 |
 |
 |
| |
Characterization of Host, Viral, and Treatment-Related Factors Associated With Antiviral Efficacy of Tenofovir Alafenamide and Tenofovir Disoproxil Fumarate
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
Edward J. Gane, 1 Masaki Saito, 2 Si-Hyun Bae, 3 Pietro Andreone, 4 John F. Flaherty, 5 Kyungpil Kim, 5 Anuj Gaggar, 5 Robert P. Myers, 5 Florin A. Caruntu, 6 Florence Wong, 7 Xiaoli Ma, 8 Patrick Marcellin 9
1New Zealand Liver Transplant Unit, Auckland Clinical Studies, Auckland, New Zealand; 2Hyogo College of Medicine, Hyogo, Japan; 3Seoul St. Mary's Hospital, Seoul, Korea; 4Center for Research and Studies on Hepatitis, University of Bologna, Italy; 5Gilead Sciences, Inc., Foster City, CA;
6Institutul National de Boli Infectioase "Prof. Dr. Matei Bal," Bucharest, Romania; 7University Health Network, Toronto, Ontario, Canada; 8Xiaoli Ma, PC, Bryn Mawr, PA; 9Hôpital Beaujon, Clichy, France
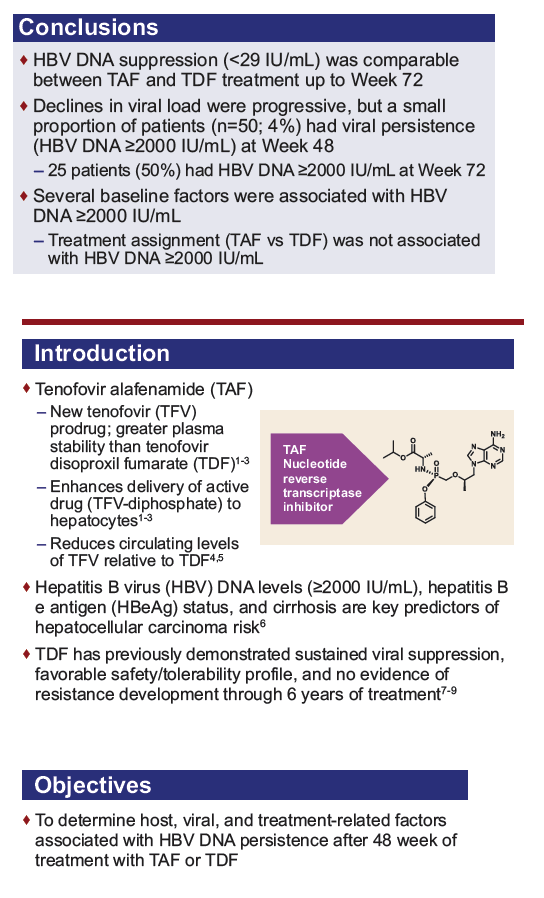
*Normal alanine aminotransferase (ALT) ranges according to AASLD criteria10 are <19 and <30 U/L for females and males, respectively, and
according to central laboratory criteria are ≤34 and ≤43 U/L, respectively, aged <69 y, and ≤32 and ≤35 U/L, respectively, aged >69 y. eGFRCG,
estimated glomerular filtration rate by Cockcroft-Gault (CG) formula.
