 |
 |
 |
| |
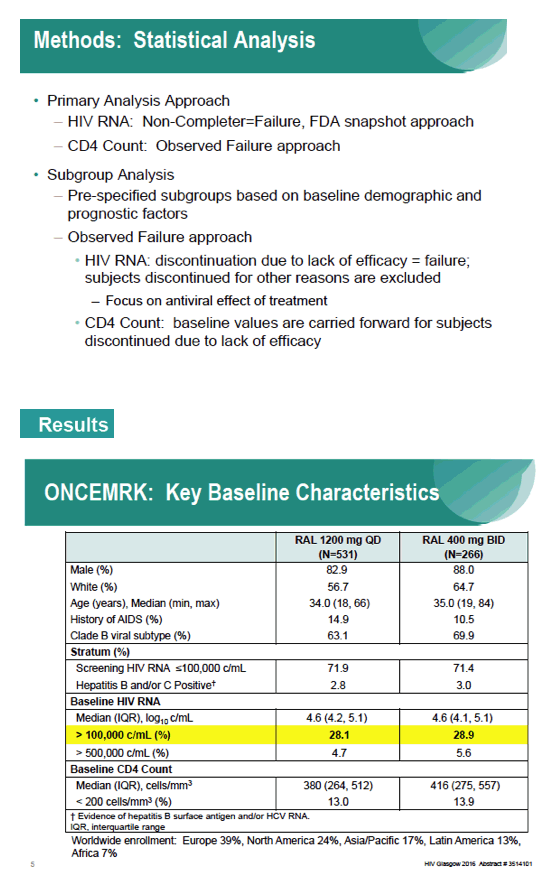
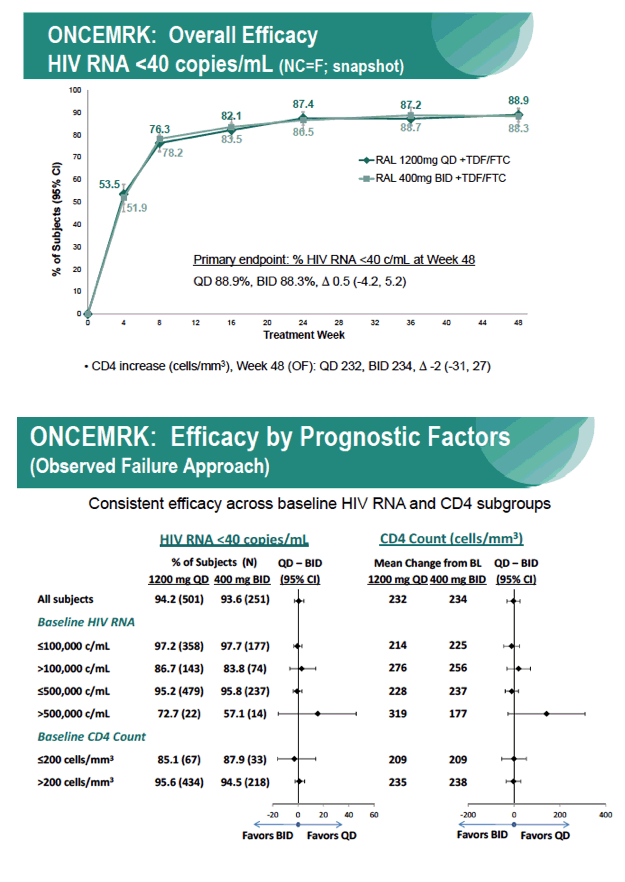
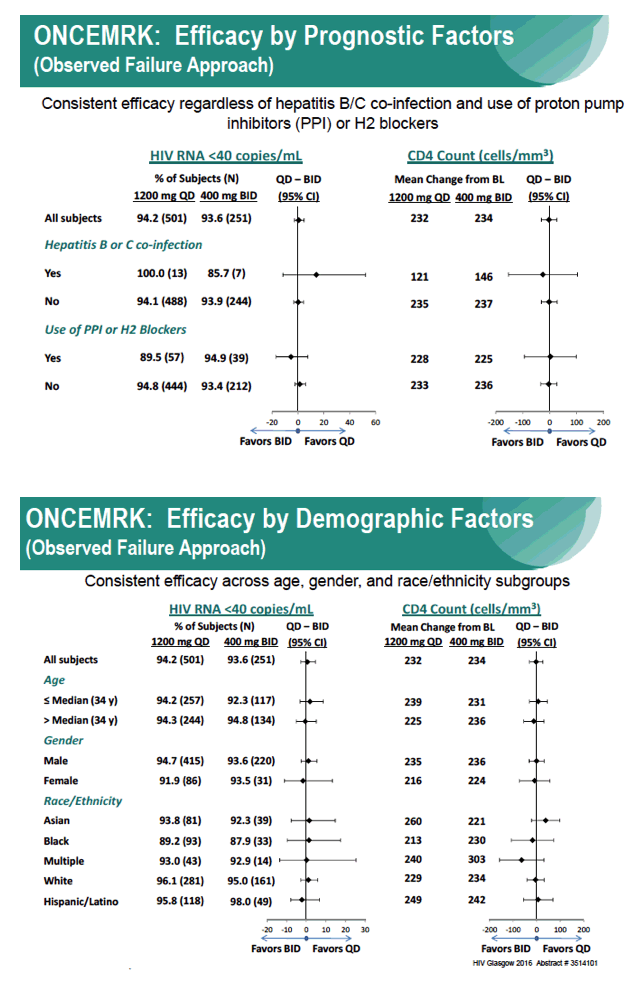
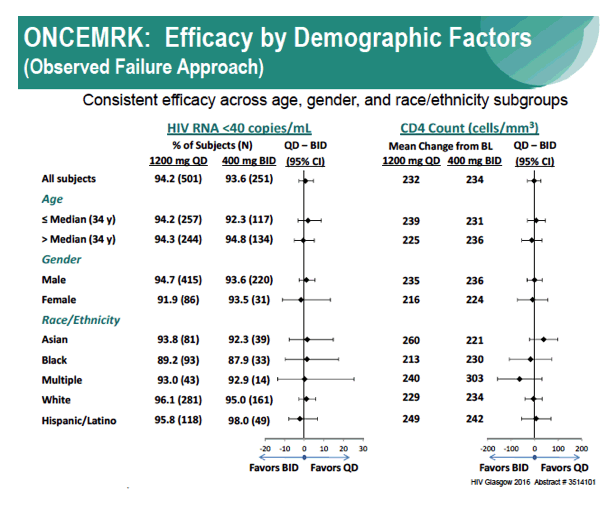
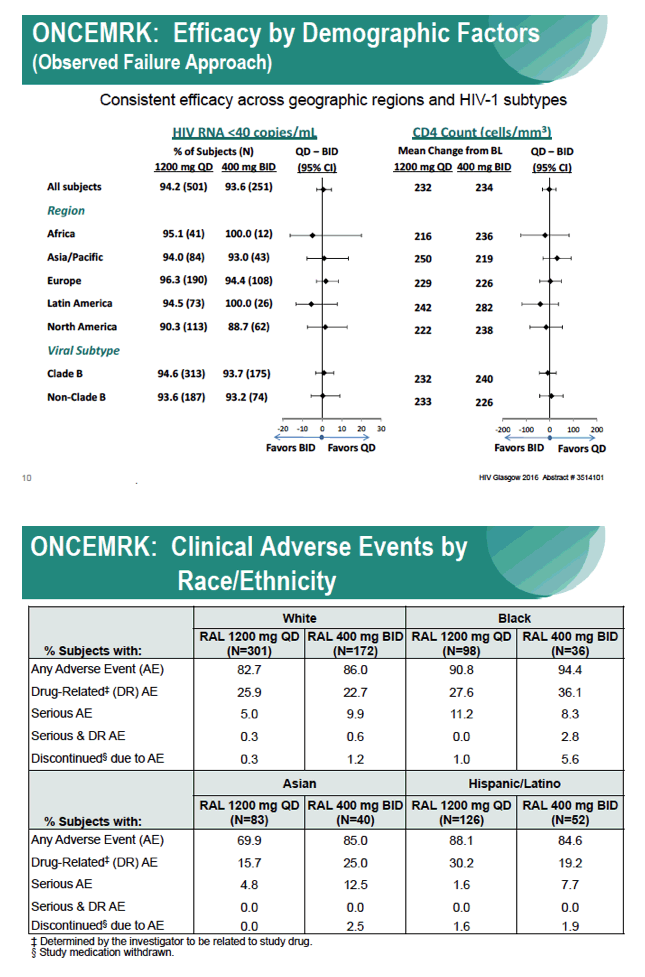
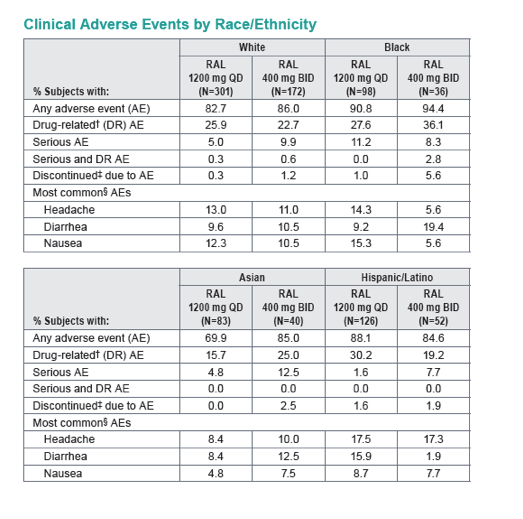
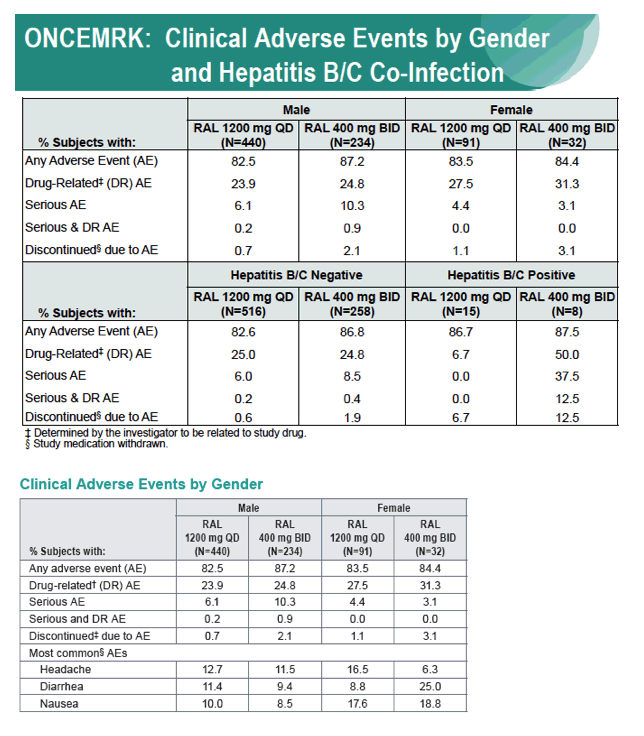
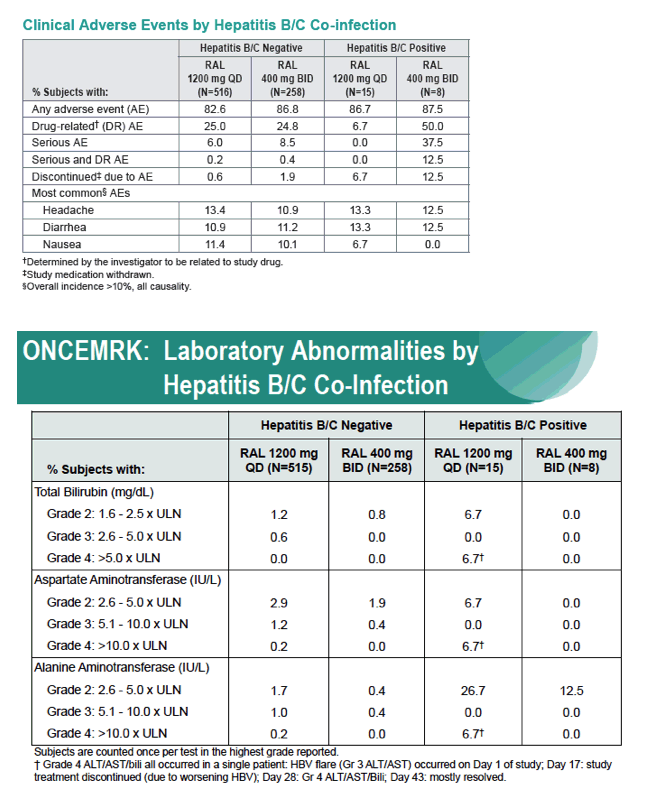
Subgroup Analyses from ONCEMRK, a Phase 3 Study of Raltegravir (RAL) 1200 mg Once Daily vs RAL 400 mg Twice Daily, in Combination with Tenofovir/Emtricitabine, in Treatment-Naïve HIV-1 Infected Subjects
|
| |
| |
