 |
 |
 |
| |
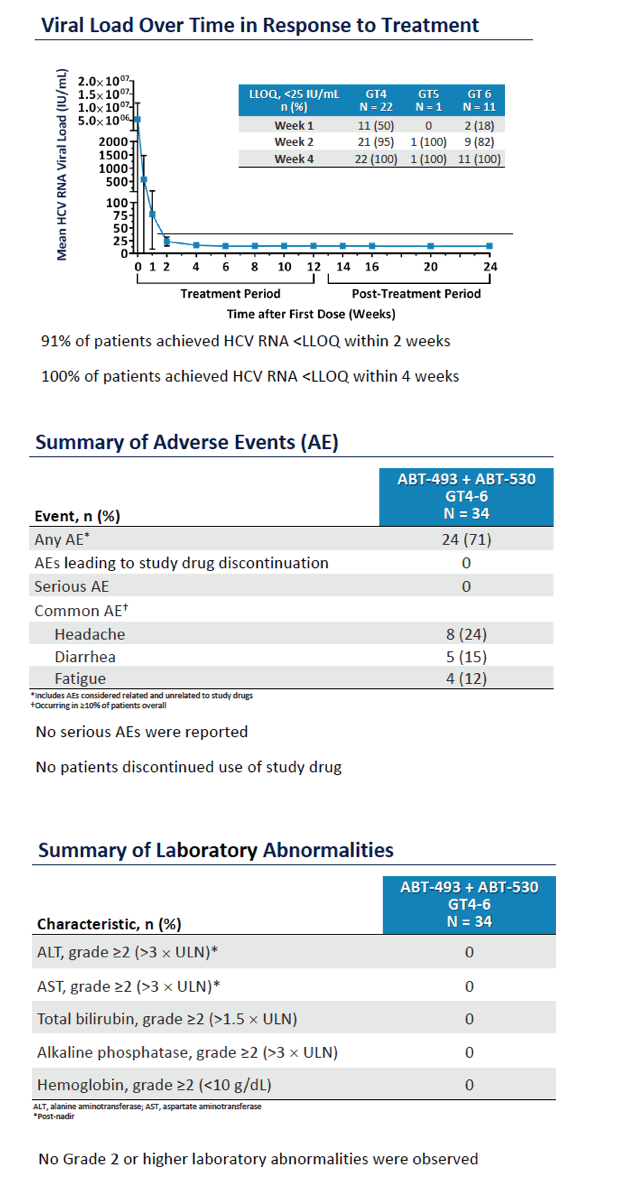
SURVEYOR-I: 100% SVR12 AND FAVORABLE SAFETY OF ABT-493 + ABT-530 ADMINISTERED FOR 12 WEEKS IN NON-CIRRHOTIC PATIENTS WITH GENOTYPES 4, 5, OR 6 INFECTION
|
| |
| |
Reported by Jules Levin
Digestive Disease Week
San Diego, California
23 May 2016
Edward Gane1, Jacob Lalezari2, Armen Asatryan3, Susan Greenbloom4, Tarek Hassanein5, Teresa I Ng3, Ran Liu3, Chih-Wei Lin3, Jens Kort3, Federico J Mensa3
1Liver Unit, Auckland City Hospital, Auckland, New Zealand; 2eStudySite, San Diego, California, United States;
3AbbVie Inc., North Chicago, Illinois, United States; 4Toronto Digestive Disease Associates, Toronto, Ontario, Canada; 5Southern California GI and Liver Centers and Southern California Research Center, Coronado, California, United States


|
| |
|
 |
 |
|
|