 |
 |
 |
| |
AbbVie Presents New Phase 2 Data for Investigational, Once-Daily, Ribavirin-Free,
Pan-Genotypic Regimen of ABT-493 and ABT-530 for Hepatitis C Genotypes 1-6
|
| |
| |
Apr 16, 2016
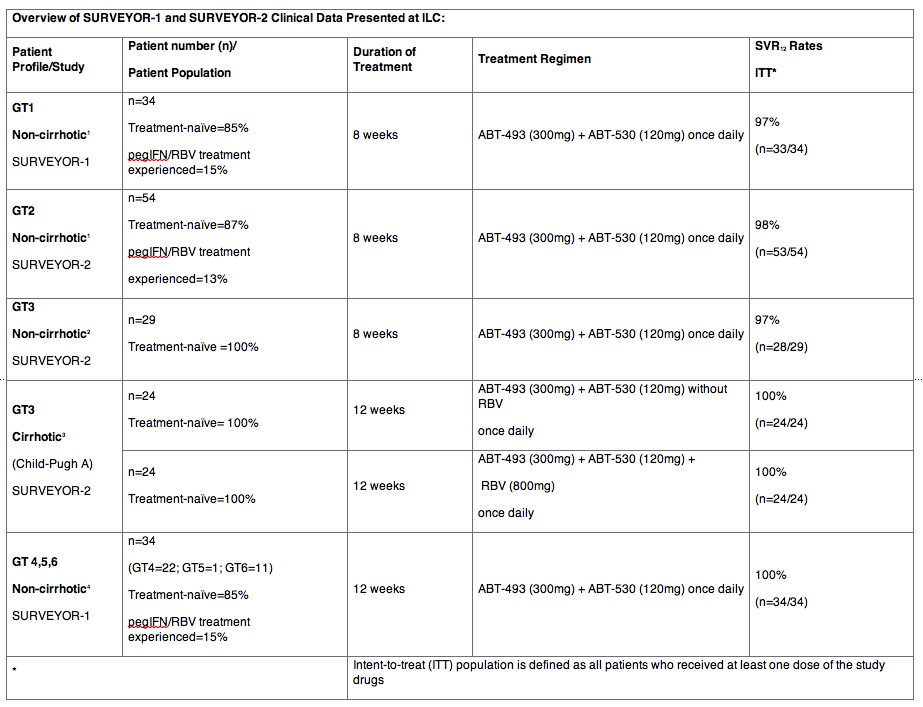
- 97-98 percent SVR12 achieved with eight weeks of ABT-493 and ABT-530 in genotypes 1-3 hepatitis C virus patients without cirrhosis in SURVEYOR-1 and 2 studies(1,2)
- 100 percent SVR12 achieved with 12 weeks of treatment in difficult-to-treat genotype 3 patients with compensated cirrhosis (Child-Pugh A) new to therapy (3)
- 100 percent SVR12 achieved with 12 weeks of treatment in genotypes 4-6 patients without cirrhosis; eight-week duration investigated in this ongoing study (4)
BARCELONA, April 16, 2016 /PRNewswire/ -- AbbVie (NYSE: ABBV), a global biopharmaceutical company, today announced that with eight weeks of treatment, 97-98 percent of genotype 1-3 (GT1-3) chronic hepatitis C virus (HCV) infected patients without cirrhosis treated with AbbVie's investigational, once-daily, ribavirin (RBV)-free, pan-genotypic regimen of ABT-493 and ABT-530 achieved sustained virologic response at 12 weeks post-treatment (SVR12).1,2 Results for GT1 (n=33/34), GT2 (n=53/54) and treatment-naïve GT3 (n=28/29) patients were based on an Intent-to-Treat (ITT) analysis.1,2 Additionally, 100 percent (n=34/34) of genotype 4-6 (GT4-6) chronic HCV infected patients without cirrhosis achieved SVR12 with 12 weeks of treatment.4 These new data from the Phase 2 SURVEYOR-1 and SURVEYOR-2 studies will be presented at The International Liver Congress™ (ILC) 2016 in Barcelona, Spain.
"These results move us closer to our ultimate goal of providing a treatment option for as many hepatitis C patients as possible. We will continue to examine our investigational, pan-genotypic regimen through our dedicated clinical trial program, including an eight-week duration across all genotypes," said Rob Scott, M.D., vice president, development and chief medical officer, AbbVie.
In separate late-breaking data from the SURVEYOR-2 study, 100 percent of GT3 chronic HCV infected patients with compensated cirrhosis (Child-Pugh A) new to therapy achieved SVR12 with 12 weeks of treatment both with and without RBV (n=24/24 in each arm).3 No patients discontinued treatment due to adverse events.3 Data in GT3 chronic HCV infected patients with and without cirrhosis were featured in the official ILC 2016 press program.
"The recent evolution in hepatitis C treatment has resulted in high cure rates for many patients with specific genotypes, but there remain distinct areas of unmet need," said Paul Kwo, M.D., professor of medicine at the Indiana University School of Medicine. "These new data show us the potential of ABT-493 and ABT-530 in genotype 3 patients new to therapy even with the added complication of compensated cirrhosis."
