 |
 |
 |
| |
EFFECTIVENESS AND SAFETY OF OMBITASVIR, PARITAPREVIR, RITONAVIR AND DASABUVIR PATIENTS WITH GENOTYPE 1 CHRONIC HEPATITIS C VIRUS INFECTION: RESULTS FROM THE SPANISH REAL WORLD COHORT
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Jose Luis Calleja* 1, 2, Diego Rincon3, Belen Ruiz-Antoran4, Begoña Sacristan5, Christie Perello1, Sabela Lens2, 6, Inmaculada Fernanadez7, Francisco Gea2, 8, Rosa Maria Morillas9, Joaquin Cabezas10, Jose Manuel Pascasio11, Martin Prieto12, Juan Turnes13, Miguel A Serra14, Juan Arenas15, Xavier Torras2, 16, Lucia Bonet17, Conrado Fernandez18, Javier Samaniego2, 19, Alicia Hernandez-Albujar20, Javier Ampuero2, 21, Jose Maria Moreno22, Federico Saez-Royuela23, Carmen Alvarez-Navascues24, Moises Diago25, Gloria Sanchez-Antolin26, Juan de la Vega27, Juan Jose Sanchez-Ruano28, Raul Andrade2, 29, Maria Butti2, 30, Jose A Carrion31, Esther Molina32, Miguel A Simon33, Jose Ramon Salcines34, Francisco Jorquera35, Silvia Montoliu36, Adriana Ahumada3, Tania Hernaez5, Javier Crespo10
1Department of Gastroenterology, Hospital Universitario Puerta de Hierro Majadahonda, 2CIBERehd, 3Department of Gastroenterology, Hospital Universitario Gregorio Marañon, 4Department of Clinical Pharmacology, Hospital Universitario Puerta de Hierro Majadahonda, Madrid, 5Department of Gastroenterology, Hospital San Pedro, Logroño, 6Liver Unit, Hospital Clinic, Barcelona, 7Department of Gastroenterology, Hospital Universitario 12 de Octubre, 8Department of Gastroenterology, Hospital Universitario Ramon y Cajal, Madrid, 9Department of Gastroenterology, Hospital Germans Trias I Pujol, Barcelona, 10Department of Gastroenterology, Hospital Universitario Marques de Valdecilla, Santander, 11Department of Gastroenterology, Hospital Universitario Virgen del Rocio, Sevilla, 12Department of Gastroenterology, Hospital Universitario La Fe, Valencia, 13Department of Gastroenterology, Hospital de Montecelo, Pontevedra, 14Department of Gastroenterology, Hospital Clinico de Valencia, Valencia, 15Department of Gastroenterology, Hospital Universitario de Donostia, San Sebastian, 16Department of Gastroenterology, Hospital Santa Creu i Sant Pau, Barcelona, 