 |
 |
 |
| |
RG-101 DEMONSTRATES FAVORABLE IN VITRO ANTIVIRAL ACTIVITY AND CROSS RESISTANCE PROFILE TO SUPPORT CLINICAL COMBINATION STUDIES IN HCV PATIENTS
|
| |
| |
Reported by Jules Levin
European Association for the Study of the Liver April 13-17, 2016 Barcelona, Spain
Steven Neben, Scott Davis, Paul Grint, Amy K. Patick Regulus Therapeutics, Inc, San Diego, CA
AASLD: Towards a Single Treatment Cure for HCV: Reformulation of the Pangenotypic NS5B NNI GSK2878175 as a Long Acting Parenteral (LAP) - (11/21/16)
LA JOLLA, Calif., June 1, 2016 /PRNewswire/ -- Regulus Therapeutics Inc. (NASDAQ: RGLS), a biopharmaceutical company leading the discovery and development of innovative medicines targeting microRNAs (miR), today announced that it has expanded its clinical trial collaboration agreement with GSK (NYSE: GSK) for the development of RG-101, Regulus' wholly-owned, GalNAc-conjugated anti-miR that targets miR-122. In the expanded collaboration, the companies plan to conduct a multi-centered, randomized, dose-ranging Phase II study evaluating the combination of RG-101 and GSK's long-acting parenteral ("LAP") formulation of GSK2878175 as a potential single-visit cure in patients chronically infected with HCV. This study will be conducted outside the United Statesand is planned to begin in the fourth quarter of 2016. Based on predicted enrollment rates, interim results