 |
 |
 |
| |
Integrated Analysis Of Studies Evaluating The Treatment of Chronic Hepatitis C Genotype 4 With Interferon-Free Sofosbuvir-Based Regimens
|
| |
| |
Reported by Jules Levin
Presented at DDW 2016
21-24 May 2016
San Diego, CA
P Ruane2, G Shiha3, A Abergel4, T Asselah4, A Kohli5, S Kottilil6, S Naggie7, W Doss8,
G Esmat9, CM Letterio1; T Hahambis1, M Natha1, B Kreter1, K Kersey1, D Brainard1, S Knox1
1Gilead Sciences, Foster City, CA, United States; 2Ruane Medical and Liver Health Institute, Los Angeles, CA, United States; 3Egyptian Liver Research Institute and Hospital, Mansoura, Egypt; 4Beaujon Hospital, Clichy, France; 5Creighton University School of Medicine, Phoenix, AZ, United States; 6NIAID, Baltimore, MD, United States; 7Duke Clinical Research Institute, Durham, NC, United States; 8National Hepatology and Tropical Medicine Research Institute, Cairo, Egypt; 9University of Cairo, Cairo, Egypt
AASLD: A Phase 3 Double-Blind Placebo-Controlled Evaluation of Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks in Genotype 1, 2, 4, 5, 6 HCV-Infected Patients:
Results of the ASTRAL-1 Study - (11/17/15) published:
http://www.natap.org/2015/AASLD/AASLD_52.htm
HepDart/2015: High Efficacy of Sofosbuvir/Velpatasvir Across 7 HCV Genotypes and 46 Subtypes: Pooled Data From the ASTRAL1, 2 and 3 Trials (12/14/15)
HepDart/2015: High Efficacy of Sofosbuvir/Velpatasvir In HCV Genotypes 1-6 Infected Patients With Cirrhosis: Pooled Data From the ASTRAL 1, 2 and 3 Trials (12/14/15)
AASLD/2015: Sofosbuvir/Velpatasvir Fixed-Dose Combination for the Treatment of HCV in Patients With Decompensated Liver Disease: the Phase 3 ASTRAL-4 Study / ASTRAL 1, 2 and 3- (11/30/15)



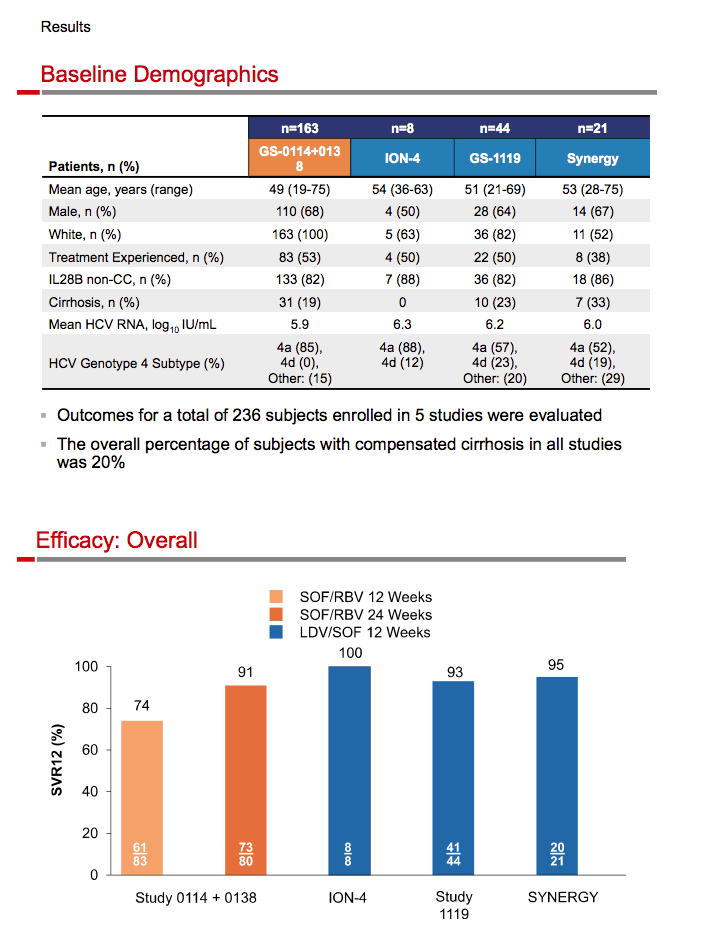
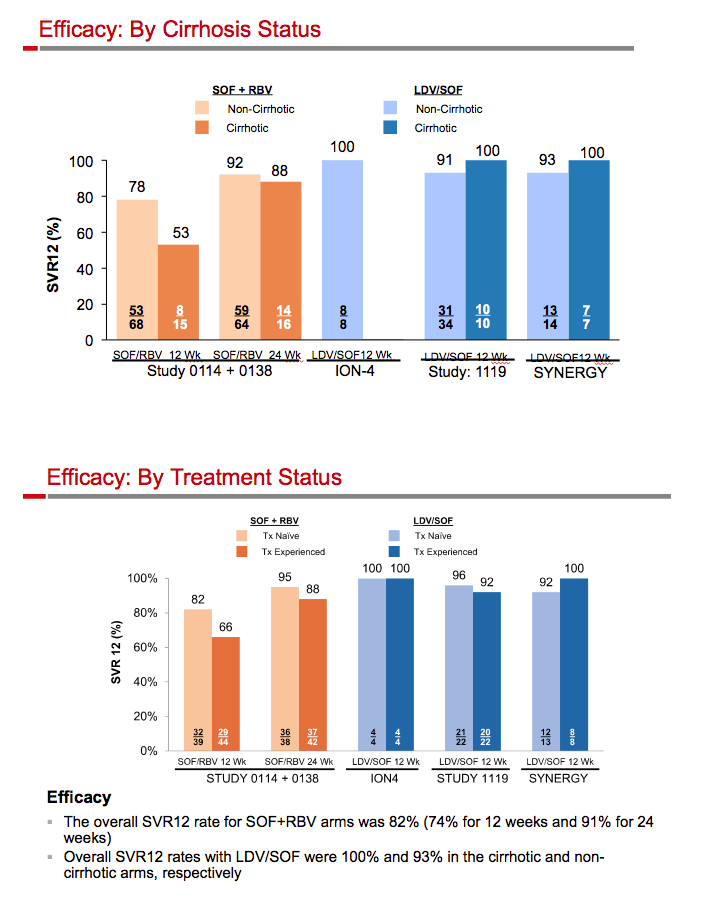
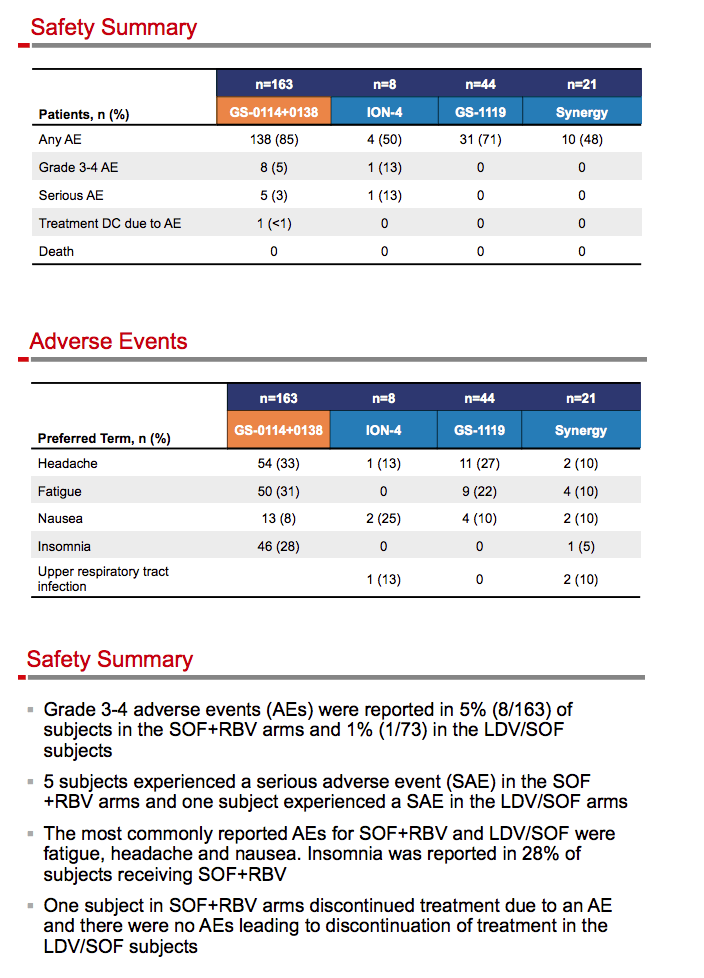

|
| |
|
 |
 |
|
|