 |
 |
 |
| |
Real-World Data on the Use of Ribavirin With Ombitasvir/Paritaprevir/r With or Without Dasabuvir in HCV Genotype 1- or 4-Infected Pati ents From the German Hepatitis C Registry
|
| |
| |
Reported by jules Levin
EASL 2016 April 14-17 Barcelona
Tania M Welzel1, Peter Buggisch2, Thomas Berg3, Kersti n Stein4, Jan Hetti nger5, Henning Kleine5
1JW Goethe University, Frankfurt, Germany; 2IFI Hamburg, Hamburg, Germany; 3Clinic of Gastroenterology and Rheumatology, University Hospital Leipzig, Leipzig, Germany; 4Ott o-von-Guericke-University Magdeburg, Magdeburg, Germany; 5AbbVie Deutschland GmbH & Co. KG, Wiesbaden, Germany


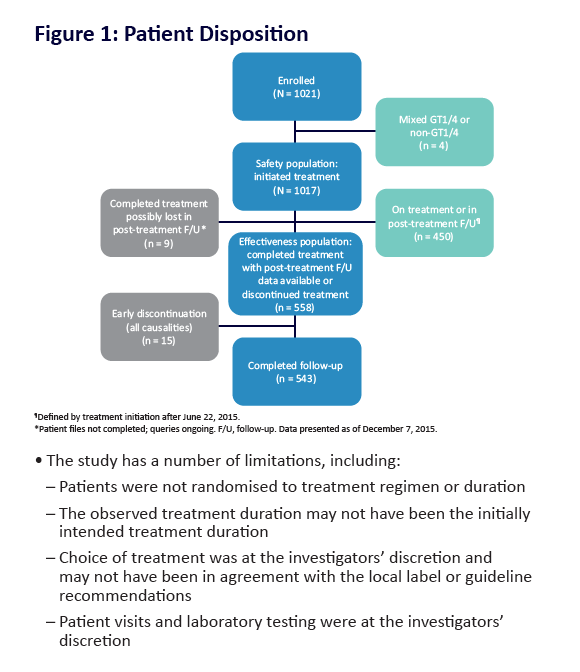

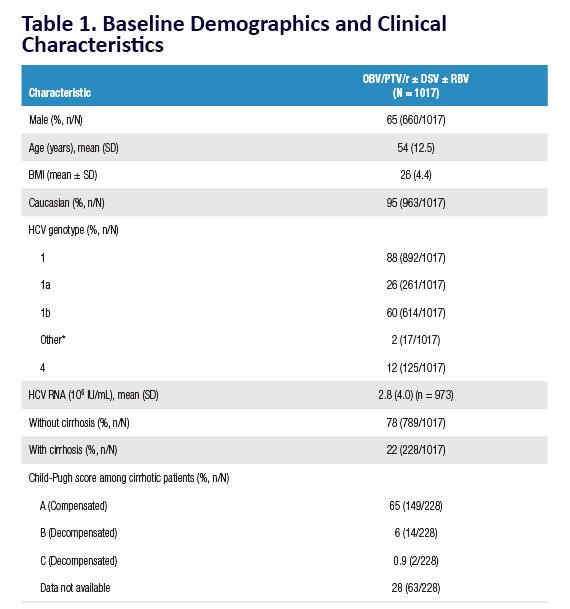

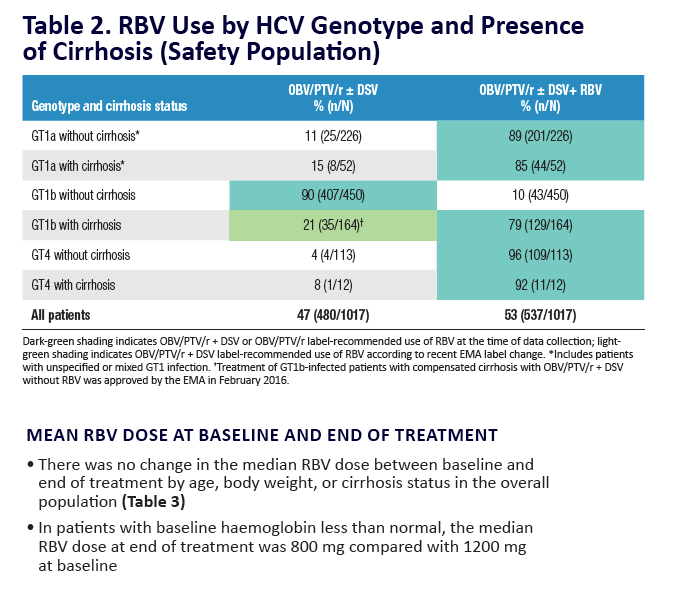
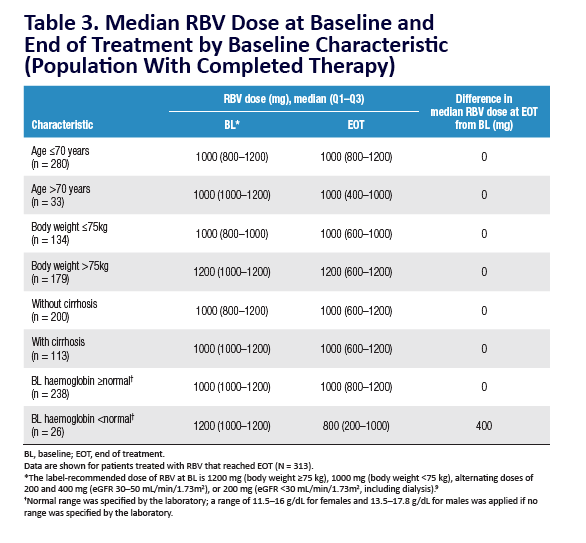
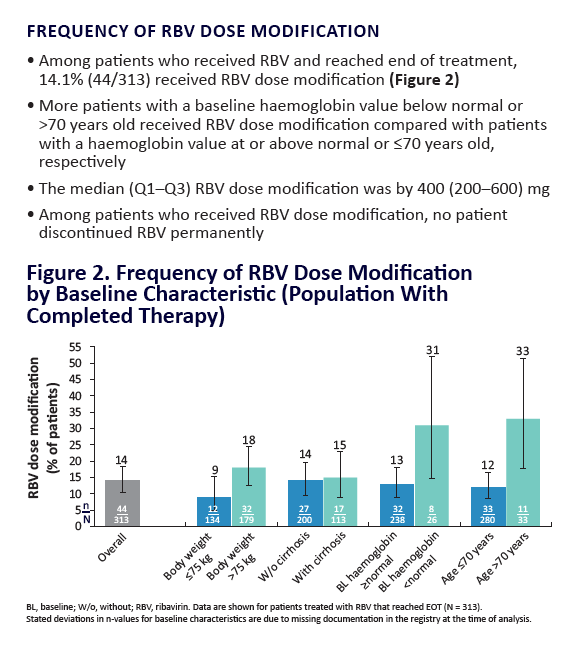
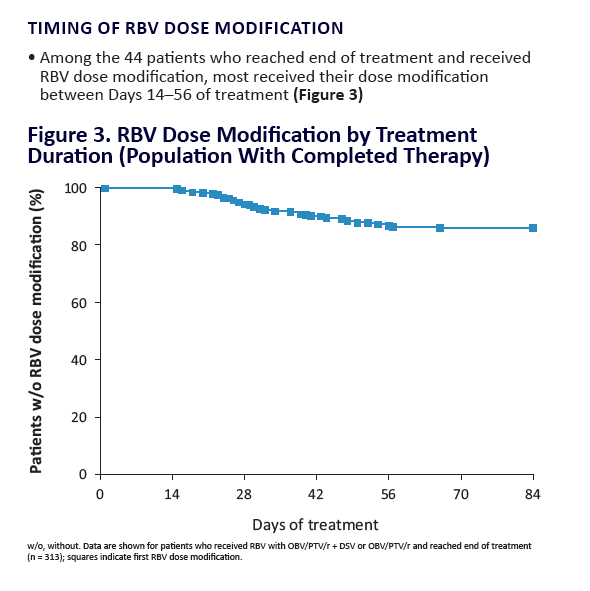

