 |
 |
 |
| |
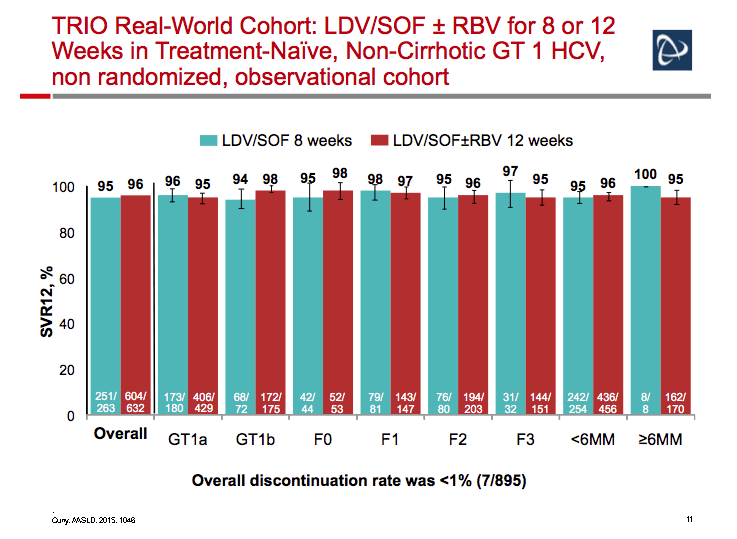
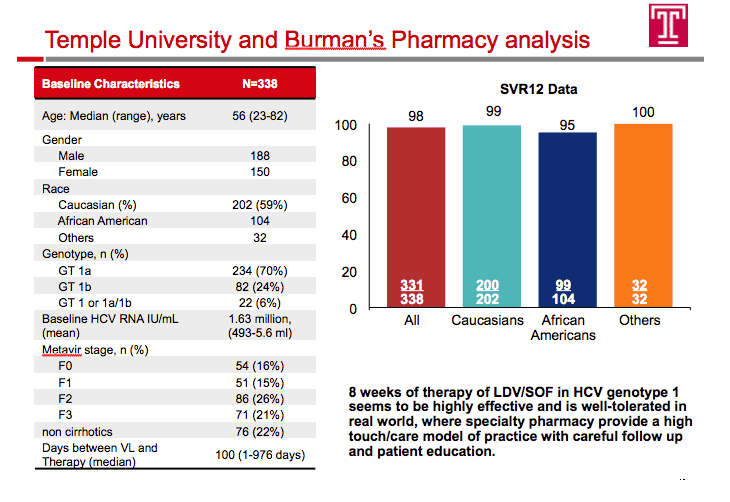
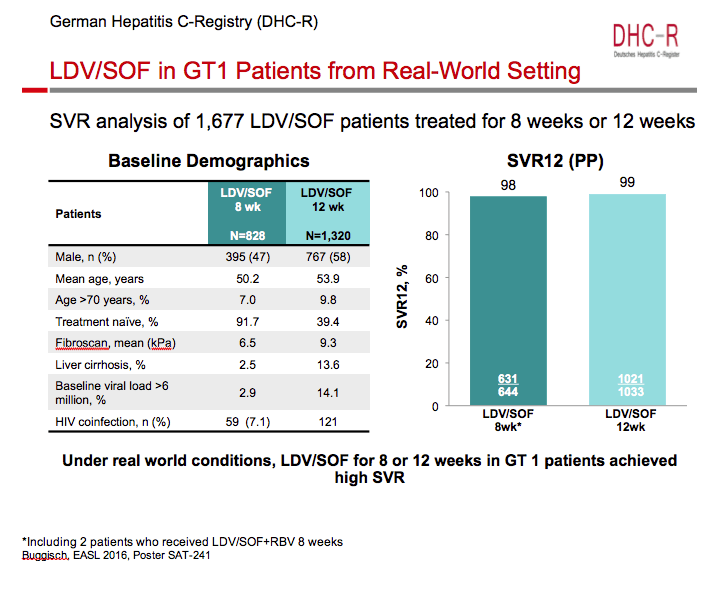
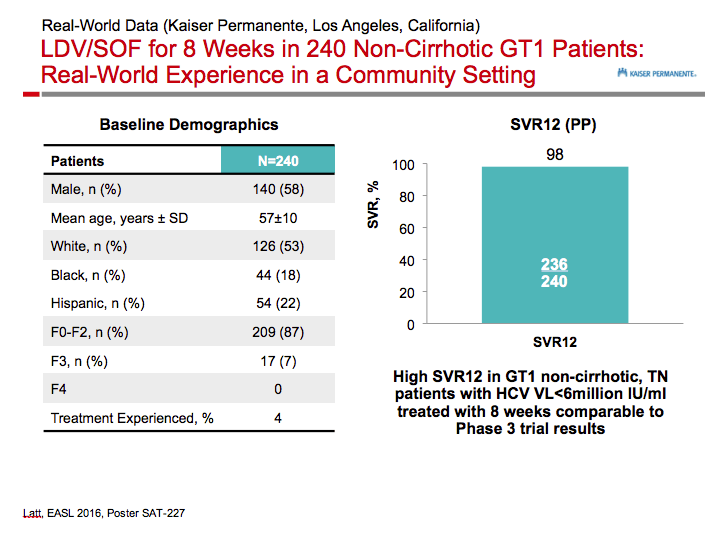
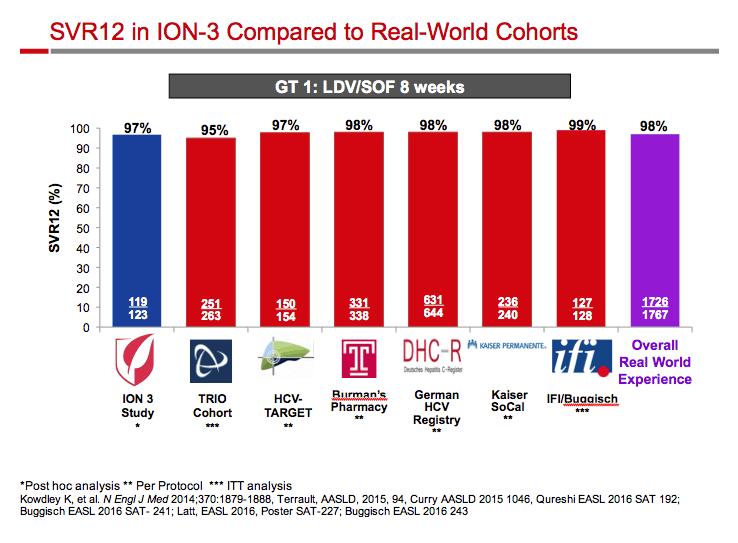
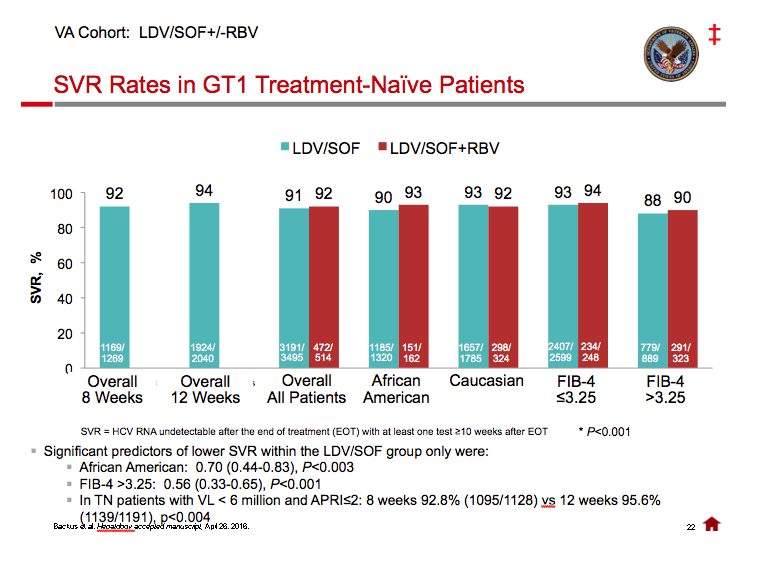
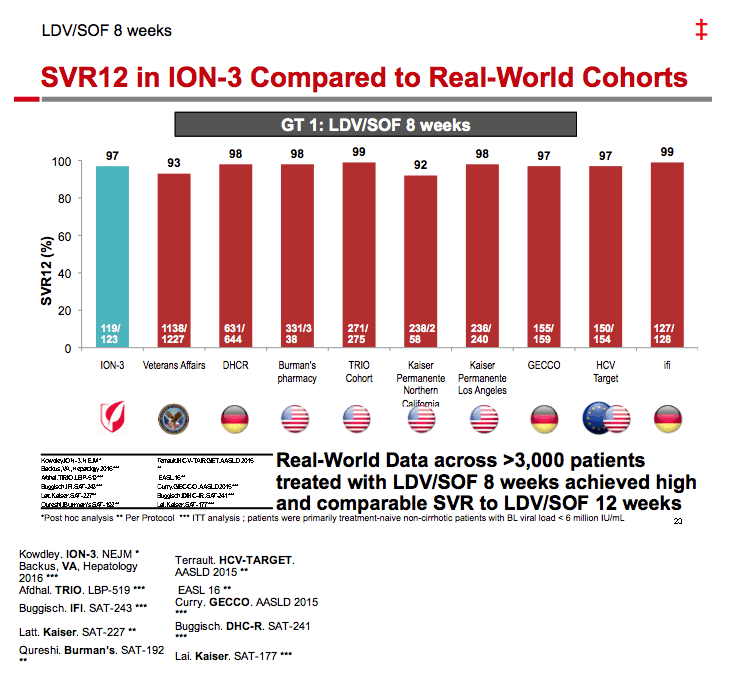
