 |
 |
 |
| |
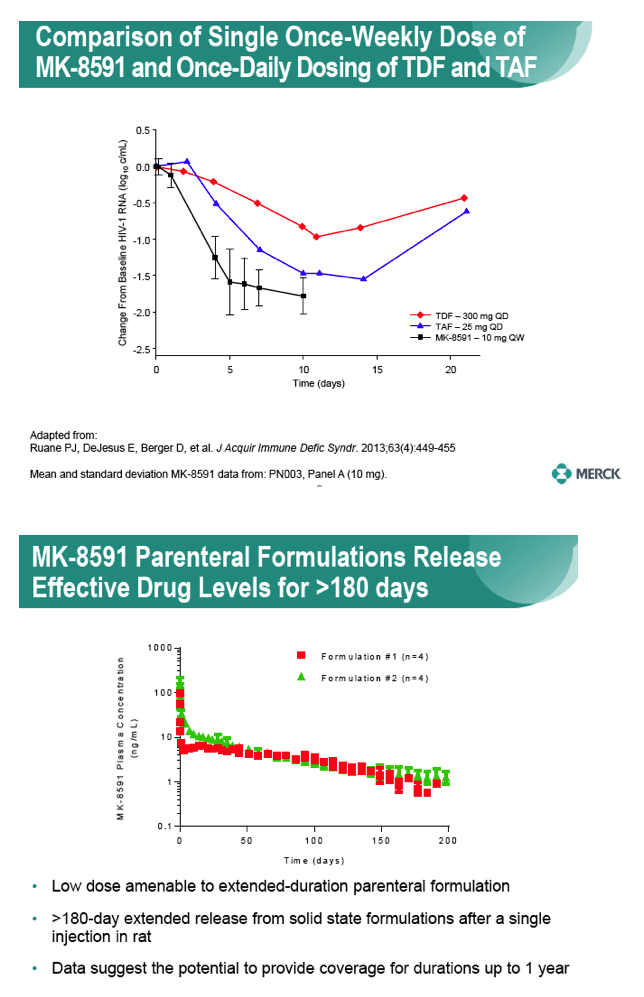
Long-Acting Oral and Parenteral Dosing of MK-8591 for HIV Treatment or Prophylaxis
|
| |
| |
Reported by jules Levin
CROI 2016 Feb 22-24 Boston
Jay A. Grobler, Ming-Tain Lai, Stephanie E. Barrett, Marian Gindy, Kerry Fillgrove, Wendy Ankrom, Sandra Wood, Evan Friedman, Marian Iwamoto, Daria J. Hazuda on