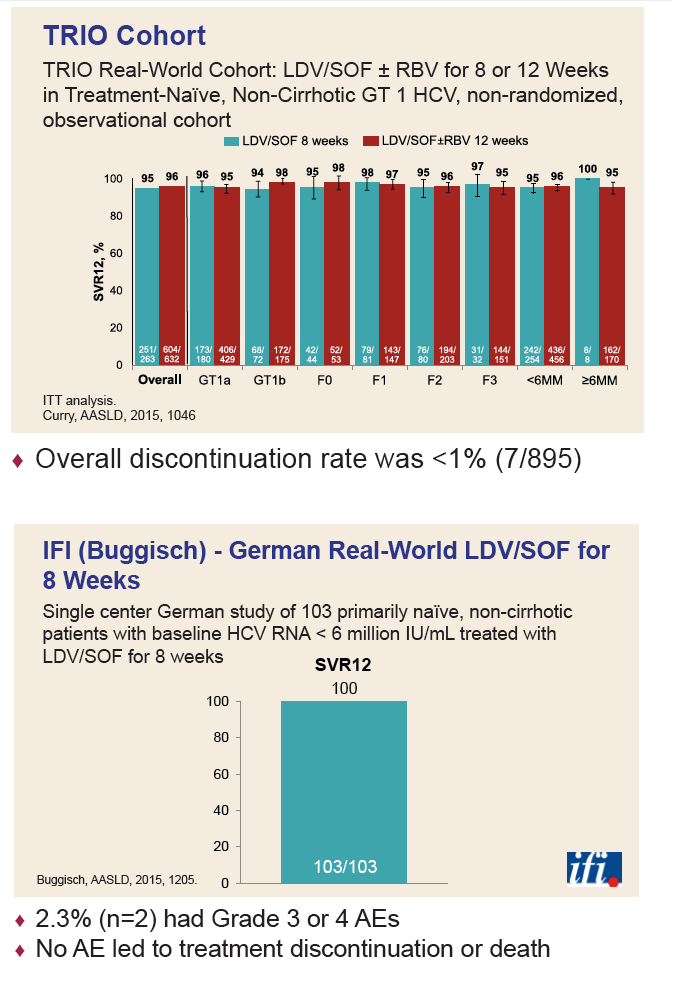
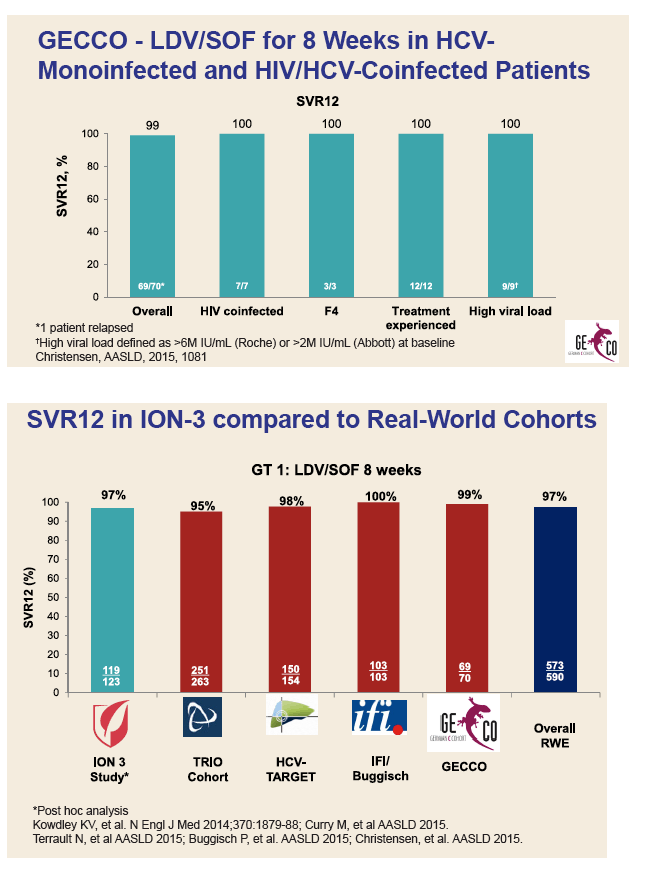
|
| |
|
 |
 |
|
|