 |
 |
 |
| |
Hepatitis B Reactivation Associated with Direct Acting Antiviral Therapy for Hepatitis C:
A review of Spontaneous Post-Marketing Cases
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
Susan J. Bersoff-Matcha MD; Kelly Cao PharmD; Mihaela Jason PharmD; Adebola Ajao PhD; S. Christopher Jones PharmD, MPH, MS*; Tamra Meyer PhD, MPH; Allen Brinker MD, MS
Center for Drug Evaluation and Research, Office of Surveillance and Epidemiology 10903 New Hampshire Avenue, Silver Spring, MD 20993 *Commissioned Officer of the US Public Health Service
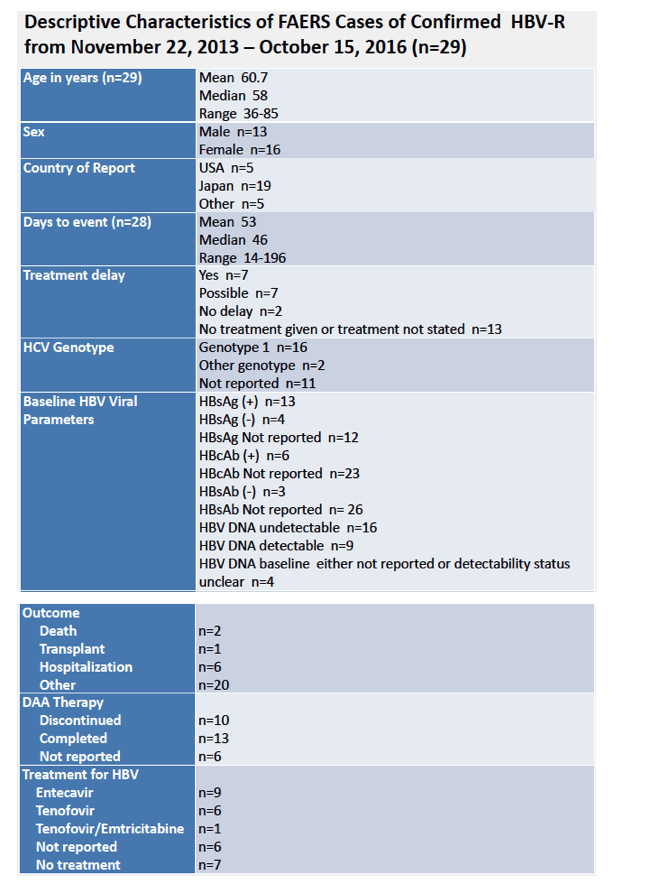
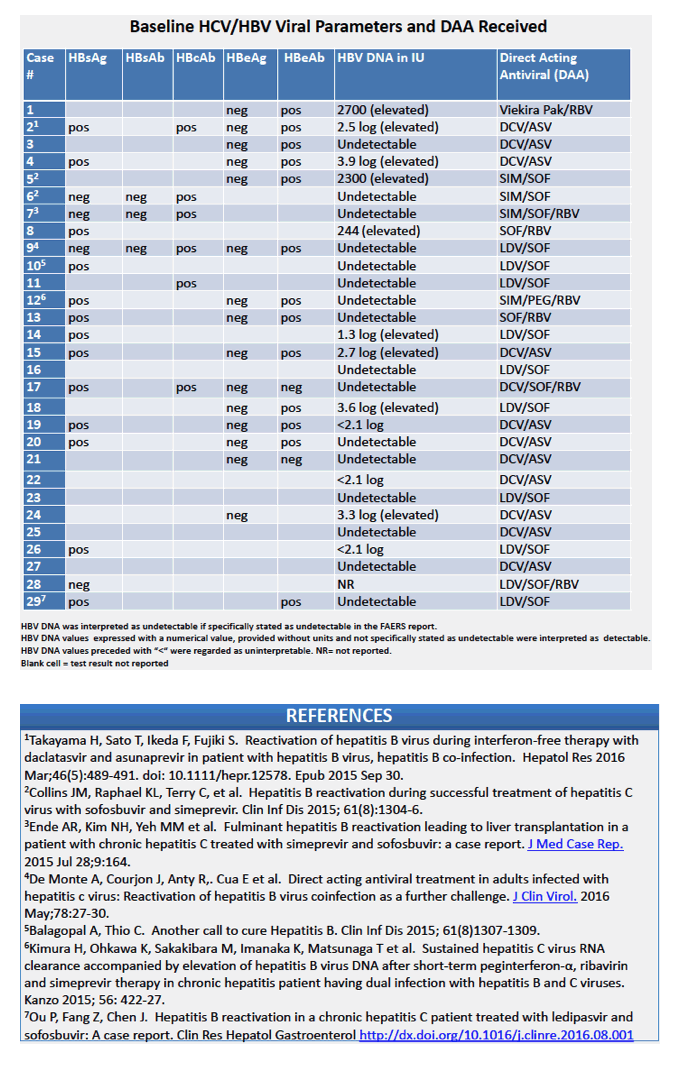
AASLD: Hepatitis B Reactivation in Patients Receiving Interferon-Free Direct Acting Antiviral Agents for Chronic Hepatitis C Virus Infection - (11/23/16)
AASLD: HEPATITIS B (HBV) REACTIVATION DURING ANTI-HEPATITIS C (HCV) THERAPY WITH INTERFERON (IFN)-FREE REGIMENS: A PROSPECTIVE STUDY - (11/22/16)

Center for Drug Evaluation and Research, Office of Surveillance and Epidemiology 10903 New Hampshire Avenue, Silver Spring, MD 20993 *Commissioned Officer of the US Public Health Service


|
| |
|
 |
 |
|
|