 |
 |
 |
| |
UK EAP - Antiviral treatment in patients with advanced HCV cirrhosis using sofosbuvir and ledipasvir/ daclatasvir, with or without ribavirin -
outcomes compared to untreated patients and long term outcomes....... ANTIVIRAL TREATMENT IN PATIENTS WITH ADVANCED HCV CIRRHOSIS USING SOFOSBUVIR AND LEDIPASVIR/ DACLATASVIR WITH OR WITHOUT RIBAVIRIN - 6 AND 12 MONTH OUTCOMES COMPARED TO UNTREATED PATIENTS
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Michelle CM Cheung1, Graham R Foster1, William L Irving2, Alex J Walker2, Benjamin E Hudson3, Suman Verma4, John McLauchlan5, David J Mutimer6, Ashley Brown7, William TH Gelson8, Douglas C MacDonald9, Kosh Agarwal4
HCV Research UK


Program abstract
ANTIVIRAL TREATMENT IN PATIENTS WITH ADVANCED HCV CIRRHOSIS USING SOFOSBUVIR AND LEDIPASVIR/ DACLATASVIR WITH OR WITHOUT RIBAVIRIN - 6 AND 12 MONTH OUTCOMES COMPARED TO UNTREATED PATIENTS
Michelle C. M. Cheung* 1, Graham R Foster1, William L Irving2, Alex J Walker3, Benjamin E Hudson4, Suman Verma5, JohnMcLauchlan6, David J Mutimer7, Ashley Brown8, William Gelson9, Douglas MacDonald10, Kosh Agarwal5 and HCV Research UK
1Blizard Institute, Barts and The London School of Medicine & Dentistry, Queen Mary University of London, London,2NIHR Nottingham Digestive Diseases Biomedical Research Unit, 3Faculty of Medicine & Health Sciences, University of Nottingham, Nottingham, 4Hepatology Department, University Hospitals Bristol NHS Trust, Bristol, 5Institute of Liver Studies, King's College London, London, 6MRC-Unviersity of Glasgow Centre for Virus Research, University of Glasgow, Glasgow, 7Centre for Liver Research, Queen Elizabeth Hospital, Birmingham, 8Department of Hepatology, St Mary's Hospital, Imperial College London, London, 9Department of Hepatology, Cambridge University Hospitals NHS Foundation Trust, Cambridge, 10UCL Institute of Liver and Digestive Health, Royal Free London NHS Foundation, London, United Kingdom
Background and Aims: Direct acting antivirals (DAAs) successfully clear hepatitis C (HCV) infection without the use of interferon, allowing treatment of patients with advanced liver disease. Since April 2014 the English Expanded Access Program (EAP) has provided DAAs for patients with advanced HCV cirrhosis, using sofosbuvir (SOF) combined with ledipasvir (LDV) or daclatasvir (DCV). Aim: to study the virological and functional outcomes up to 12 months after end of DAA therapy in advanced HCV cirrhosis, in comparison to untreated patients.
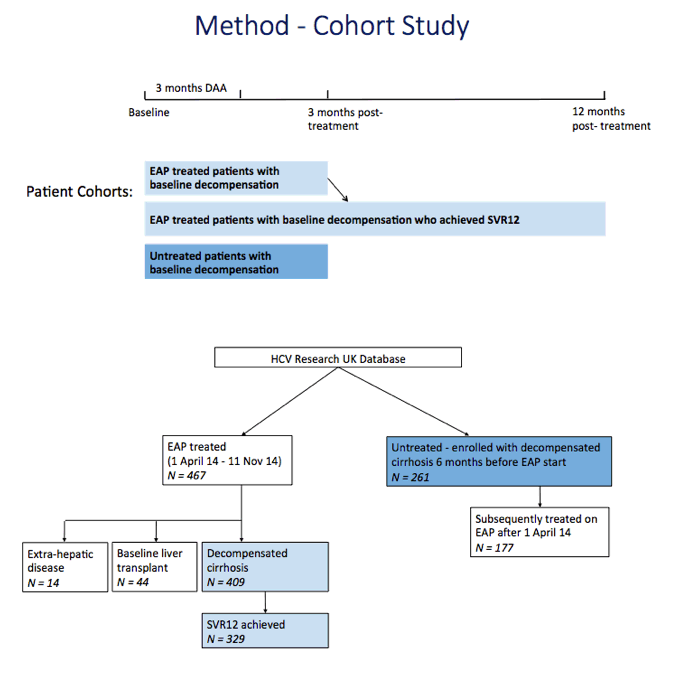
Methods: We included patients fulfilling NHS England EAP criteria, who received 12 weeks of SOF and LDV or DCV, with or without ribavirin. Patients consented to prospective data collection by HCV Research UK (HCVRUK). Two groups of patients with untreated HCV advanced cirrhosis were selected retrospectively from HCVRUK for functional outcome comparison (a) patients with decompensated cirrhosis enrolled at least 6 months prior to the initiation of the EAP, who did NOT subsequently enter EAP; (b) EAP patients for whom retrospective data were available for the period 6 months prior to EAP therapy. Outcome measures: HCV undetectable 12 weeks after treatment end (SVR12), change in MELD score and serious adverse events within 12 months.