behalf of the MK-8591 Early Development Team (West Point, PA)
Merck & Co., Inc. Kenilworth, NJ, USA
WEBCAST link: http://www.croiwebcasts.org/console/player/29624?mediaType=audio&


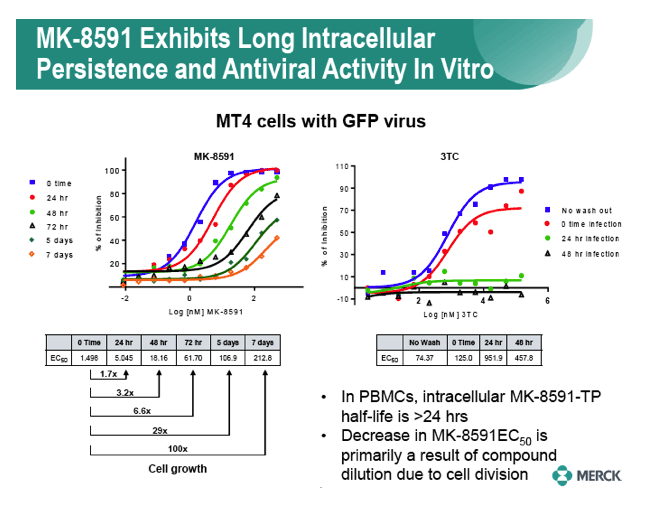
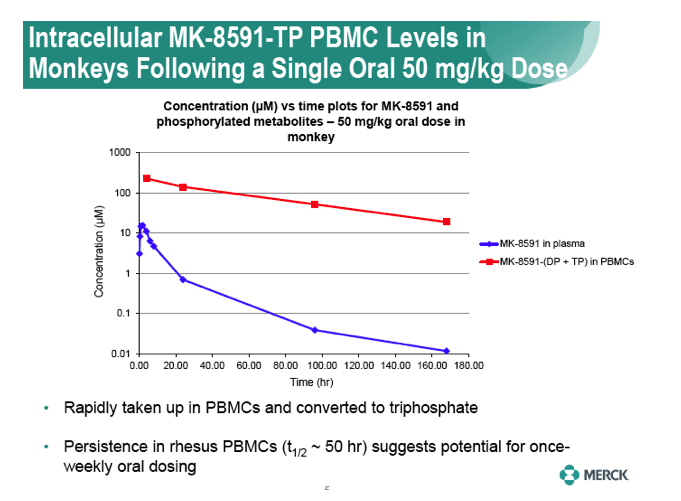
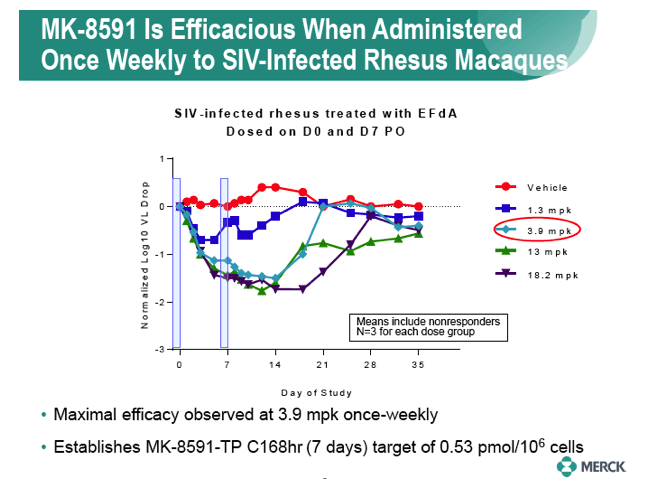
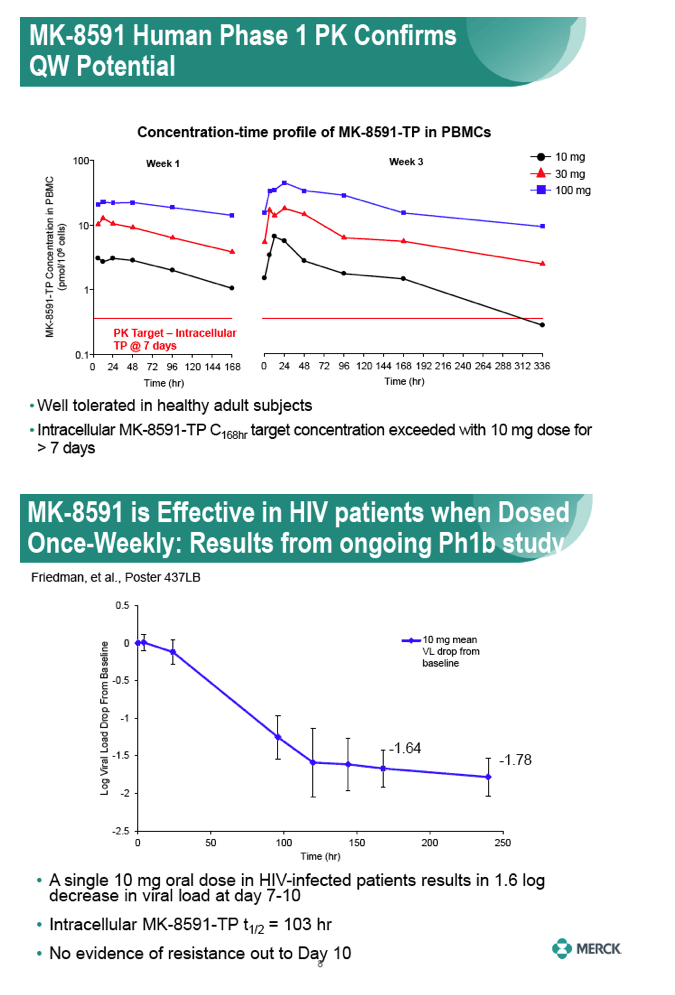
|
| |
|
 |
 |
|
|