 |
 |
 |
| |
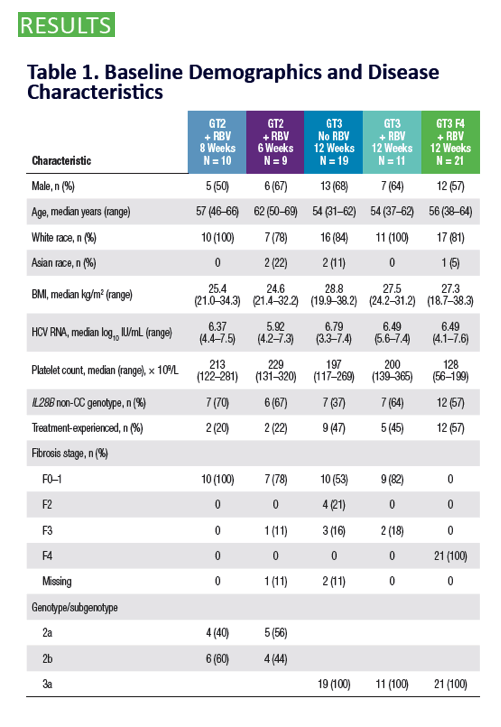
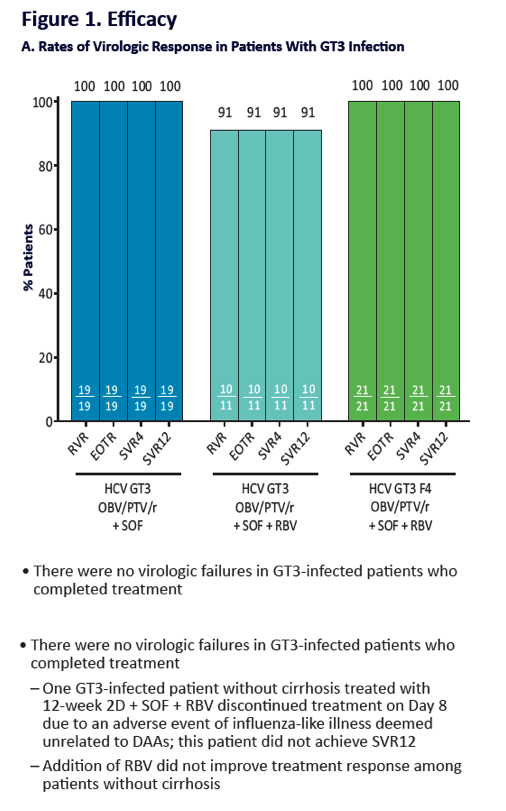
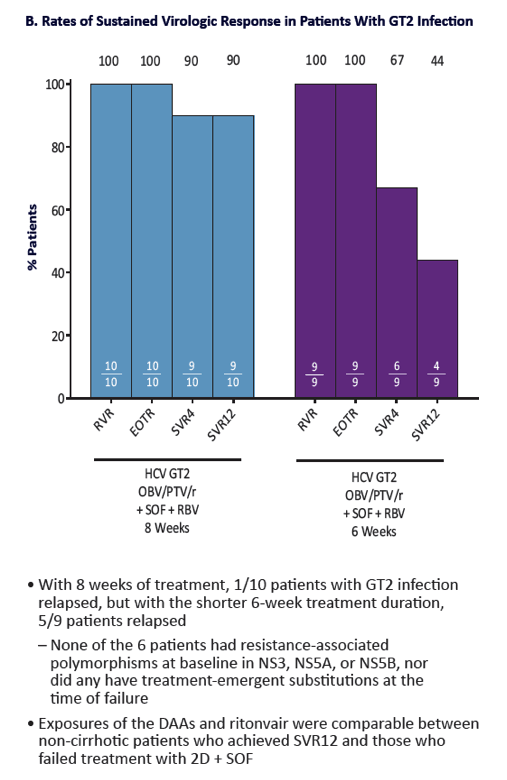
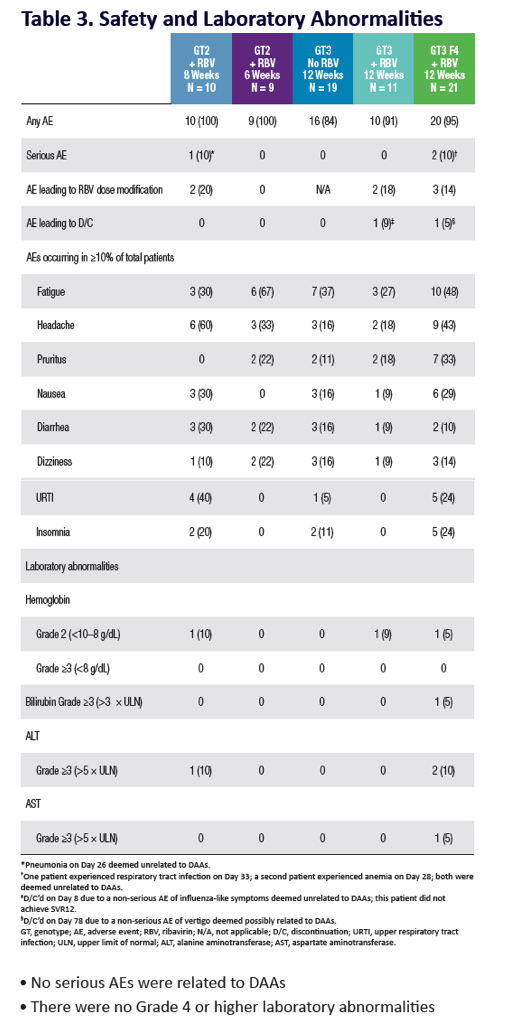
QUARTZ II-III: Final Efficacy and Safety Results in Patients With HCV Genotype 2 or 3 Infection - Abbvie 2D + Sofosbuvir in Gt3 [12 weeks] & Gt2 (6, 8 weeks) - treatment experienced & naive
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
Stephen D Shafran1, David Shaw2, Mariem Charafeddine3, Kosh Agarwal4, Graham R Foster5, Manal Abunimeh3, Bo Fu3, Tami Pilot-Mati as3, Rajvineeth Kumar Pothacamury3, Jennifer R King3, Eric Cohen3, Daniel E Cohen3, Edward Gane6
1University of Alberta Hospital, Edmonton, Alberta, Canada; 2Royal Adelaide Hospital, Adelaide, Australia; 3AbbVie Inc., North Chicago, Illinois, United States; 4Insti tute of Liver Studies, Kings College Hospital, London, United Kingdom; 5Queen Mary University of London, Barts Health, London, United Kingdom,
6Liver Unit, Auckland City Hospital, Auckland, New Zealand




|
| |
|
 |
 |
|
|