 |
 |
 |
| |
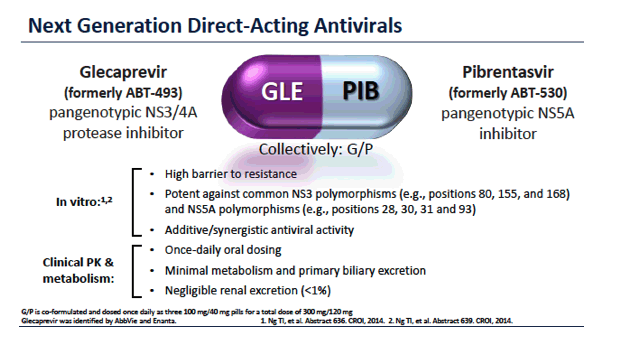
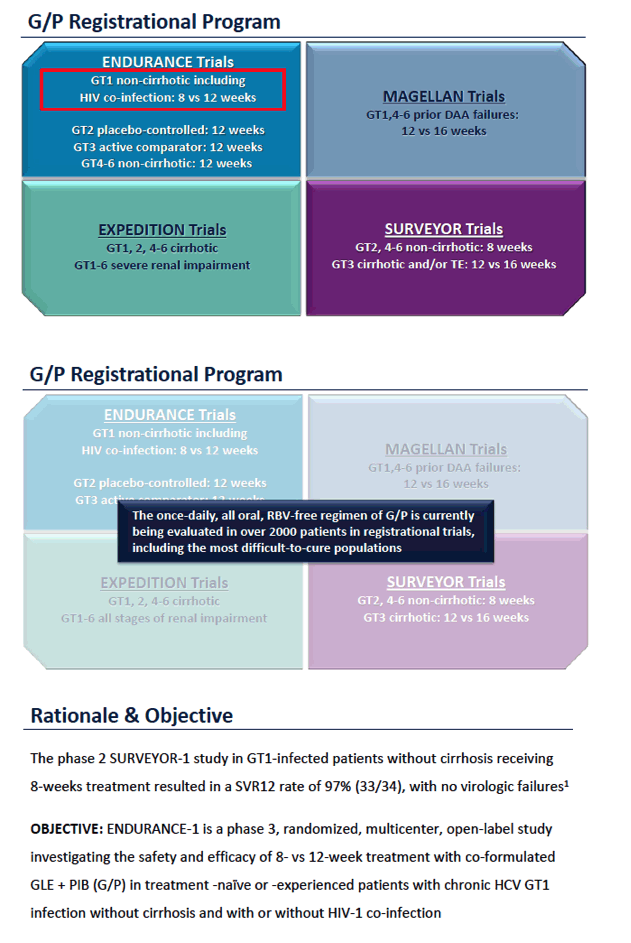
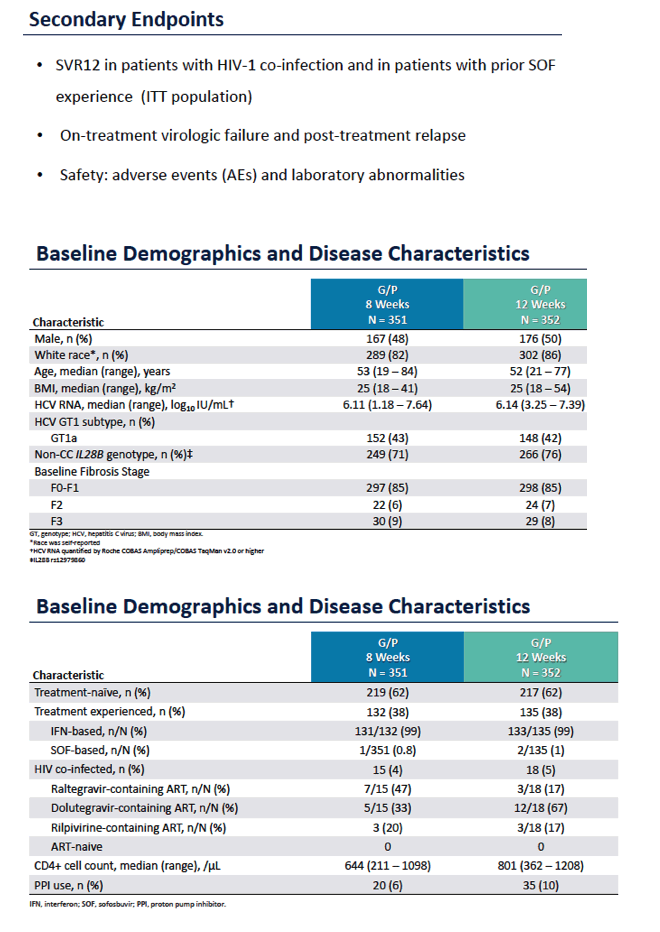
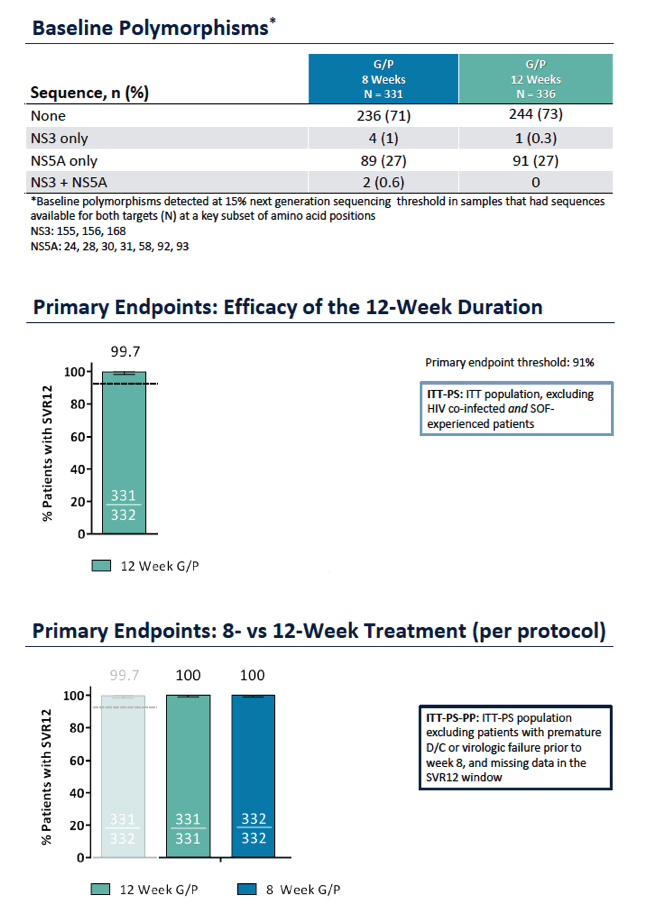
ENDURANCE-1: A Phase 3 Evaluation of the Efficacy and Safety of 8- versus 12-week Treatment with Glecaprevir/Pibrentasvir (formerly ABT-493/ABT-530) in HCV Genotype 1 Infected Patients with or without HIV-1 Co-infection and without Cirrhosis
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
Stefan Zeuzem1, Jordan Feld2, Stanley Wang3, Marc Bourliere4, Heiner Wedemeyer5, Ed Gane6, Robert Flisiak7, Wan-Long Chuang8, Steven Flamm9, Paul Kwo10, Gladys Sepulveda-Arzola11, Ruth