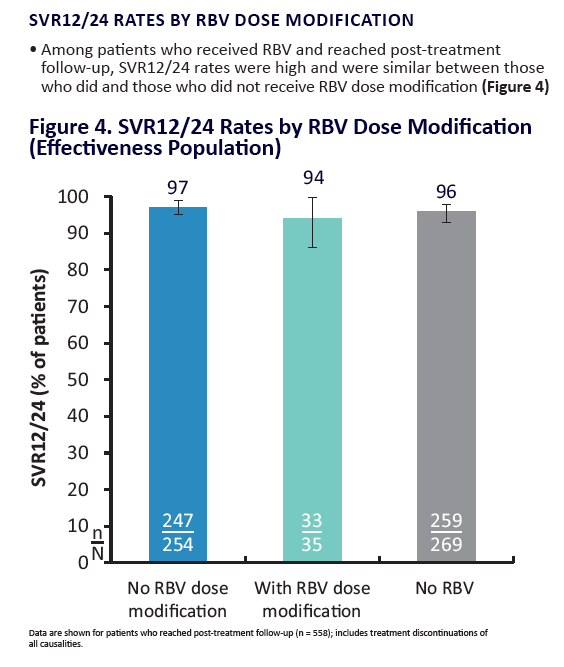

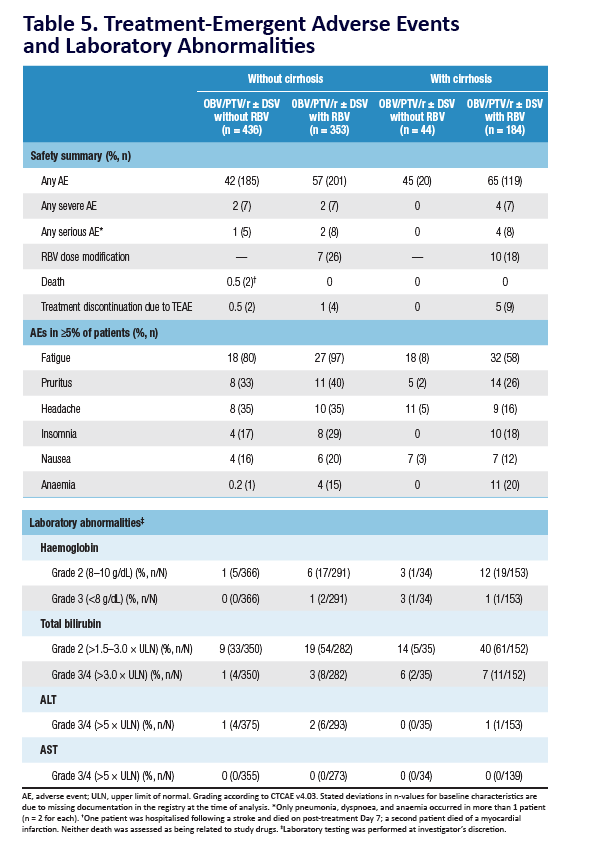
AE, adverse event; ULN, upper limit of normal. Grading according to CTCAE v4.03. Stated deviati ons in n-values for baseline characteristi cs are
due to missing documentati on in the registry at the ti me of analysis. *Only pneumonia, dyspnoea, and anaemia occurred in more than 1 pati ent
(n = 2 for each). One pati ent was hospitalised following a stroke and died on post-treatment Day 7; a second pati ent died of a myocardial
infarcti on. Neither death was assessed as being related to study drugs. Laboratory testi ng was performed at investi gator's discreti on.
|
| |
|
 |
 |
|
|