 |
 |
 |
| |
SOF/Rbv SWIFT-C Study Acute HCV Treatment: low RBV levels & relapse;
Acute HCV - studies of 4, 6, 8 & 12 weeks
|
| |
| |
Download the PDF here
Jules Levin, NATAP
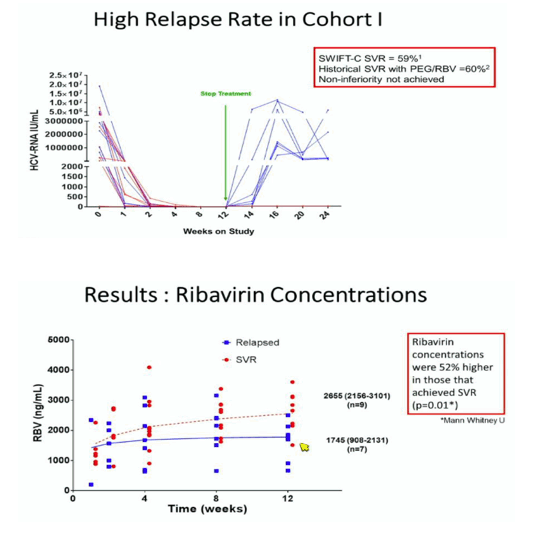
At CROI 2016 investigators reported that lower Ribavirin levels were present in the relapsers vs the SVR patients who were treated in the ACTG 5327 SWIFT-C Study reported at AASLD 2015:http://www.natap.org/2015/AASLD/AASLD_108.htm; in this ACTG study where 17 HIV/HCV coinfected patients with acute HCV in Cohort 1 received SOF + weight-based ribavirin for 12 weeks there was a high relapse rate of 41%, after 12 weeks treatment 100% were undetectable and SVR12 was only 59%, SUGGESTING non-adherence to RBV as the cause for relapse & suggesting that short-term HCV treatment as reported in SLAM-C where 4 weeks SOF/LDV achieved 100% SVR might be effective. Self-reported adherence, by patients themselves, found no statistical significant difference in self-reported 100% adherence: 9/10SVR patients reported 100% adherence vs 5/7 (71%), although there is no statistical
ly significant difference the numbers of patients is small; still this raises the question of whether patient/self-reported adherence is reliable, which in some studies it has been found not to be reliable. SWIFT-C is a 2 cohort study & Cohort 2 is ongoing. Here at CROI the study investigators report the relapsers had 52% lower ribavirin levels. The SLAM-C study was conducted in the setting of NYC inner city drug rehabs, [link below] treated acute HCV for 4 weeks with SOF/LDV with a 100% SVR rate, or for 8 weeks with SOF/Simeprevir where 14/15 achieved SVR12, the 1 failure was a dropout who restarted IV drug use.
And then at AASLD, Daniel Fierer reported acute HCV was treated with SOF/RBV for 12 weeks with 11/12 achieving SVR12 with 1 unexplained failure.
SOFOSBUVIR IN THE TREATMENT OF ACUTE HCV INFECTION IN HIV-INFECTED MEN - (12/04/15)
CROI:Ledipasvir/Sofosbuvir for 6 Weeks in HIV-Infected Patients with Acute HCV Infection - (03/01/16)

High Relapse Rate - RBV low-levels/non-adherence?
CROI 2016: Lower Ribavirin Exposures in HIV+ Patients That Relapsed to Acute HCV Treatment (ACTG SWIFT-C
Study)......http://www.croiwebcasts.org/console/player/29625?mediaType=audio&
APASL: SLAM C study Sofosbuvir and Ledipasvir versus Sofosbuvir and Simeprevir combination therapy in the management of acute hepatitis C: A randomized open label prospective clinical pilot study. (drug rehabs in NYC] - (02/22/16)
CROI 2016 Program Abstract:
Lower Ribavirin Exposures in HIV+ Patients That Relapsed to Acute HCV Treatment
Christine E. MacBrayne1; Michael Hughes2; Kimberly M. Hollabaugh2; Jhoanna C. Roa3; Xinhui Chen4; Diana M. Brainard5; Raymond T. Chung6; Susanna Naggie7; Jennifer J. Kiser1; for the A5327 Study Team
1Univ of Colorado, Denver, CO, USA;2Harvard Sch of PH, Boston, MA, USA;3Social & Scientific Systems, Silver Spring, MD, USA;4Univ of Colorado, Aurora, CO, USA;5Gilead Sciences, Foster City, CA, USA;6Harvard Med Sch, Boston, MA, USA;7Duke Univ Sch of Med, Durham, NC, USA
Abstract Body:
ACTG 5327 (SWIFT-C) Cohort I determined the efficacy of sofosbuvir (SOF) and weight-based ribavirin (RBV; 1000 or 1200mg daily) in 17 HIV-1 infected individuals with acute hepatitis C virus (HCV). The rate of relapse following 12 weeks of treatment was 41% in this Cohort. The objective of this analysis was to determine the contribution of RBV pharmacokinetics to viral relapse.
RBV plasma concentrations were determined using a validated LC/MS-MS method. RBV concentrations at weeks 4, 8, and 12 of treatment, RBV dose, IL28B genotype, ITPA phenotype, race, and antiretroviral (ARV) regimen were compared in those who achieved sustained virologic response (SVR) vs. relapse using the Mann-Whitney U test.
Self-reported adherence was measured using a written four-day recall at each visit.
Seventeen HIV-infected males (11 Hispanic, 6 Caucasian, mean + SD age 42.8+10.6yrs, weight 75.9+10.3kg, and CrCl 120.2+27.3 mL/min, 11/2/1/3 HCV genotype 1a/1b/2b/indeterminate, 4/10/3 IL28B CC/CT/TT, 12/3/2 with 100%/60%/30% ITPA activity) received 12 weeks of SOF/RBV for acute HCV. No patient required a RBV dose reduction. Self-reported adherence to SOF/RBV was >95%. RBV concentrations are shown in the table. Median RBV exposures were 34% lower (p=0.01) at week 12 in those that relapsed vs. those that achieved SVR. The other variables tested were not significantly associated with relapse.
RBV exposures were lower at end of treatment in HIV-infected patients that relapsed to SOF/RBV therapy for acute HCV compared to those that achieved SVR. The cause of this discrepancy in exposures is unclear, but could relate to pharmacokinetic variability, a chance imbalance between groups, or differences in adherence despite the high levels of self-reported adherence.
|
| |
|
 |
 |
|
|