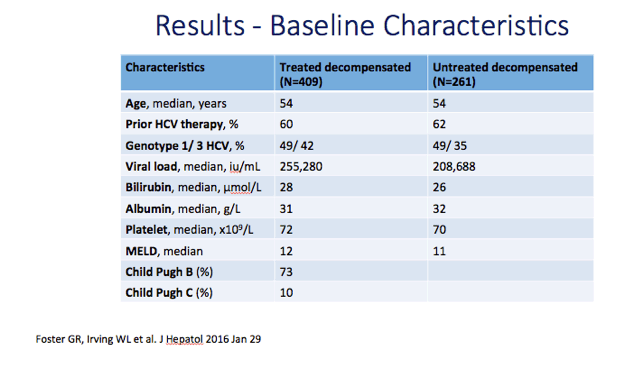
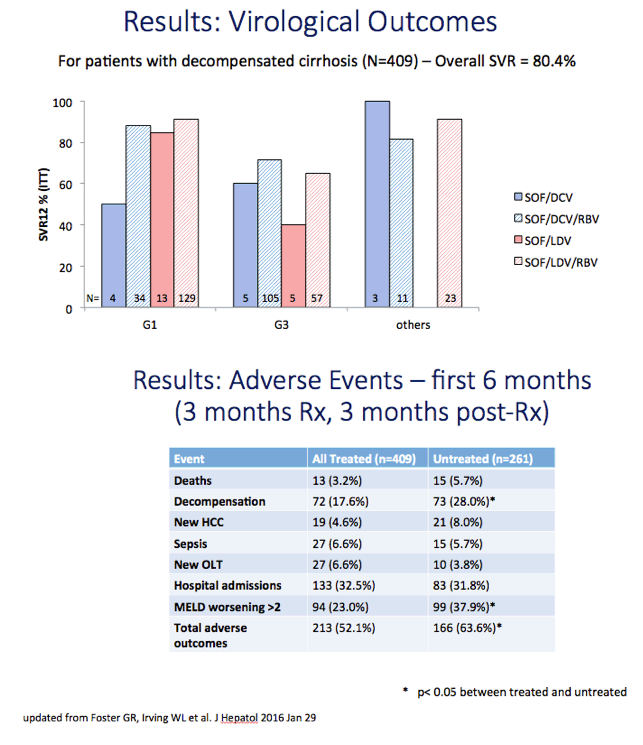
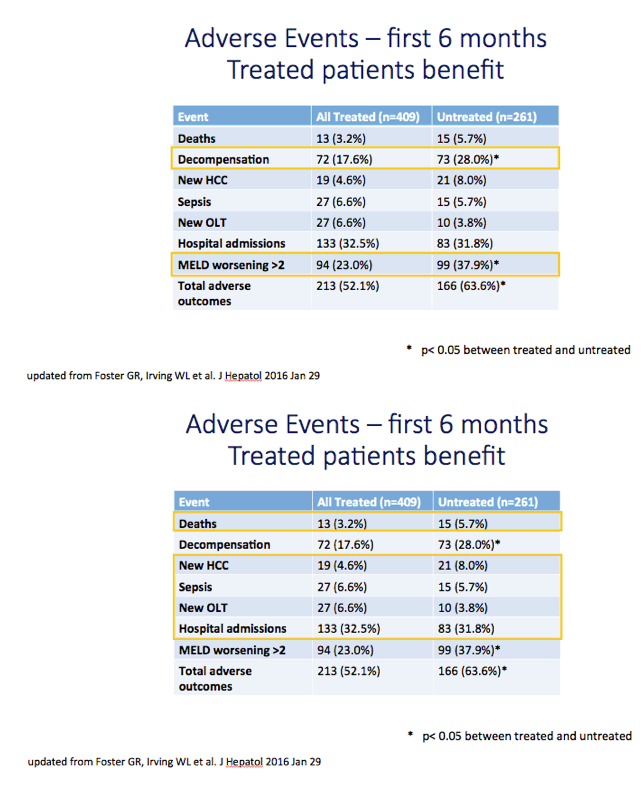
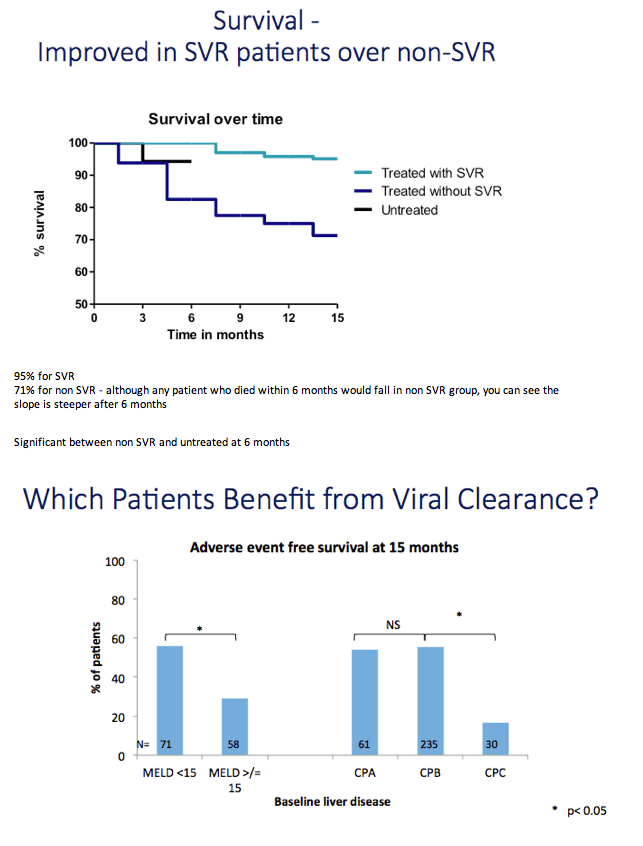
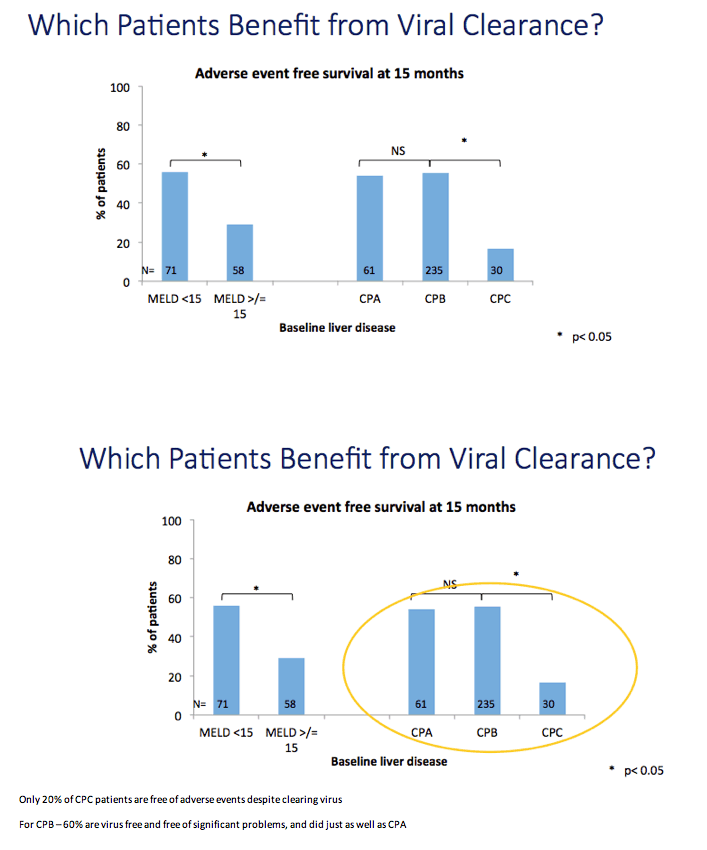
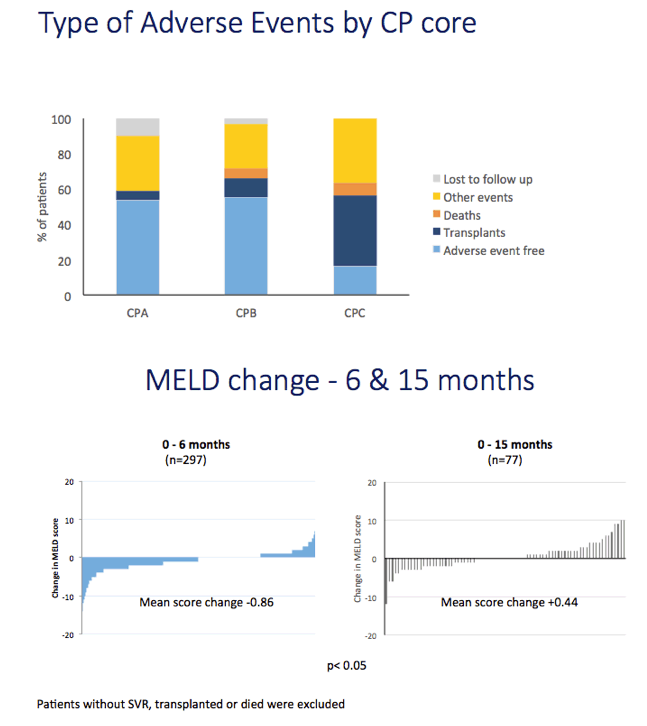
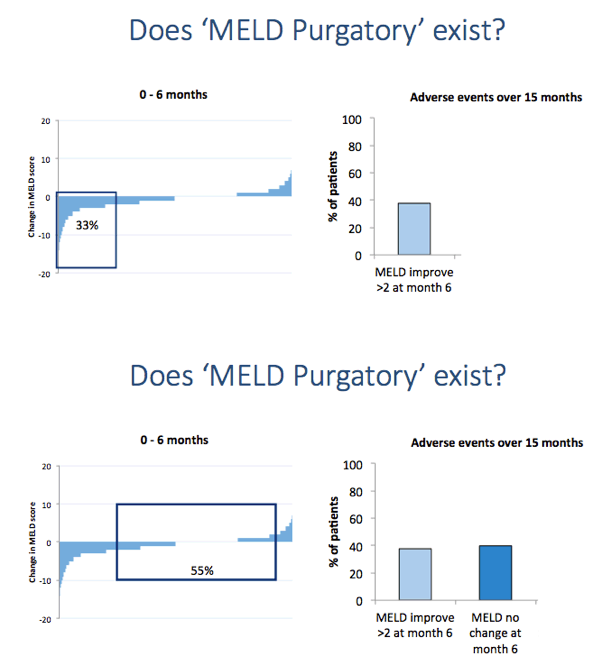
Results: We report on 467 treated patients, consisting mainly genotypes 1 (G1) and 3 (G3) HCV infections. Patients had advanced liver disease - 88% with past or current decompensation, median MELD score was 11. SVR12 was significantly higher for G1 than G3 (90 vs. 69%, p <0.0001), and in ribavirin-containing regimens. High body mass index and detectable virus at treatment week 2 were associated with lower SVR. Compared to untreated patients (n = 261) over a 6 month period, treated patients had more frequent MELD score improvements, and fewer decompensation events (18 vs. 28%, p = 0.0006). All-cause adverse outcomes (MELD increase ≥2 and any serious adverse event) were reduced (52% vs. 64%, p = 0.004) but there was no significant difference in incidence of primary liver cancer (6 vs. 8%), sepsis (7 vs. 6%), liver transplantation (4 vs. 6%) or death (6 vs. 3%).
Conclusions: High SVR rates were achieved with 12 weeks of DAAs in this large real-life patient cohort with advanced cirrhosis, who previously had limited treatment options. HCV treatment was associated with early improvements in liver function. This is clinically relevant in a population with poor prognosis. The longer term outcome of the treated patients over 12 months follow-up will be reported.




|
| |
|
 |
 |
|
|