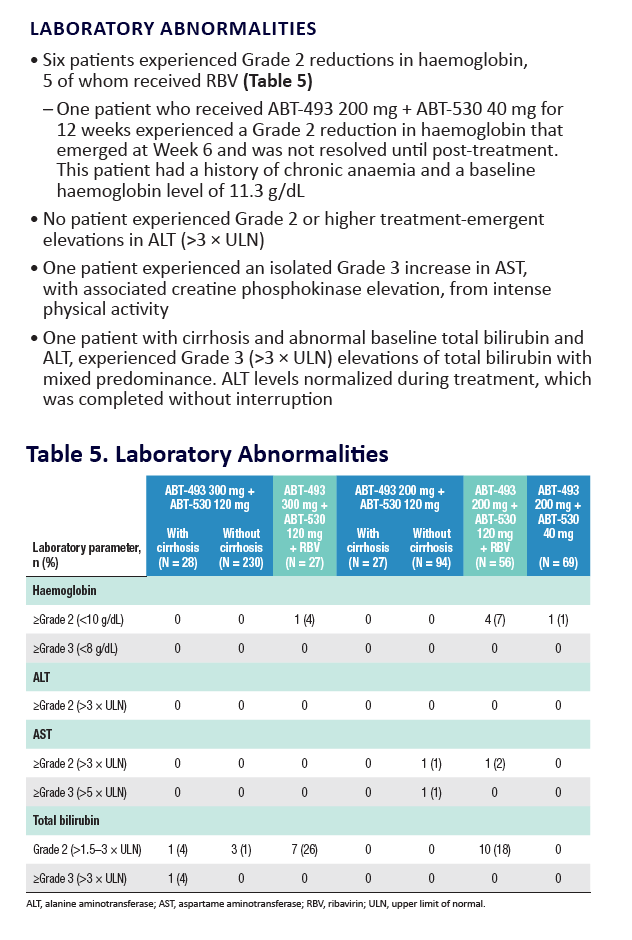
 |
 |
 |
| |
Safety of ABT-493 + ABT-530 Co-Administered in Patients With HCV
Genotype 1-6 Infection:
Results From the SURVEYOR-I and SURVEYOR-II Studies
|
| |
| |
Reported by Jules Levin
EASL 206 April 14-17 Barcelona
Paul Kwo1, Fred Poordad2, Ariel Porcalla3, Edward Gane4, Jacob Lalezari5, David Wyles6, Ran Liu3, Jens Kort3, Federico J Mensa3
1Indiana University School of Medicine, Indianapolis, Indiana, United States; 2Texas Liver Insti tute, University of Texas Health Science Center, San Antonio, Texas, United States; 3AbbVie Inc., North Chicago, Illinois, United States;
4Liver Unit, Auckland City Hospital, Auckland, New Zealand; 5Quest Clinical Research, San Francisco, California, United States; 6University of California San Diego, La Jolla, California, United States