In a pooled analysis of 531 patients across both SURVEYOR studies, of five treatment regimens of ABT-493 and ABT-530 evaluated, the most commonly reported adverse events were fatigue (18 percent), headache (17 percent), nausea (13 percent) and diarrhea (10 percent).5 Three patients across all study arms evaluated to date, two of whom received RBV, discontinued study drugs early due to adverse events.5
About SURVEYOR-11,4,5
SURVEYOR-1 is an ongoing Phase 2 two-part study designed to evaluate the safety and efficacy of ABT-493 and ABT-530, with and without RBV, for eight to 12 weeks, in cirrhotic and non-cirrhotic adult genotype 1 patients, and non-cirrhotic adult patients with genotypes 4, 5 or 6 chronic HCV infection who were new to therapy or did not respond to previous treatment with pegylated interferon (pegIFN)/RBV (null responder).
About SURVEYOR-21,2,3,5
SURVEYOR-2 is an ongoing Phase 2, four-part study designed to evaluate the safety and efficacy of ABT-493 and ABT-530, with or without RBV, in adult patients with genotypes 2, 3, 4, 5 or 6 chronic HCV infection who were new to therapy or had failed previous treatment with pegylated interferon (pegIFN)/RBV.
The primary endpoint of both studies is the percentage of subjects achieving SVR12.
Safety and efficacy data for Part 1 of the studies were presented at The Liver Meeting® 2015, the Annual Meeting of the American Association for the Study of Liver Diseases (AASLD) in San Francisco.
About pooled safety analysis of SURVEYOR-1 and SURVEYOR-25
531 patients were included in this safety analysis: 26 percent GT1, 24 percent GT2, 43 percent GT3, and 6 percent with GT4, 5 or 6 infection. Patients across genotypes received ABT-493/ABT-530 at five doses: 300/120mg (n=258), 300/120mg with RBV (n=27), 200/120mg (n=121), 200/120mg with RBV (n=56), and 200/40mg (n=69).
About AbbVie's HCV Clinical Development Program
AbbVie's HCV clinical development program is intended to advance scientific knowledge and the clinical care of people with chronic HCV infection by investigating pan-genotypic (genotypes 1-6), all-oral, ribavirin-free, once-daily treatment for 12 weeks. An eight-week treatment duration with ABT-493 and ABT-530 will be investigated across all genotypes in our comprehensive Phase 2/Phase 3 clinical trial program, which focuses on areas of ongoing need in HCV.
AbbVie's investigational regimen includes 300mg ABT-493, an NS3/4A protease inhibitor, and 120mg ABT-530, an NS5A inhibitor.
ABT-493 was discovered during the ongoing collaboration between AbbVie and Enanta Pharmaceuticals (NASDAQ: ENTA) for HCV protease inhibitors and regimens that include protease inhibitors.
About AbbVie
AbbVie is a global, research-based biopharmaceutical company formed in 2013 following separation from Abbott Laboratories. The company's mission is to use its expertise, dedicated people and unique approach to innovation to develop and market advanced therapies that address some of the world's most complex and serious diseases. Together with its wholly-owned subsidiary, Pharmacyclics, AbbVie employs more than 28,000 people worldwide and markets medicines in more than 170 countries. For further information on the company and its people, portfolio and commitments, please visit www.abbvie.com. Follow @abbvie on Twitter or view careers on our Facebook or LinkedIn page.
1 Poordad, F et al. High SVR Rates with the Combination of ABT-493 + ABT-530 for 8 Weeks in Non-Cirrhotic Patients with HCV Genotype 1 or 2 Infection. Poster presentation #SAT-157; presented at The International Liver Congress™ (ILC), the Annual Meeting of the European Association for the Study of the Liver (EASL) in Barcelona, Spain, April 13-17, 2016.
2 Muir, A et al. High SVR Rates with ABT-493 + ABT-530 Co-Administered for 8 Weeks in Non-Cirrhotic Patients with HCV Genotype 3 Infection. Oral presentation #PS098; presented at The International Liver Congress™ (ILC), the Annual Meeting of the European Association for the Study of the Liver (EASL) in Barcelona, Spain, April 13-17, 2016.
3 Kwo, P et al. 100% SVR12 with ABT-493 And ABT-530 with or without Ribavirin in Treatment-Naïve HCV Genotype 3-Infected Patients with Cirrhosis; Late Breaker presentation #LB01; presented at The International Liver Congress™ (ILC), the Annual Meeting of the European Association for the Study of the Liver (EASL) in Barcelona, Spain, April 13-17, 2016
4 Gane, E et al. 100% SVR4 and Favorable Safety of ABT-493 + ABT-530 Administered for 12 Weeks in Non-Cirrhotic Patients with Genotypes 4,5, or 6 Infection (SURVEYOR-I). Poster presentation #SAT-137; presented at The International Liver Congress™ (ILC), the Annual Meeting of the European Association for the Study of the Liver (EASL) in Barcelona, Spain, April 13-17, 2016.
5 Kwo, P et al. Safety of ABT-493 and ABT-530 Co-Administered in Patients with HCV Genotype 1-6 Infection: Results From the SURVEYOR-I and SURVEYOR-II Studies; Poster presentation #SAT-239; presented at The International Liver Congress™ (ILC), the Annual Meeting of the European Association for the Study of the Liver (EASL) in Barcelona, Spain, April 13-17, 2016.
SOURCE AbbVie
Contact(s)
Media: Jane Woo, +1 (847) 937-4754, jane.woo@abbvie.com, Investor Relations: Liz Shea, +1 (847) 935-2211, liz.shea@abbvie.com
|
| |
|
 |
 |
|
|