Ledipasvir/Sofosbuvir (LDV/SOF) for 8 Weeks in Genotype 1 Treatment-Naïve Non-Cirrhotic Patients with HCV Viral load <6 million IU/ml; A Comparative Analysis of the Phase-3 ION-3 Data to Real World Effectiveness
|
| |
| |
Reported by Jules Levin
DDW May 22-24 San Diego, CA
K Kowdley1, P Ruane2, D Ain2, P Ingiliz3, S Mauss4, P Buggisch5, J Petersen5,
N Tsai6, Y Lee7, M Natha8, E Eggleton8, B Kreter8, DM Brainard8, M Curry9
1Swedish Medical Seattle, WA; 2Ruane Medical and Liver Health, Los Angeles, CA; 3Medizinisches Infektiologie Zentrum Berlin, Berlin; 4Center for HIV and Hepatogastroenterology, Duesseldorf; 5IFI Institut für Interdisziplinäre Medizin, Asklepios Klinik St. Georg, Hamburg Germany; 6Queens Medical Center, UH, Honolulu Hawaii; 7TRIO Health Analytics; 8Gilead Sciences Foster City, CA; 9Beth Israel, Boston, MA



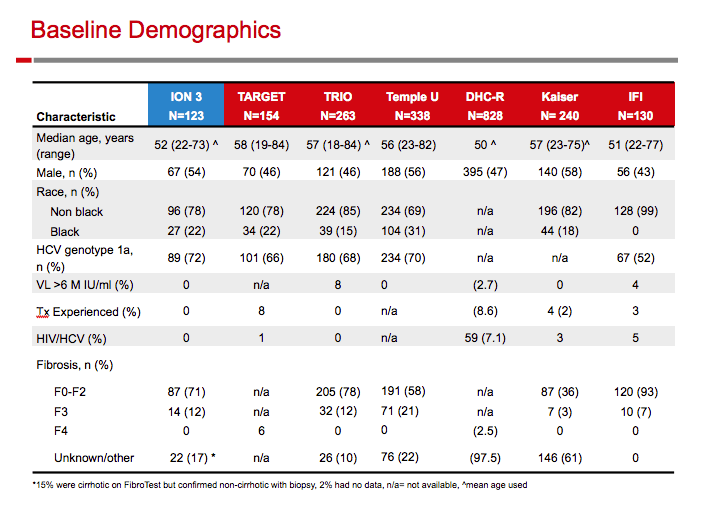
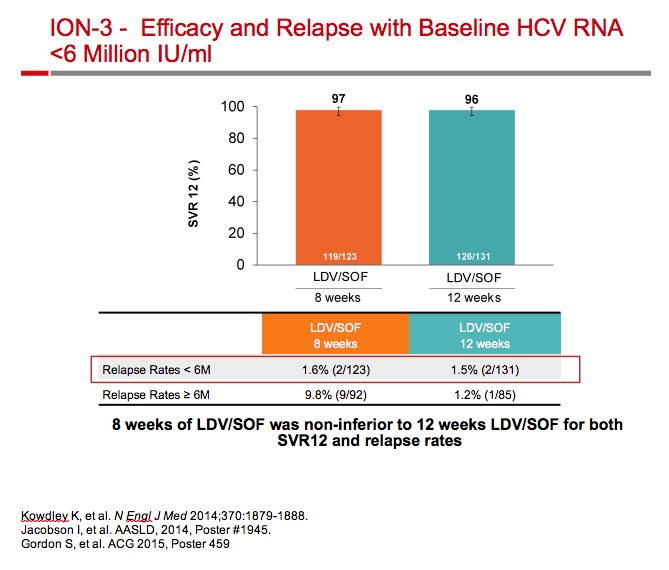
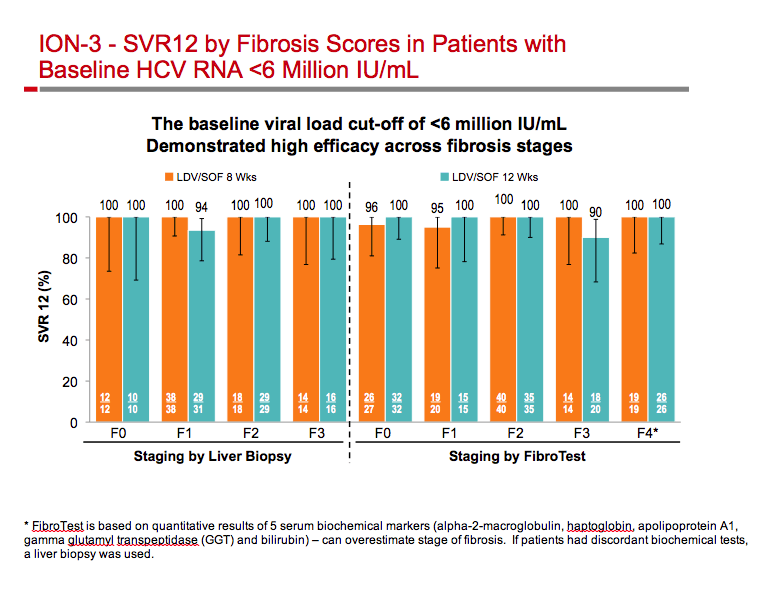
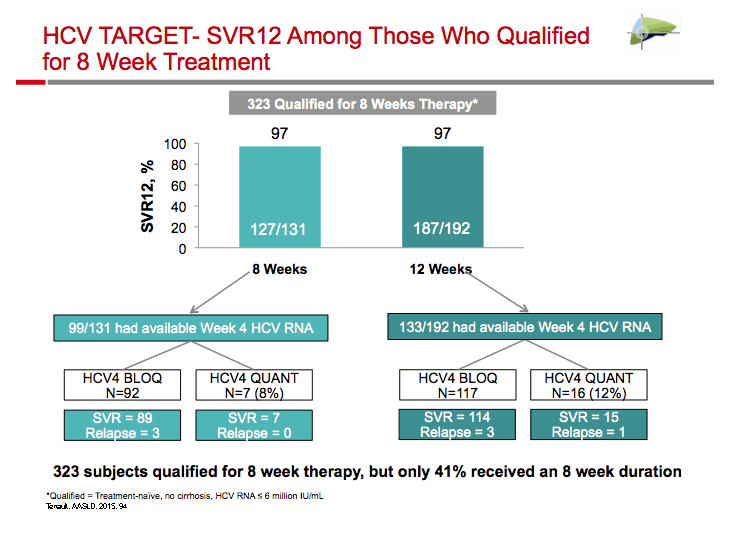
|
| |
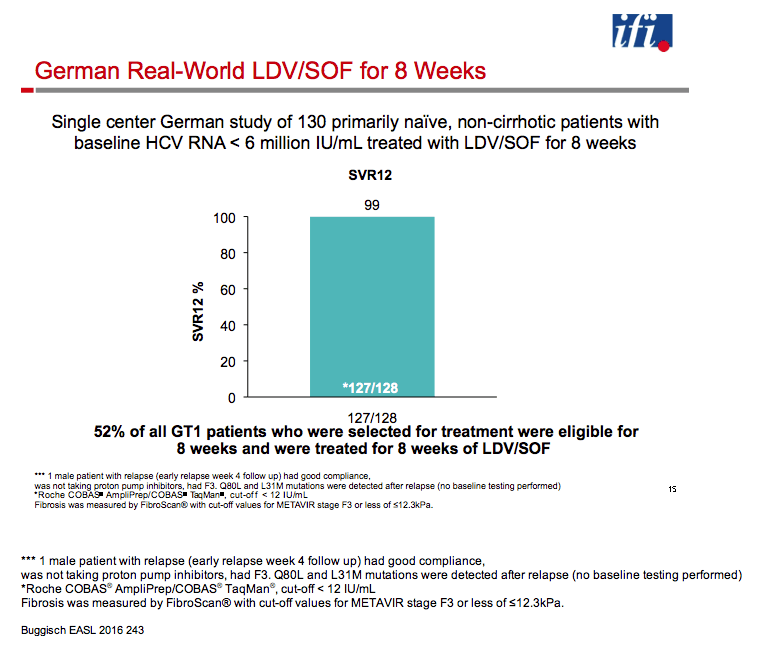
|
 |
 |
|
|