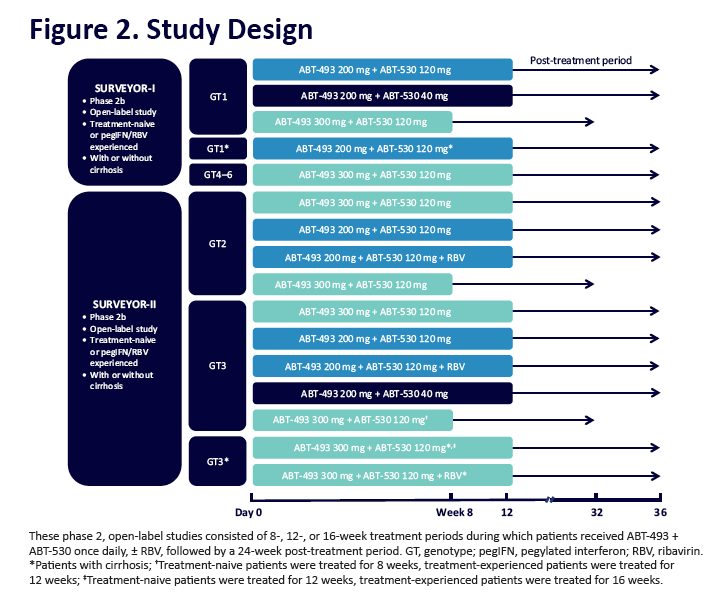

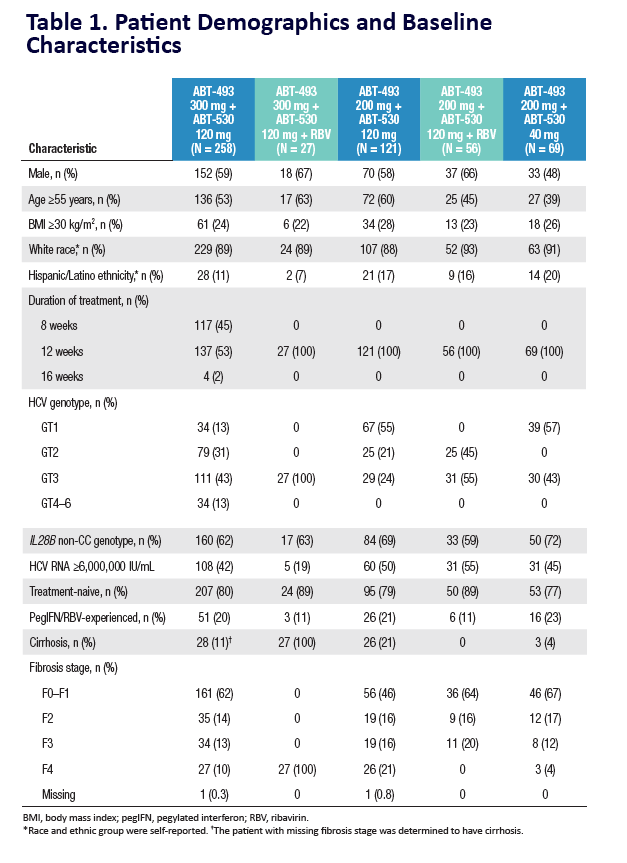
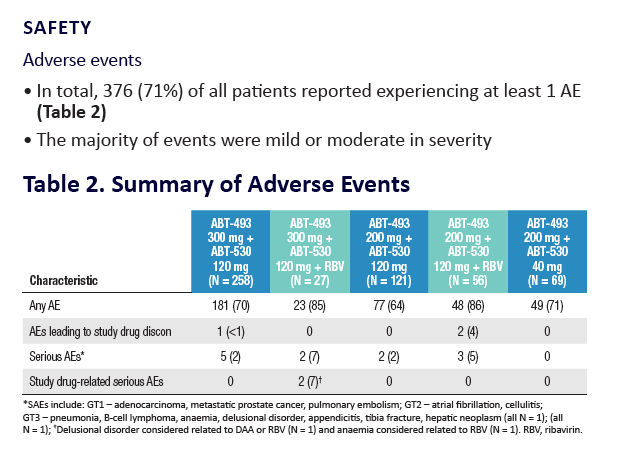

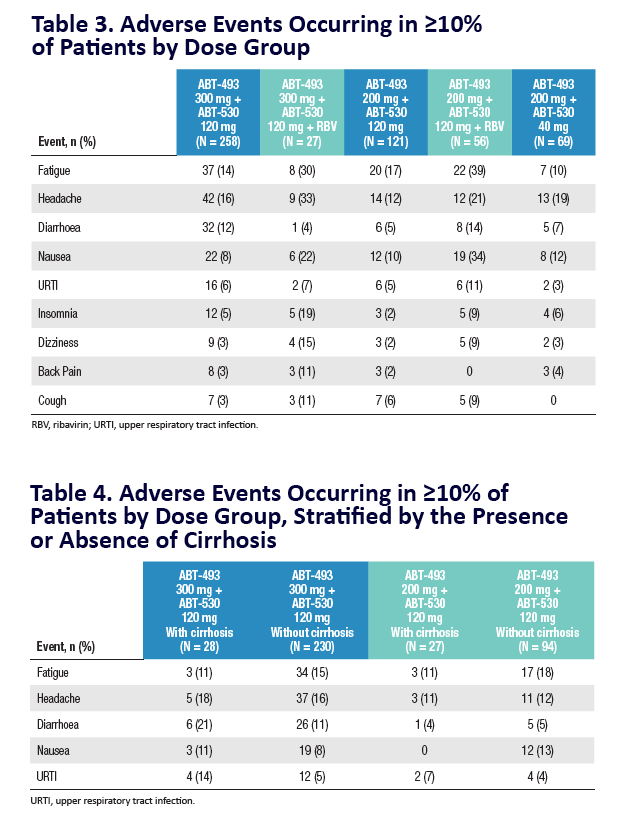
|
| |
|
 |
 |
|
|