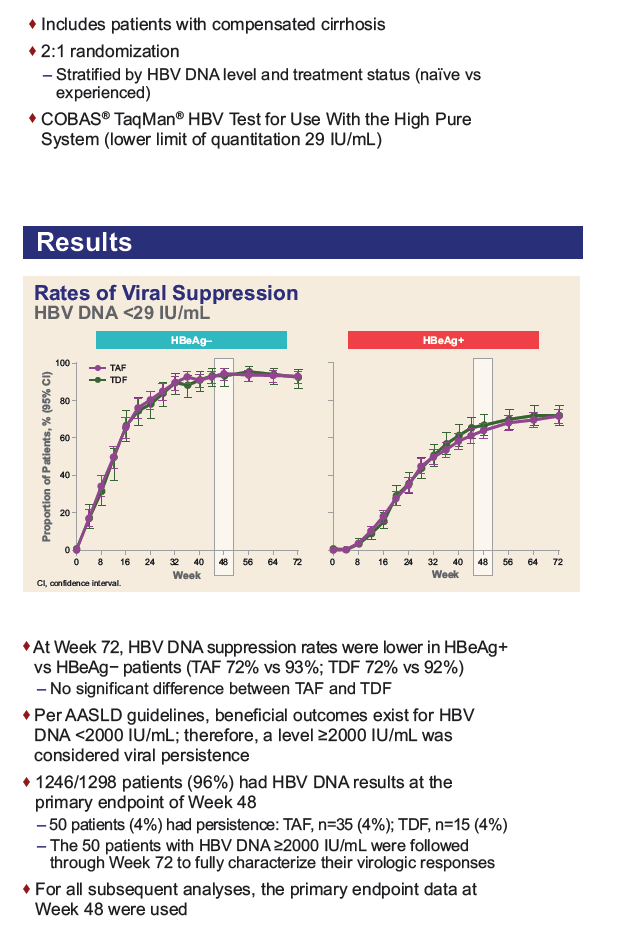
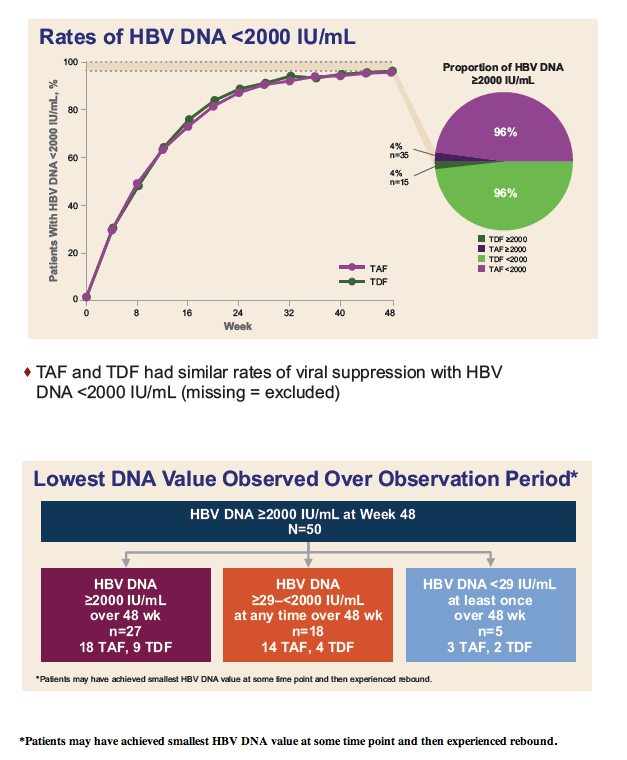
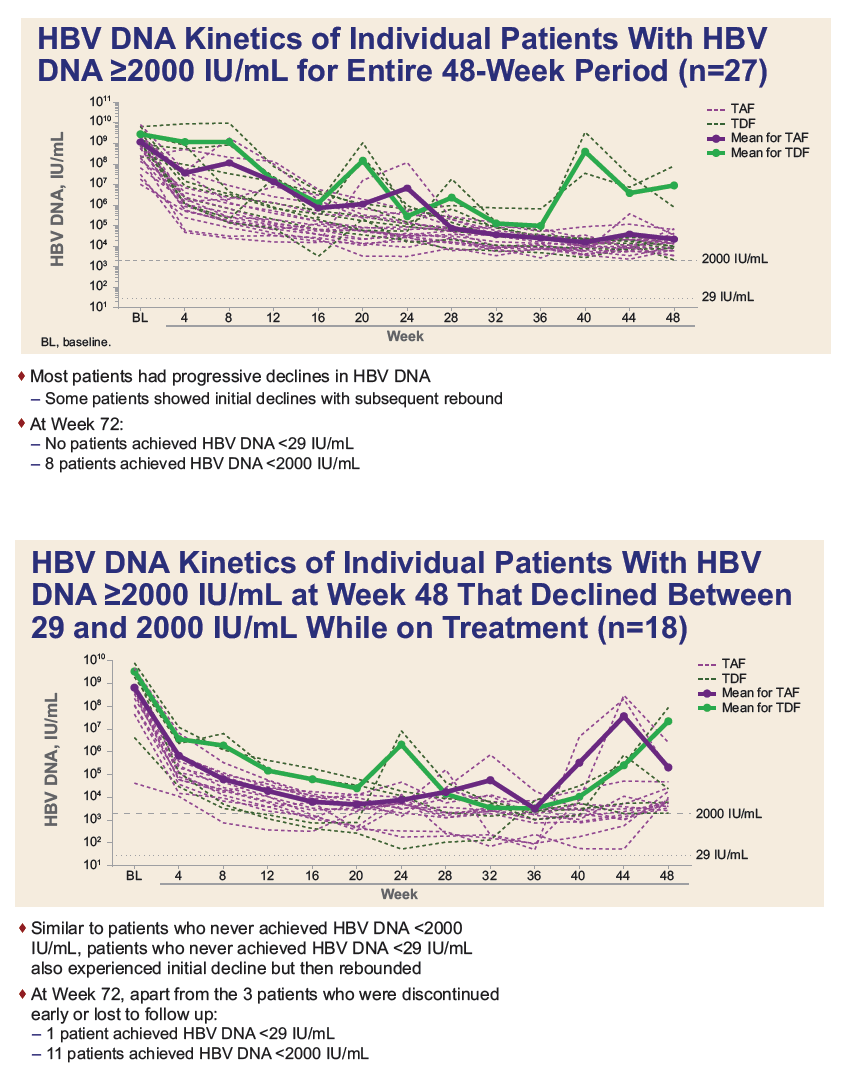