17Department of Gastroenterology, Hospital Universitario son Espases, Palma de Mallorca, 18Department of Gastroenterology, Hospital Universitario Fundación Alcorcón, 19Department of Gastroenterology, Hospital Universitario La Paz, Madrid, 20Department of Gastroenterology, Hospital General Universitario de Ciudad Real, Ciudad Real, 21Department of Gastroenterology, Hospital Universitario de Valme, Sevilla, 22Department of Gastroenterology, Complejo Hospitalario Universitario de Albacete, Albacete, 23Department of Gastroenterology, Hospital Universitario de Burgos, Burgos, 24Department of Gastroenterology, Hospital Universitario Central de Asturias, Oviedo, 25Department of Gastroenterology, Hospital Universitario General de Valencia, Valencia, 26Department of Clinical Pharmacology, Hospital Universitario Rio Hortega, Valladolid, 27Department of Gastroenterology, Hospital San Agustin, Aviles, 28Department of Gastroenterology, Hospital Universitario de Toledo, Toledo, 29Department of Gastroenterology, Hospital Universitario Virgen de la Victoria, Malaga, 30Department of Gastroenterology, Hospital Universitario Vall D´Hebrón, 31Department of Gastroenterology, Hospital del Mar, Barcelona, 32Department of Gastroenterology, Hospital Clínico Universitario Santiago, Santiago de Compostela, 33Department of Gastroenterology, Hospital Clinico Universitario Lozano Blesa, Zaragoza, 34Department of Gastroenterology, Hospital Comarcal Laredo, Laredo, 35Department of Gastroenterology, Complejo Asistencial Universitario León, Leon, 36Department of Gastroenterology, Hospital Universitario Tarragona, Tarragona, Spain
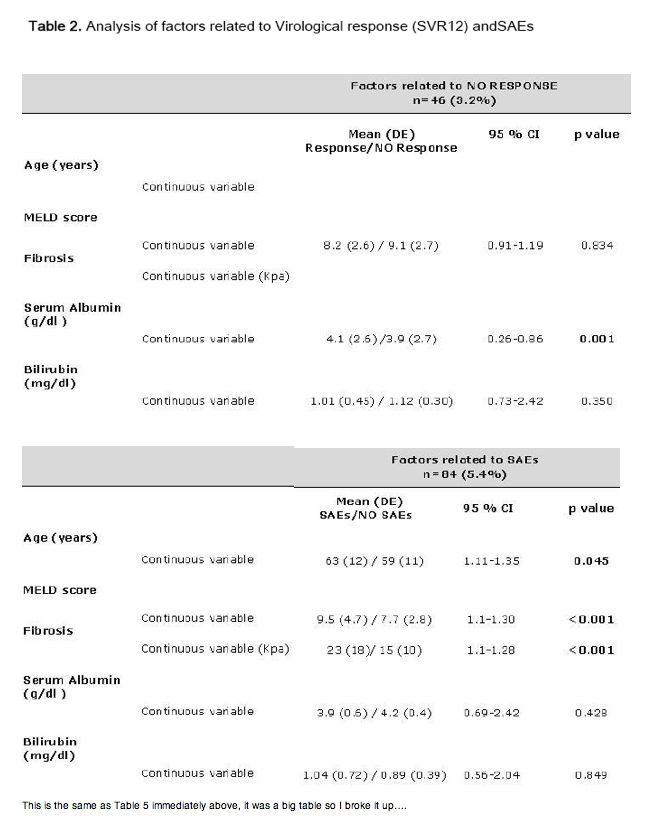
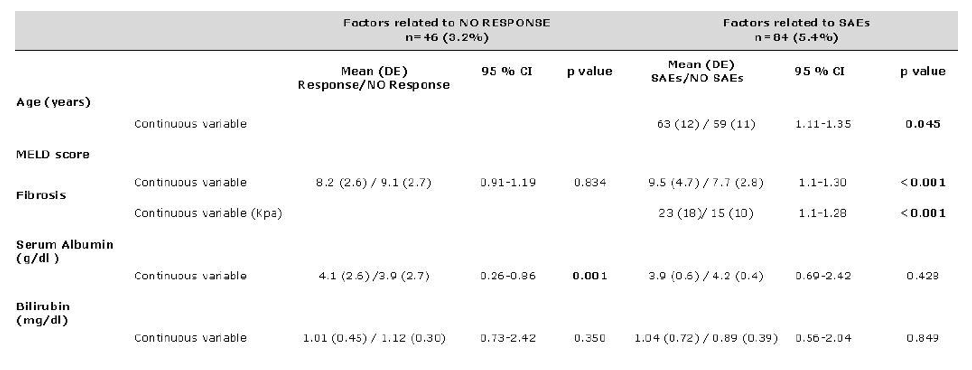
Conclusion. The regimen of ombitasvir/paritaprevir/ritonavir and dasabuvir with or without RBV achieved a high rate of efficacy SVR (96.8%) in clinical practice. It was well tolerated and was associated with low rates of treatment discontinuation. The presence of hypoalbuminemia (<3.5g/dl) was found to be a significant negative predictor of SVR12. The incidence of SAEs were significantly more frequent in patients with advanced liver disease.

|
| |
|
 |
 |
|
|