 |
 |
 |
| |
Impact of direct anti-viral agents on inactivation/de-listing of liver transplant candidates listed for decompensated C cirrhosis: a European study
|
| |
| |
Reported by Jules Levin
EASL 2016 E=April 14-17 Barcelona
Luca S Belli1, Marina Berenguer2, Paolo Cortesi3, Mario Strazzabosco4, Susanne-Rasoul Rockenschaub5, Silvia Martini6, Cristina Morelli7, Francesca Donato8, Riccardo Volpes9, Georges-Philippe Pageaux10, Audrey Coilly11, Stefano Fagiuoli12, Giuliana Amaddeo13, Giovanni Perricone1, Carmen Vinaixa2, Gabriela Berlakovich5, Rita Facchetti3, Wojciech Polak14, Paolo Muiesan15 and Christophe Duvoux13
for the European Liver and Intestine Association (ELITA)
at AASLD 2015.....
Curing Decompensated Wait-Listed HCV Patients with the New DAAs: Potential Significant Impact on Liver Transplant Wait List and Organ Allocation - (12/09/15)
Hepatitis C cure could avoid liver transplant in some Cirrhotic patients on Dialysis listed for simultaneous liver-kidney transplantation - (12/16/15).....
"We describe our experience of 9 HCV infected patients with cirrhosis and end stage renal disease (ESRD) on hemodialysis who were initially listed for SLKT (simultaneous liver kidney transplant) at the Miami Transplant Institute from 2011 to 2014.......Seven (78%) patients achieved SVR12 and from them, all (100%) were able to be delisted from liver transplant and were considered only for kidney transplant."
Current Trends in Liver Transplantation among HCV-Infected Baby Boomer Generation in the United States: A Case for Birth-Cohort Screening - (12/09/15).....over 90% listed or undergoing liver transplant were baby boomers
HCV Infection in Aging Baby Boomers is an Independent Predictor of Poor Outcome in Liver Transplant Recipients: An Analysis of the UNOS database - (12/09/15) ......chronic HCV predicts in baby boomers lower survival following liver transplant
Hepatitis C Cure Leads to a Durable Decrease in Liver Stiffness..... "cirrhosis persisted in 58%" - (12/18/15)
Regression of Advanced Fibrosis or Cirrhosis Measured by Elastography in Patients with Chronic Hepatitis C who Achieve Sustained Virologic Response after Treatment for HCV - (12/01/15) AND THIS STUDY found ONLY 55% with cirrhosis REGRESSED, 45% had NO CHANGE, a small percentage worsened. Among F3/4 at baseline 69% regressed, 17% worsened, 15% were unchanged

Program Abstract
Luca S. Belli* 1, Marina Berenguer2, Susanne-Rasoul Rockenschaub3, Silvia Martini4, Cristina Morelli5, Francesca Donato6, Riccardo Volpes7, Georges-Philippe Pageaux8, Audrey Coilly9, Stefano Fagiuoli10, Paolo Cortesi11, Christophe Duvoux12 and for the European Liver and Intestine Association (ELITA)
1Gastroenterology and Hepatology. Liver Unit, Niguarda Hospital, Milan, Italy, 2Hepatology & Liver Transplantation Unit, Hospital Universitari i Politècnic La Fe, Valencia, Spain, 3Transplantation and Surgery, University of Vienna, , Vienna, Austria, 4Liver Transplantation Center, Gastrohepatology Unit, Molinette Hospital, Turin, 5Liver and Multi-organ Transplantation, Sant'Orsola-Malpighi Hospital, Bologna, 6Digestive Diseases, 'Maggiore' University Hospital, Milan, 7Liver Transplantation, Mediterranean Institute for Transplant, Palermo, 8Hepatology, Gastroenterology, and Liver Transplantation, Centre Hospitalier Universitaire (CHU) Saint Eloi, Montpellier, Italy, 9Centre Hepato-Biliaire, Paul Brousse Hospital, Paris-Sud University, France, 10Gastroenterology and Transplant Hepatology, Papa Giovanni XXIII Hospital, Bergamo, 11Research Centre on Public Health (CESP), , University of Milan-Bicocca, Monza, Italy, 12Hepatology and Liver Transplant Unit, Henri Mondor Hospital, Henri Mondor Hospital, France
Background and Aims: Preliminary reports indicate that in Liver Transplant candidates listed for Hepatitis C virus (HCV) decompensated cirrhosis, Direct-Acting Antivirals (DAA) therapy may result in liver function improvement. Whether this clinical improvement translates into the delisting of some patients is presently unknown. Therefore, the major object of this study was to assess if and which patients can be first inactivated due to clinically improvement and subsequently delisted in a real life setting.