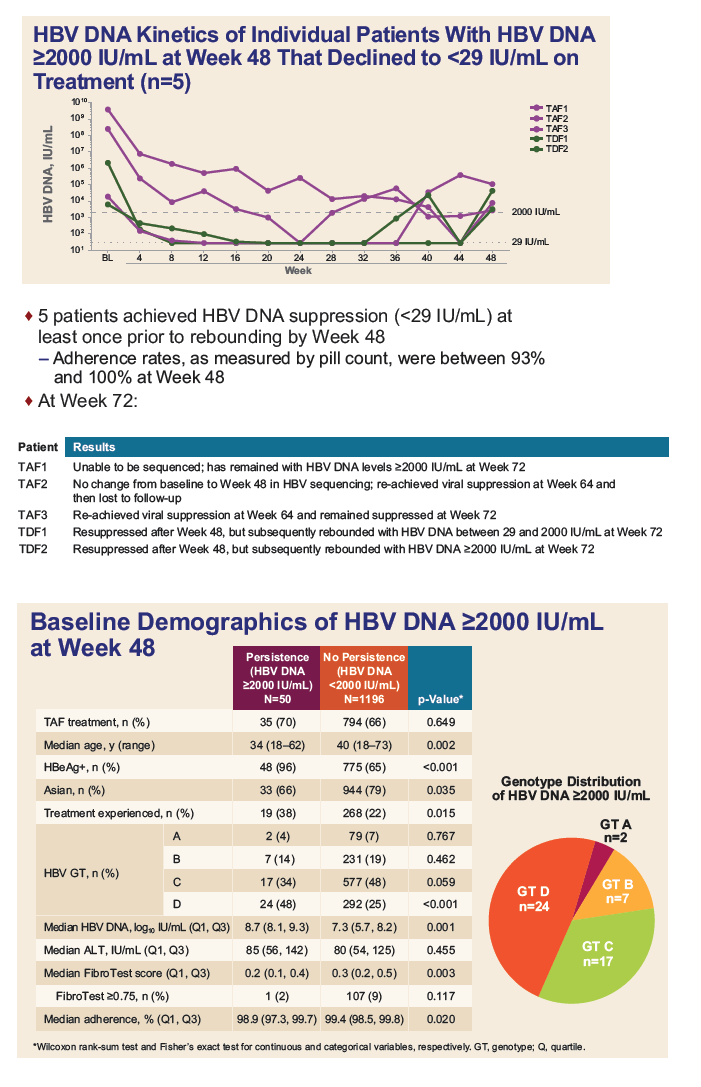
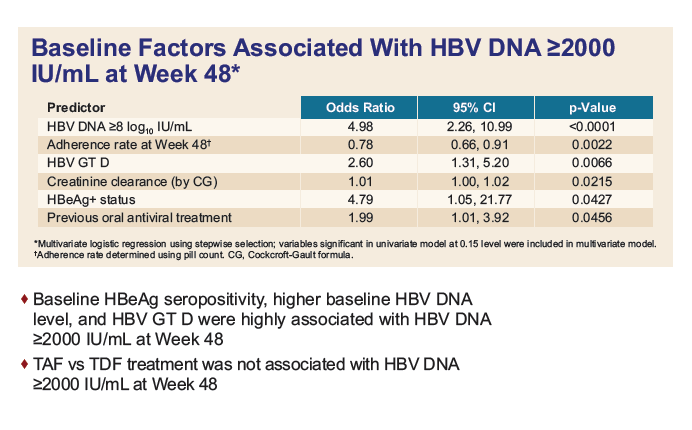
|
| |
|
 |
 |
|
|