 |
 |
 |
| |
Pharmacokinetics (PK), safety and tolerability of the 2-and
3-direct acting antiviral (DAA)
combination of AL-335, odalasvir
(ODV) and simeprevir
(SMV) administered once-daily (QD) in healthy volunteers (HVs)
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
TN Kakuda1, MW McClure1, C Westland1, J Vuong1, M-C Homery2, G Poizat2, L Viguerie2, C Denot2, A Patat2, Q Zhang1, J Hui3, D Apelian3, D Smith1, S Chanda1, J Fry11Alios BioPharma, South San Francisco, CA, USA; 2Biotrial, Rennes, France; 3Achillion Pharmaceuticals Inc., New Haven, CT, USA
EASL/Paris-2016: Short duration treatment with AL-335 and odalasvir (ODV), with or without simeprevir (SMV), in treatment naïve patients with hepatitis C virus (HCV) genotype (GT) 1 infection - (09/26/16)
Achillion Announces 100% SVR12 in the 6-Week and 8-Week Cohorts in Janssen's Phase 2 Trial Evaluating the Triple Combination Treatment Regimen Including Odalasvir, AL-335, and Simeprevir for Genotype 1 Treatment-Naïve HCV - (09/26/16)

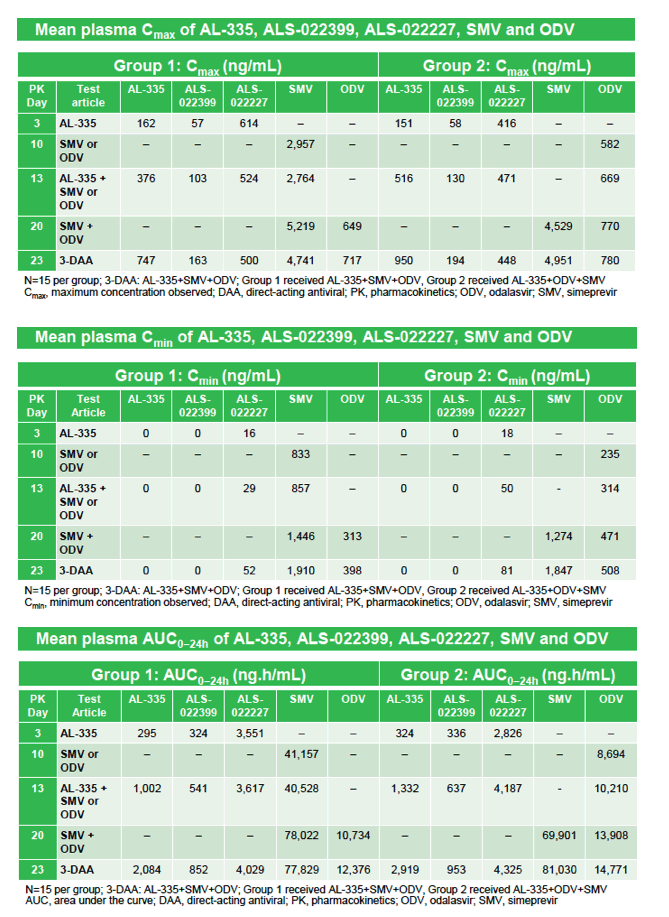
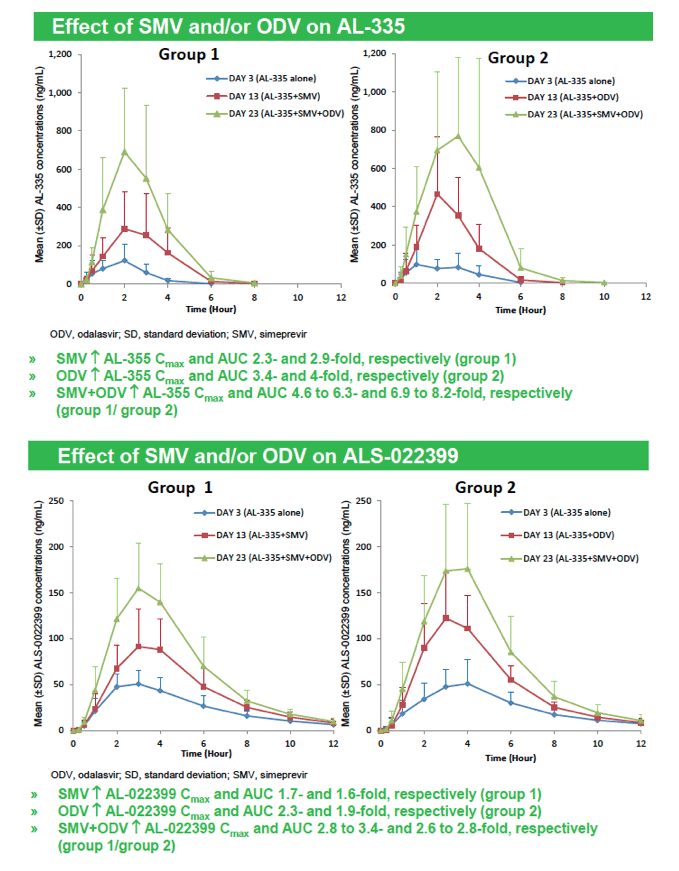
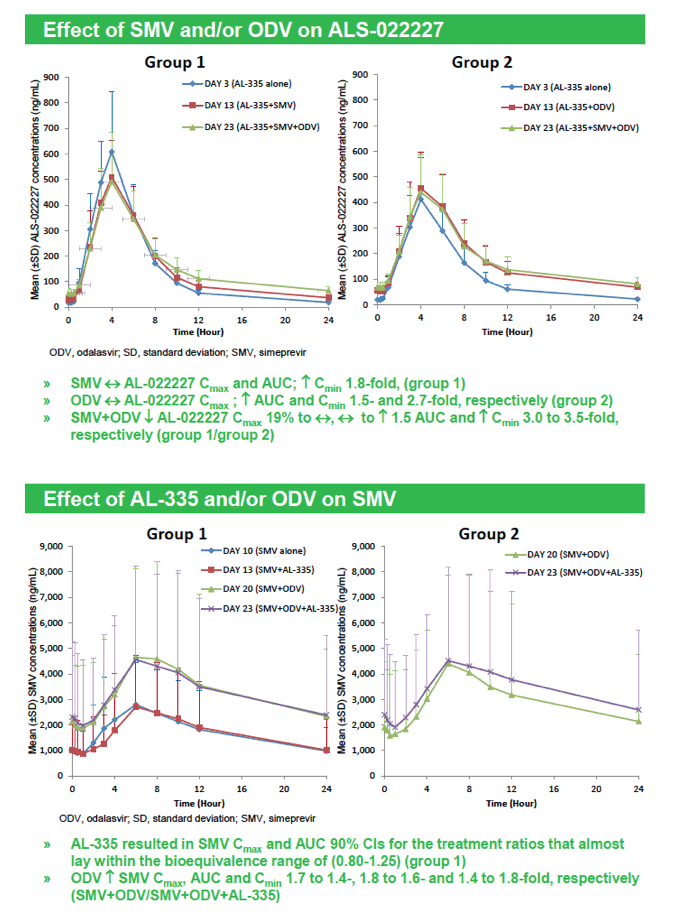
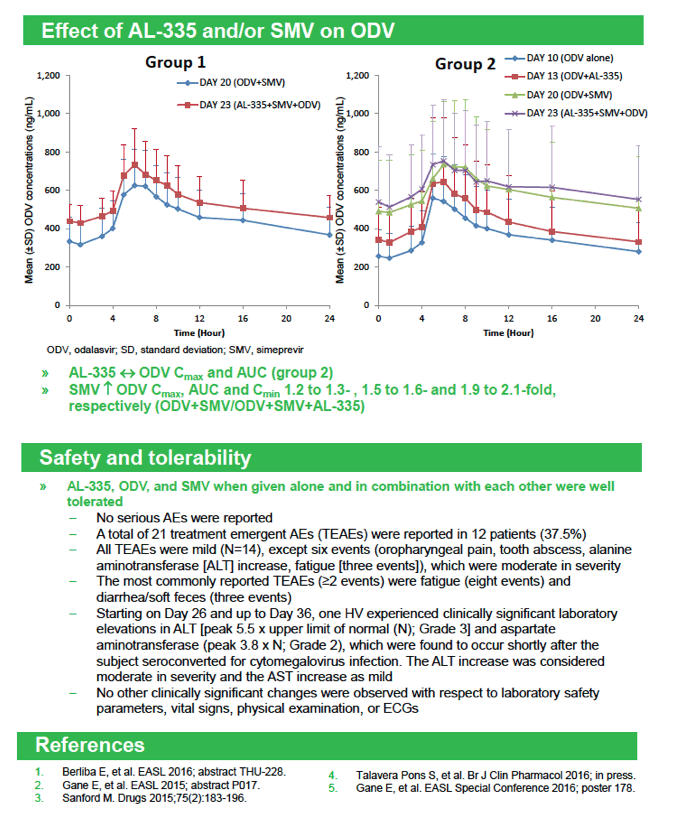
|
| |
|
 |
 |
|
|