 |
 |
 |
| |
Phase 2, Randomized, Open-Label Clinical Trials of the Efficacy and Safety of Grazoprevir and MK-3682 (NS5B Polymerase Inhibitor) with Either Elbasvir or MK-8408 (NS5A Inhibitor) in Patients with Chronic HCV GT1, 2 or 3 Infection (Part A of C-CREST 1 & 2)
|
| |
| |
Reported by Jules Levin
HEP DART 2015, December 6-10, 2015, Wailea, Hawaii
Edward J. Gane, Stephen Pianko, Stuart K. Roberts, Alex J. Thompson,
Stefan Zeuzem, Eli Zuckerman, Ziv Ben Ari, Graham R. Foster, Kosh Agarwal, Alex L. Laursen, Jan Gerstoft, Wei Gao, Hsueh-cheng Huang,
Brian Fitzgerald, Jerry J. Li, Shuyan Wan, Frank Dutko, Bach-Yen T. Nguyen, Janice Wahl, Eliav Barr, Joan R. Butterton, Rafael Esteban
AASLD: Phase 2, Randomized, Open-Label Clinical Trials of the Efficacy and Safety of Grazoprevir and MK-3682 (NS5B Polymerase Inhibitor) with Either Elbasvir or MK-8408 (NS5A Inhibitor) in Patients with Chronic HCV GT1, 2 or 3 1nfection (Part A of C-CREST-1 & 2) - (11/23/15)
AASLD: Merck Announces Presentation of Phase 3 Results of Investigational Elbasvir/Grazoprevir in People with Chronic Hepatitis C Genotypes 1, 4 or 6 Infection Who Inject Drugs and Are on Opioid Agonist Therapy - (11/18/15)
AASLD: Eliminate Restrictions to Substance Users/Injection Drug Users - SVR Rates the Same in New Study as in Non-IDUs 96% - Routine HCV Testing is needed - (11/18/15)