Soto-Malave12, Massimo Puoti13, Edward Tam14, Rafi Bruck15, Francisco Fuster16, Seung Woon Paik17, Franco Felizarta18, Bo Fu3, Teresa I. Ng3, Chih-Wei Lin3, Federico Mensa3
1J.W. Goethe University, Frankfurt, Germany; 2Toronto Centre for Liver Disease, University of Toronto, Toronto, Ontario, Canada; 3AbbVie Inc., North Chicago, Illinois, United States; 4Hopital Saint Joseph, Marseille, France; 5Medizinische Hochschule Hannover, Hannover, Germany; 6Liver Unit, Auckland City Hospital, Auckland, New Zealand; 7Klinika Chorób Zakaznychi Hepatologii UM w Biaiymstoku,Biaiystok, Poland; 8Kaohsiung Medical University, Kaohsiung City, Taiwan; 9Northwestern Feinberg School of Medicine, Chicago, IL, USA; 10Indiana University, Indianapolis, IN, USA; 11Instituto de Investigación Científica del Sur, Ponce, Puerto Rico; 12University of Puerto Rico, San Juan, Puerto Rico; 13AO Ospedale Niguarda Ca Granda, Milan, Italy; 14LAIR Centre, Vancouver, BC, Canada; 15Tel Aviv Medical Center, Tel Aviv, Israel; 16Centro de Investigaciones Clinicas Vina del Mar; Vina del Mar, Chile; 17Samsung Medical Center, Seoul, Republic of Korea; 18Private practice, Bakersfield, California, United States





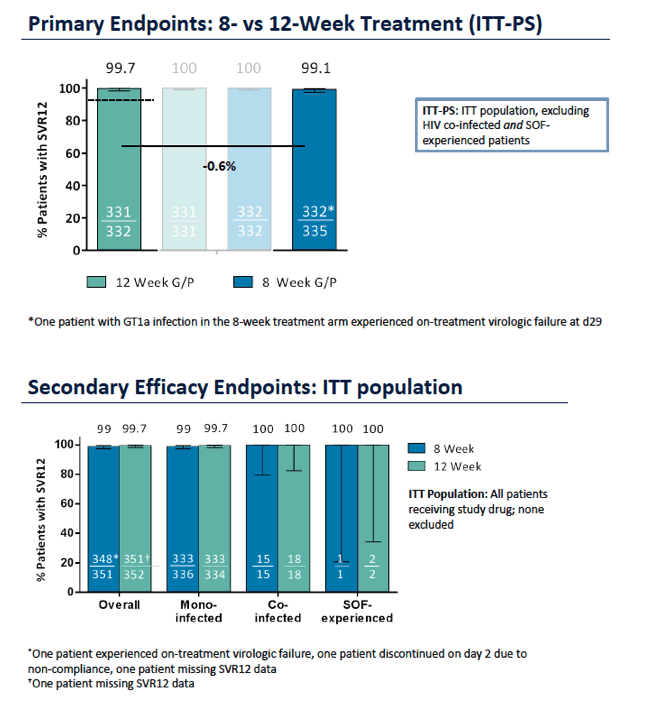
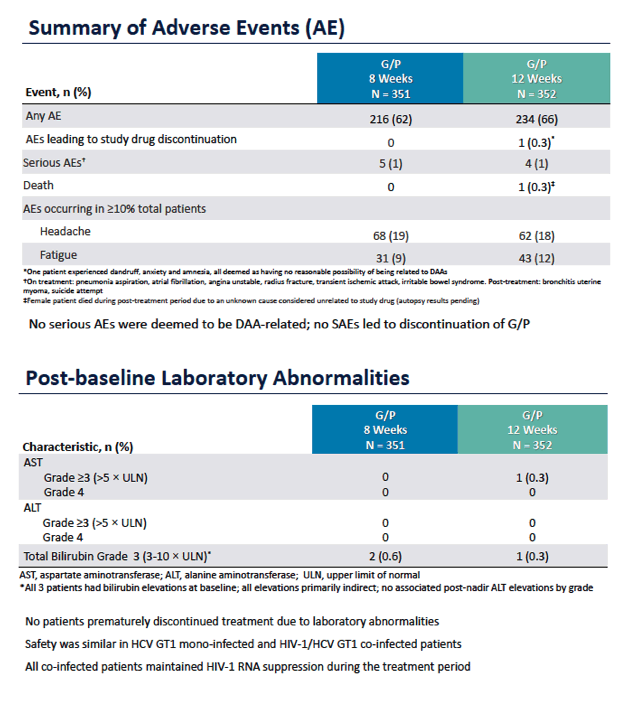
|
| |
|
 |
 |
|
|