Reported by Jules Levin
HIV Glasgow 2016 Oct 23-26
Pedro Cahn1, Richard Kaplan2, Paul Sax3, Kathleen Squires4, Jean-Michel Molina5, Anchalee Avihingsanon6, Winai Ratanasuwan7, Evelyn Rojas8, Mohammed Rassool9, Xia Xu10, Anthony Rodgers10, Sandy Rawlins10, Bach-Yen Nguyen10, Randi Leavitt10, and Hedy Teppler10 for the ONCEMRK Study Group
1 Fundación Huesped, Buenos Aires, Argentina; 2 Desmond Tutu HIV Foundation, Cape Town, South Africa; 3 Brigham & Women's Hospital, Harvard Medical School, Boston, MA; 4 Thomas Jefferson University, Philadelphia, PA; 5 Hôpital Saint-louis, Paris, France; 6 HIV-NAT Research Collaboration, Bangkok, Thailand; 7 Faculty Of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand; 8 Cericap Multiclinicas, Guatemala City, Guatemala; 9 University of Witwatersrand, Helen Joseph Hospital, Johannesburg, South Africa; 10 Merck & Co., Inc., Kenilworth NJ, USA
IAC/2016: Raltegravir (RAL) 1200 mg Once Daily (QD) is Non-Inferior to RAL 400 mg Twice Daily (BID), in Combination with Tenofovir/Emtricitabine, in Treatment-Naïve HIV-1 Infected Subjects: Week 48 Results - (07/29/16)



|
| |
|
 |
 |
|
|