AASLD: C-EDGE CO-STAR: efficacy of grazoprevir / elbasvir Fixed Dose Combination for 12 Weeks in HCV-infected Persons Who Inject Drugs on Opioid Agonist Therapy - (11/16/15)
AASLD: AN INTEGRATED ANALYSIS OF 402 COMPENSATED CIRRHOTIC PATIENTS WITH HCV GENOTYPE (GT) 1, 4 or 6 INFECTION TREATED WITH Elbasvir/GRAZOPREVIR - (11/19/15)
AASLD: Efficacy of Elbasvir (EBR) and Grazoprevir (GZR) in Black HCV-Infected Patients: Results of a Pooled Analysis of Phase 2/3 Studies - (12/03/15)
AASLD: High Efficacy of Elbasvir and grazoprevir With or Without Ribavirin in 103 Treatment-Naive and Experienced Patients With HCV Genotype 4 Infection: A Pooled Analysis - (12/01/15)
AASLD: Prevalence and Impact of Baseline NSA Resistance Associated Variants (RAVs) on the Efficacy of Elbasvir/Grazoprevir (EBR/GZR) Against GT1a Infection - (11/19/15)
AASLD: Merck Announces Presentation of Results from Two Phase 2 Studies of Investigational Triple-Combination Chronic Hepatitis C Therapy at The Liver Meeting® - (11/19/15)




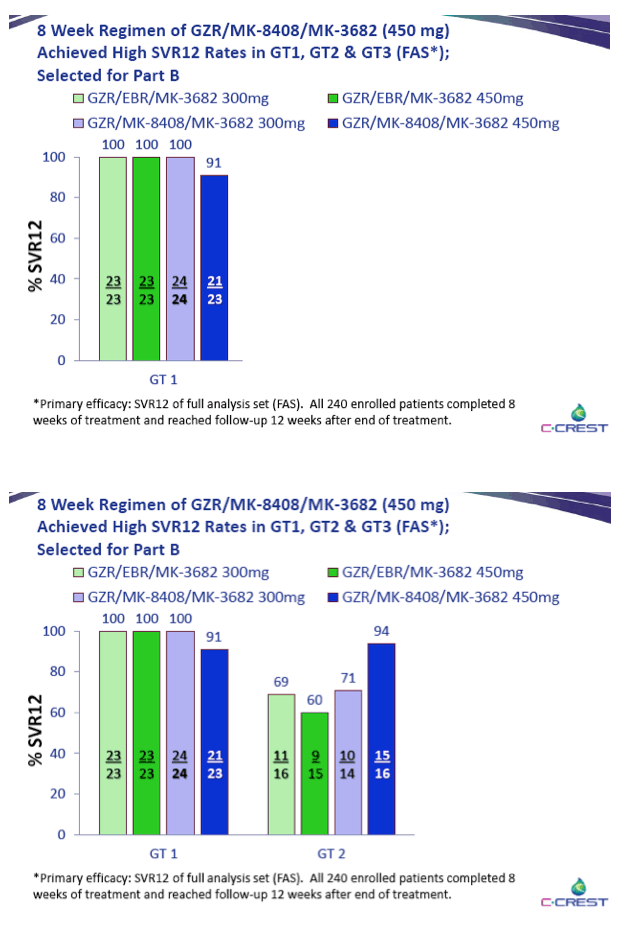
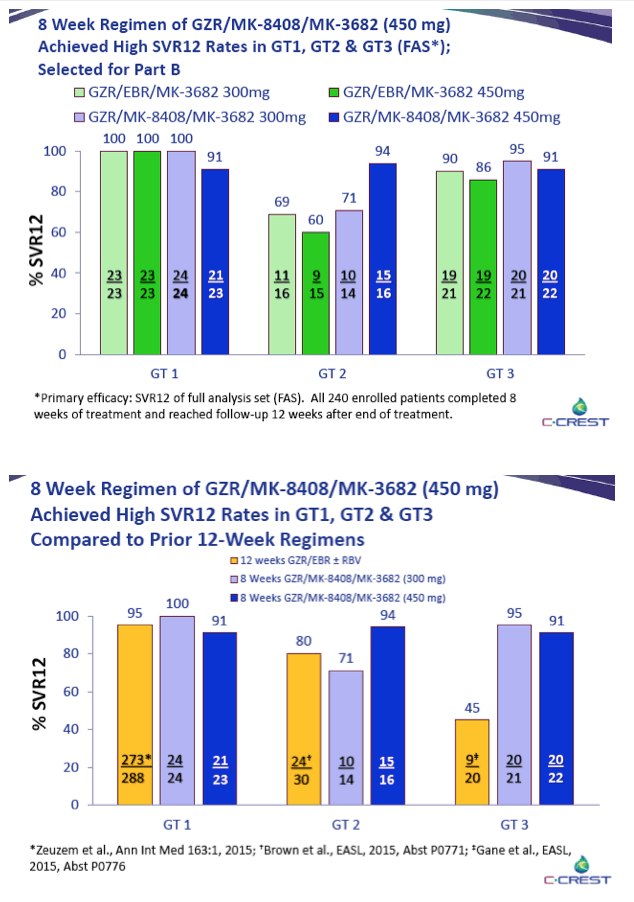
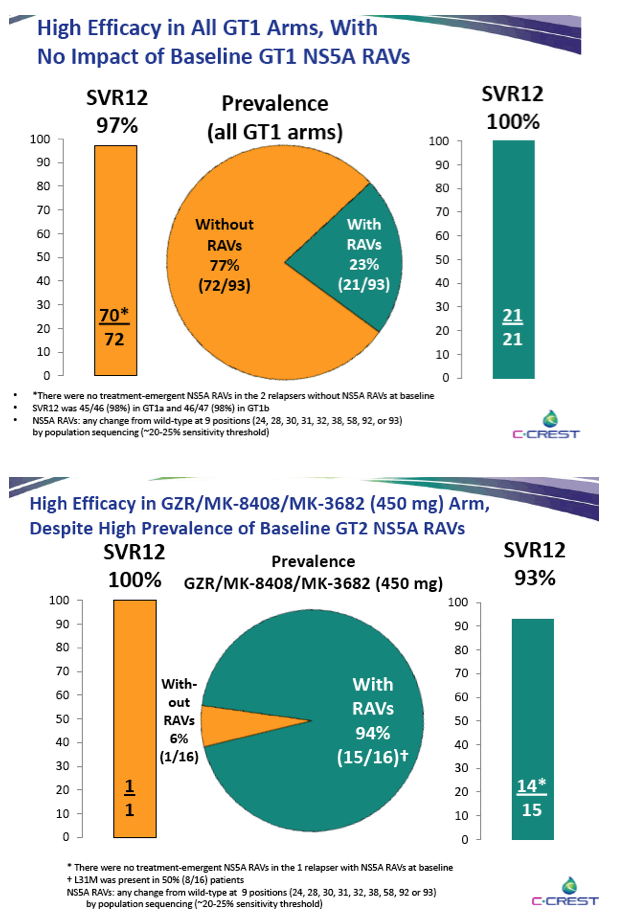
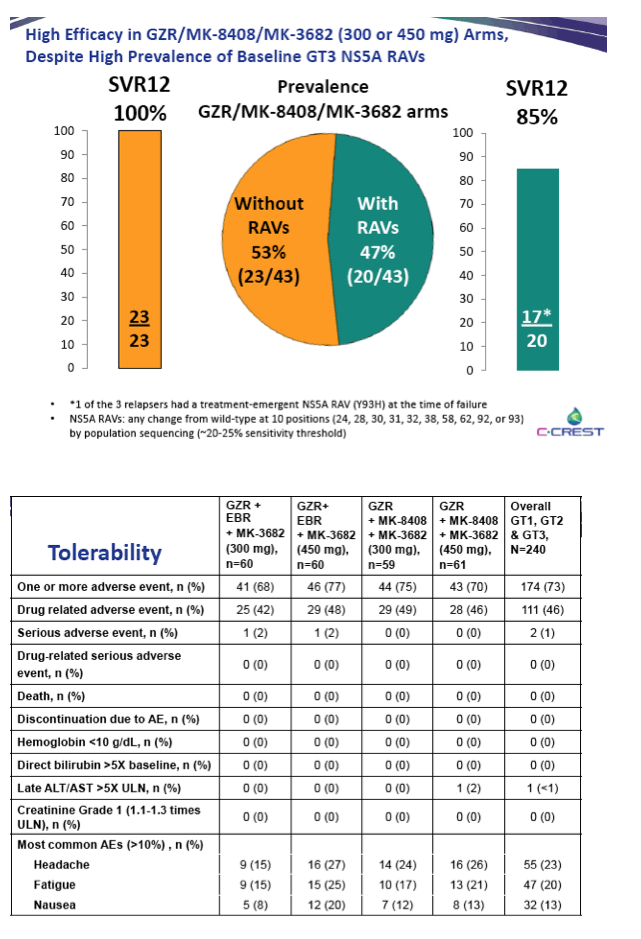
|
| |
|
 |
 |
|
|