 |
 |
 |
| |
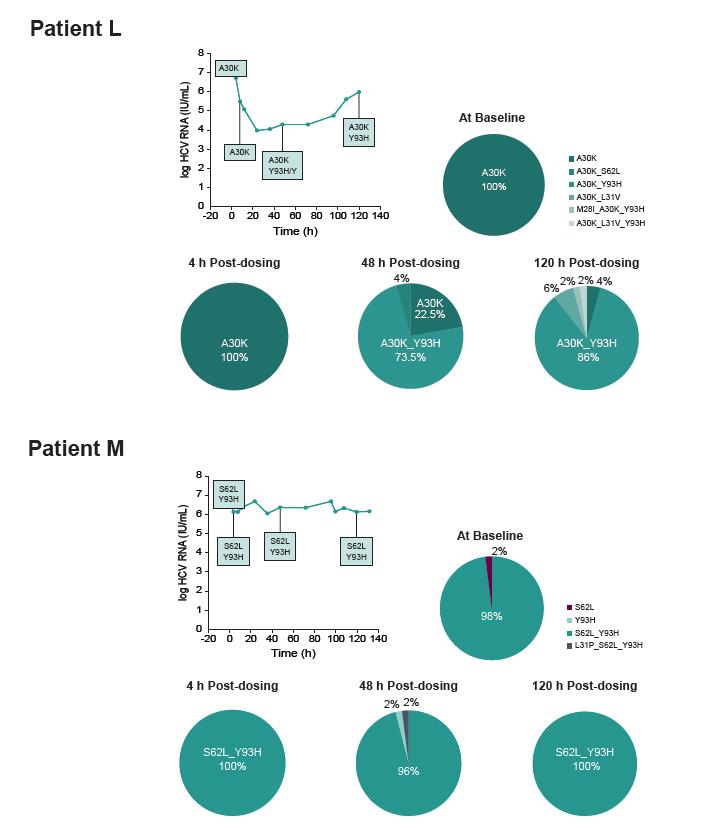
In a 5-day Monotherapy Trial, MK-8408 Demonstrated Potent
Antiviral Activity and Improved Resistance Profile in HCV
Patients With Genotypes 1, 2, and 3 Infections
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Ernest Asante-Appiah1, William Marshall1, Edward Gane2, Serghei Popa3, Patricia McMonagle1, Stephanie Curry1, Lata Maganti1, Wei Gao1, Graigory Garrett1, Caroline Cilissen4, Inge De Lepeleire4, Todd Black1, Daria Hazuda1, Joan Butterton1
1Merck & Co., Inc., Kenilworth, NJ, USA; 2Auckland Clinical Studies, Auckland, New Zealand; 3Republican Clinical Hospital of Moldova, Chisinau, Republic of Moldova; 4MSD, Brussels, Belgium



|
| |
|
 |
 |
|
|