Methods: Retrospective multicentre European study conducted on 103 consecutive liver transplant candidates with decompensated cirrhosis without hepatocellular carcinoma treated with different DAA combinations.
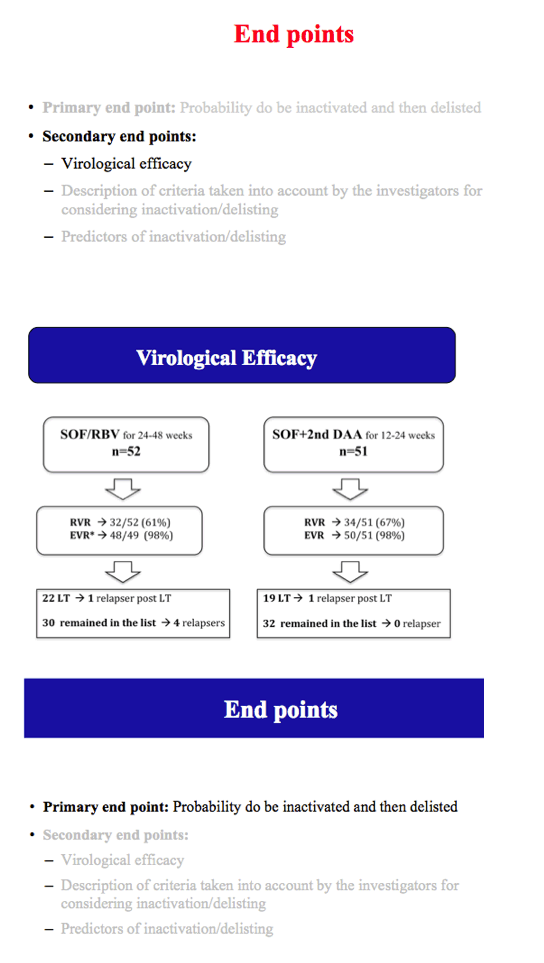
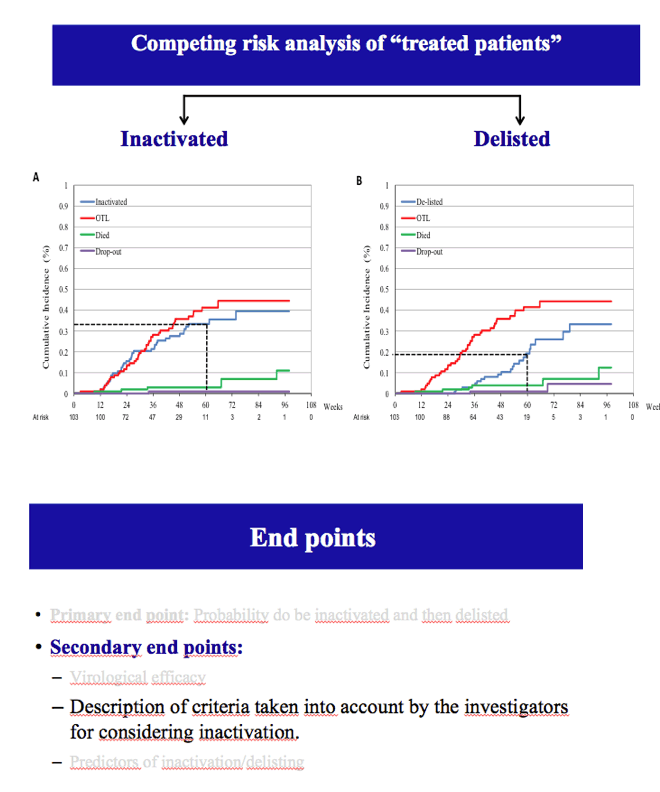
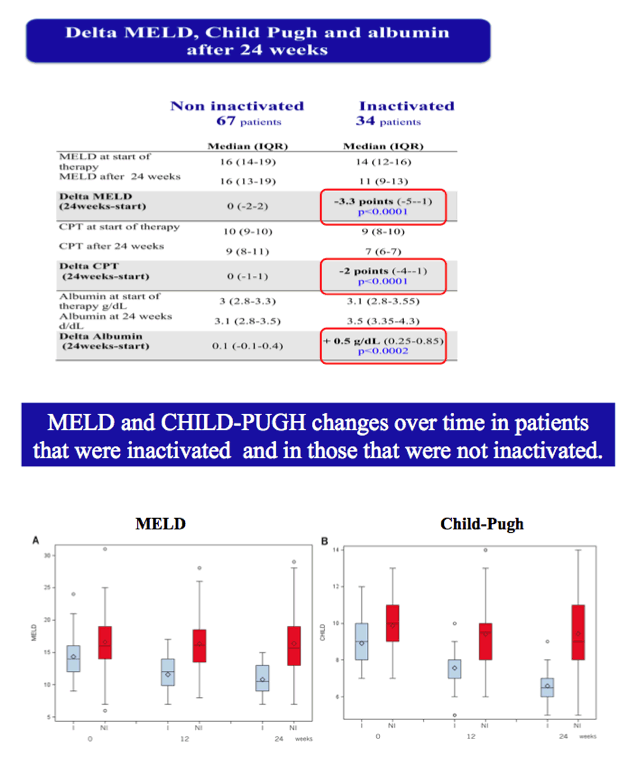
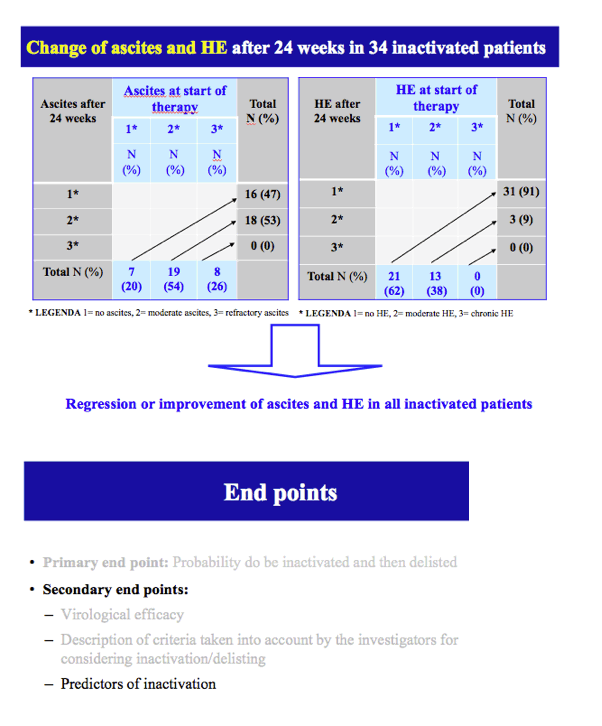
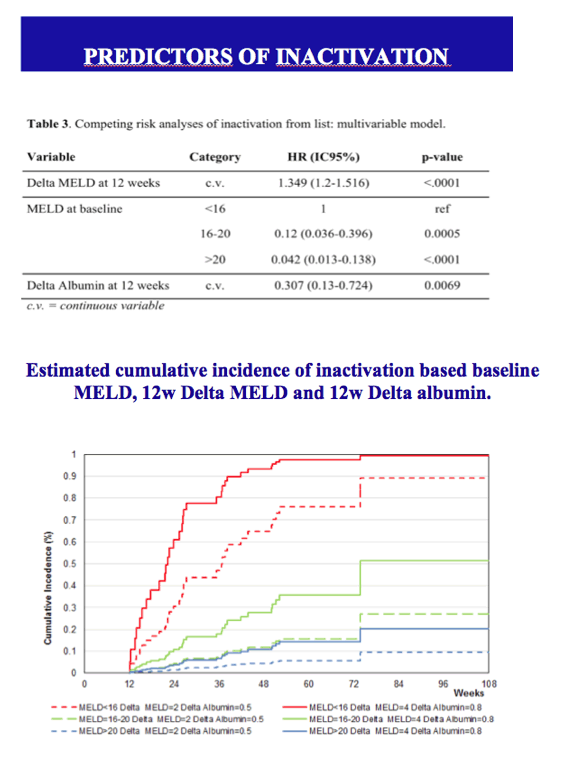
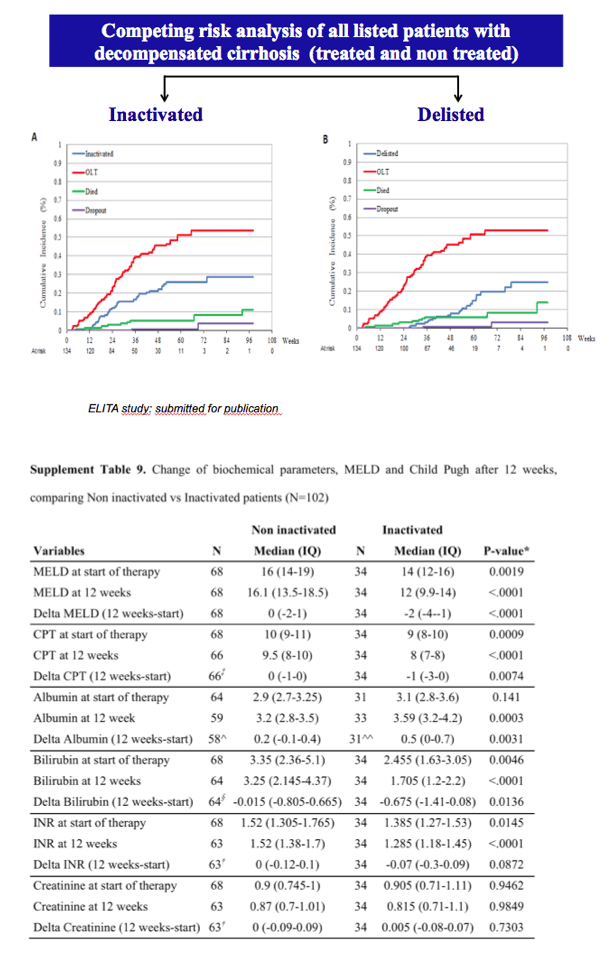
Results: The cumulative incidence of inactivated and delisted patients by competing risk analysis was 16% and 0% at 24 weeks and 35% and 20% at 48 weeks after start of DAAs. The 25 patients that were inactivated showed a median improvement of 4 points for MELD (Delta MELD, p <0.0001) and 3 points for Child-Pugh (Delta-Child-Pugh, p <0.0001). Three variables emerged from the most parsimonious Multivariate competing risk model as predictors of inactivation for clinical improvement, namely, baseline MELD (HR = 0.819; p = 0.0004), Delta-MELD (HR = 1.311; p <0.0001) and Delta-Albumin (HR = 0.419; p = 0.0041) both assessed after12 weeks of DAAs therapy.
Conclusions: In decompensated cirrhotics listed for liver transplantation, second generation DAAs favoured the inactivation and delisting of about one patient out-of-three and one patient out-of-five in 1 year, respectively. Patients with lower MELD scores have higher chances to be delisted. These results are expected to have a major impact on the management of listed patients and on organ need in programs with high prevalence of C cirrhosis.




|
| |
|
 |
 |
|
|