from this expanded collaboration should be available in the second half of 2017, enabling a potential initiation of a pivotal study in late 2017. As with the initial collaboration, both parties will share equally in the costs associated with the study. Neither Regulus nor GSK has any further obligations or commitments to each other beyond this expanded clinical collaboration agreement.
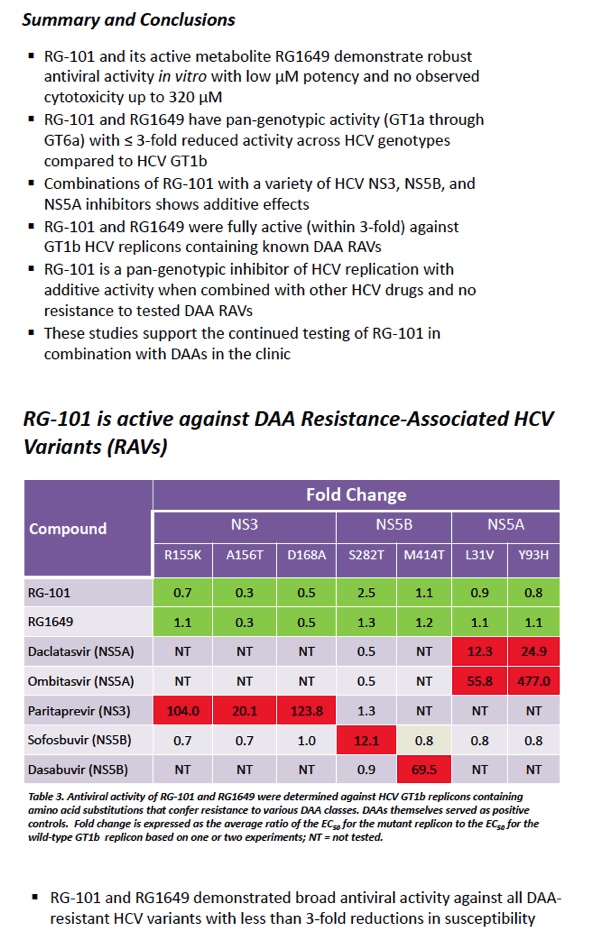

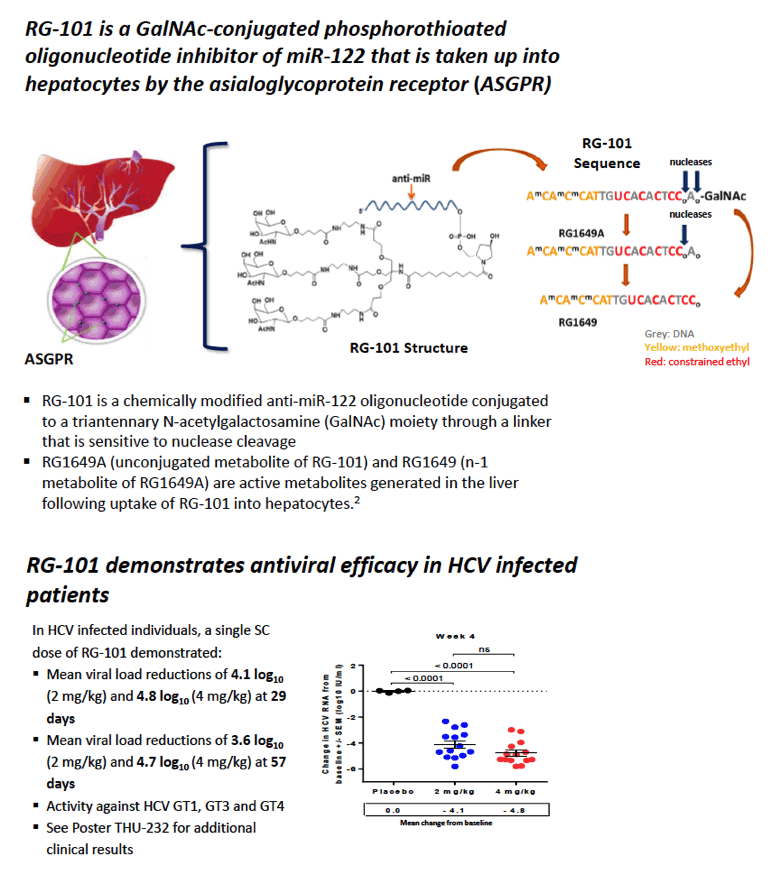
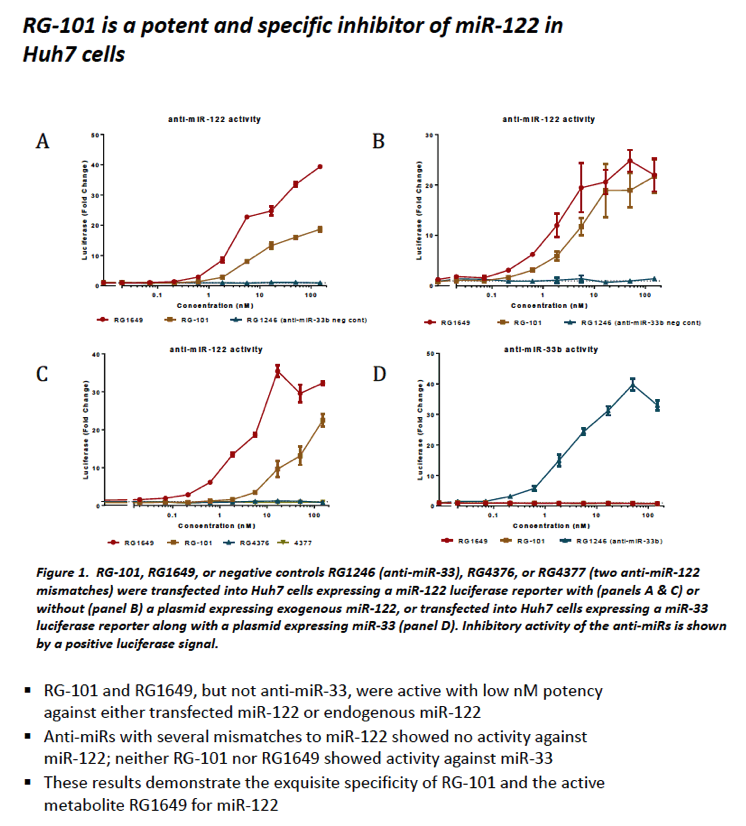
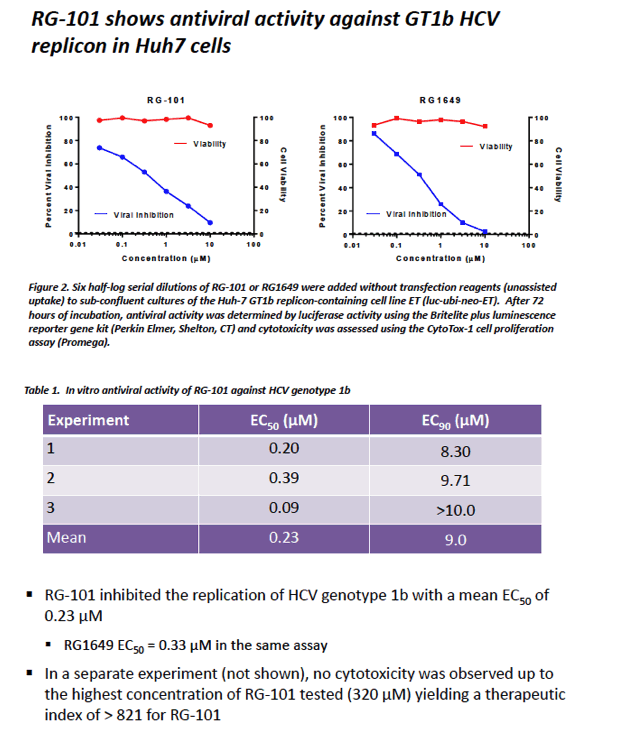
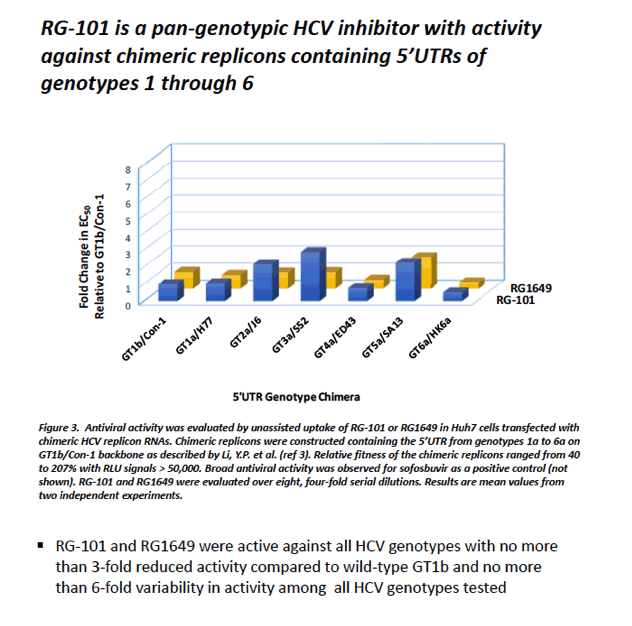
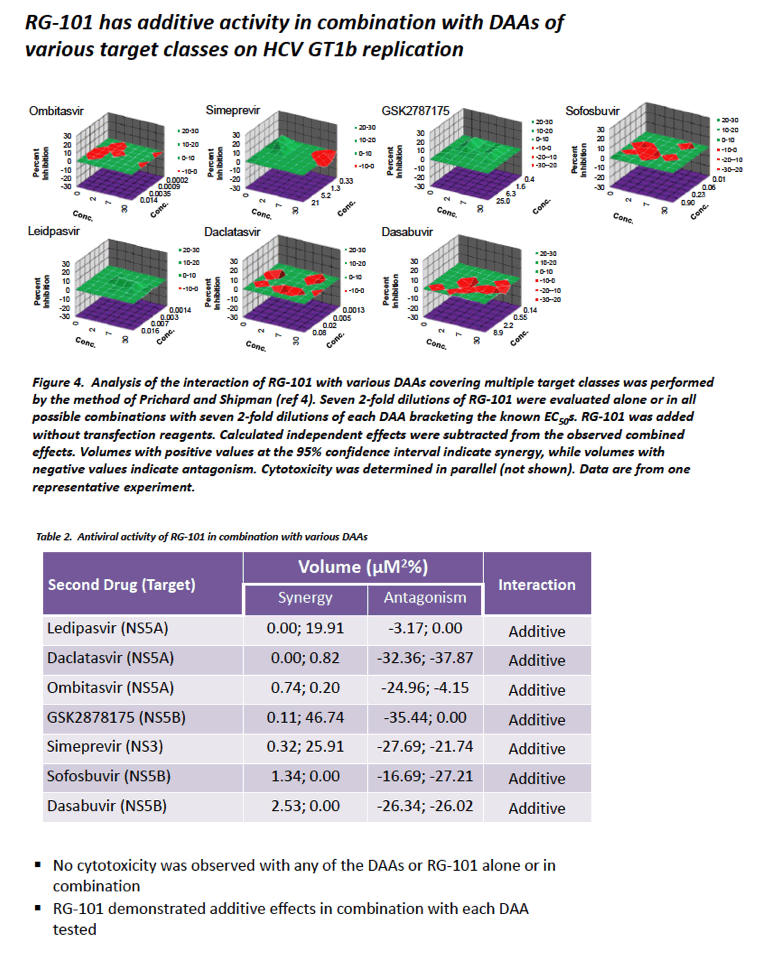
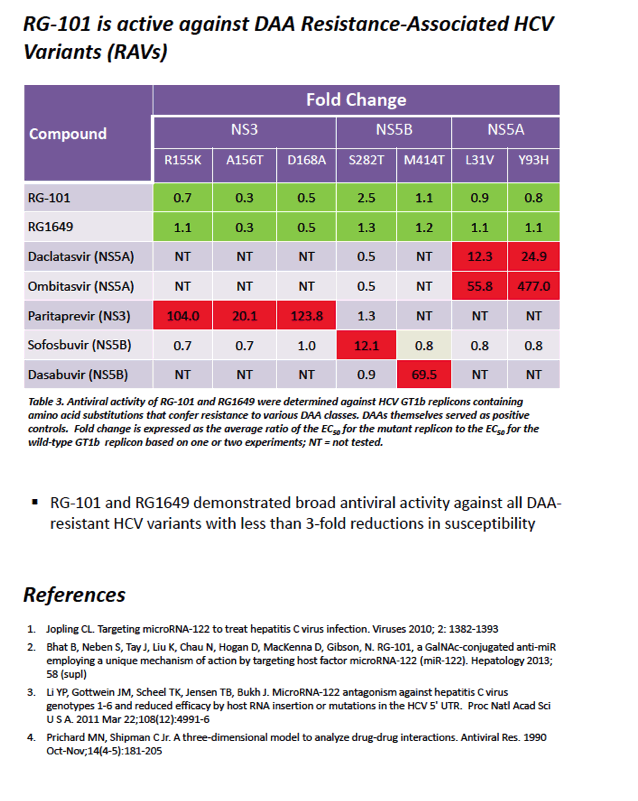
|
| |
|
 |
 |
|
|