 |
 |
 |
| |
Third ARV Does Not Affect Safety Gains in Switch From TDF/FTC to TAF/FTC - Efficacy and Safety of Emtricitabine/Tenofovir Alafenamide vs Emtricitabine/Tenofovir Disoproxil Fumarate as a Backbone for Treatment of HIV-1 Infection in Virologically Suppressed Adults: Subgroup Analysis by 3rd Agent
|
| |
| |
HIV Drug Therapy, Glasgow 2016
Mark Mascolini
Kidney and bone markers improved and HIV remained in control after a randomized switch from tenofovir disoproxil fumarate (TDF) to tenofovir alafenamide (TAF)--both coformulated with emtricitabine (FTC)--regardless of the type of third antiretroviral (ARV) in the regimen [1].
Comparisons of TAF and TDF usually involved coformulations of those drugs with elvitegravir, cobicistat, and FTC. But TAF/FTC is licensed as a two-drug coformulation that can be combined with other antiretrovirals. This randomized, double-blind, double-dummy trial (NCT02121795) aimed to compare the efficacy and safety of TAF/FTC and TDF/FTC in people taking an array of third antiretrovirals.
The study enrolled people with a viral load below 50 copies and an estimated glomerular filtration rate (eGFR) at or above 50 mL/min while taking TDF/FTC with a boosted protease inhibitor (PI) or an unboosted third agent (an integrase inhibitor, nonnucleoside, or maraviroc). Researchers randomized participants to continue TDF/FTC or switch to TAF/FTC (10/200 mg with a boosted PI, 25/200 mg with other agents).
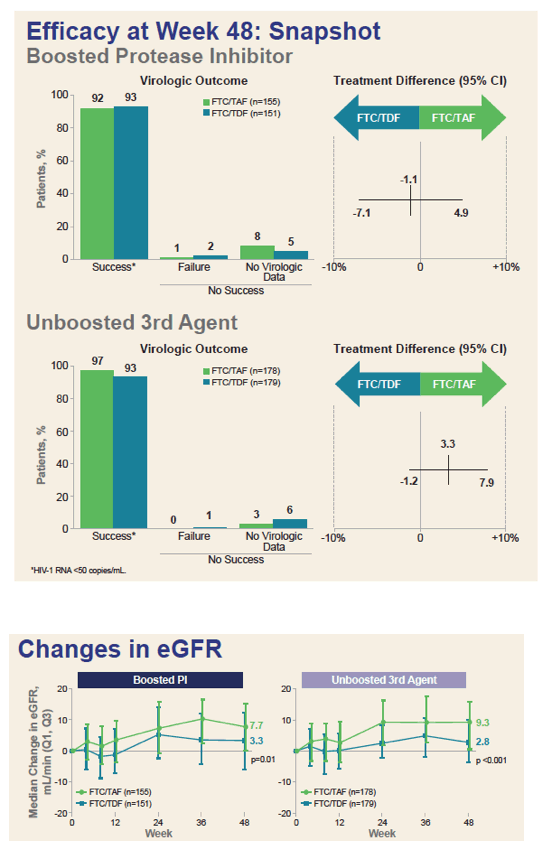
Participants had taken TDF/FTC for a median of 5.1 years. About 85% of the 333 people randomized to TAF and the 330 randomized to continue TDF were men. Respective median ages were 48 and 49, and proportions of whites 73% and 77%. About 45% of all enrollees were taking a boosted PI and the rest an unboosted agent. In a 48-week snapshot analysis of people taking a boosted PI, 92% randomized to TAF and 93% randomized to TDF had a viral load below 50 copies. Among people taking an unboosted third agent, 48-week sub-50-copy proportions were 97% with TAF and 93% with TDF. Switching to TAF/FTC was virologically noninferior to staying with TDF/FTC.
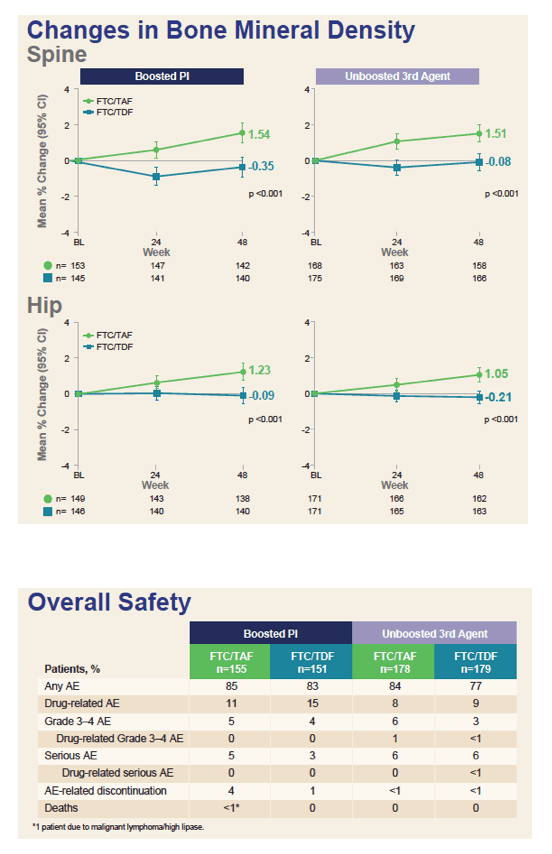
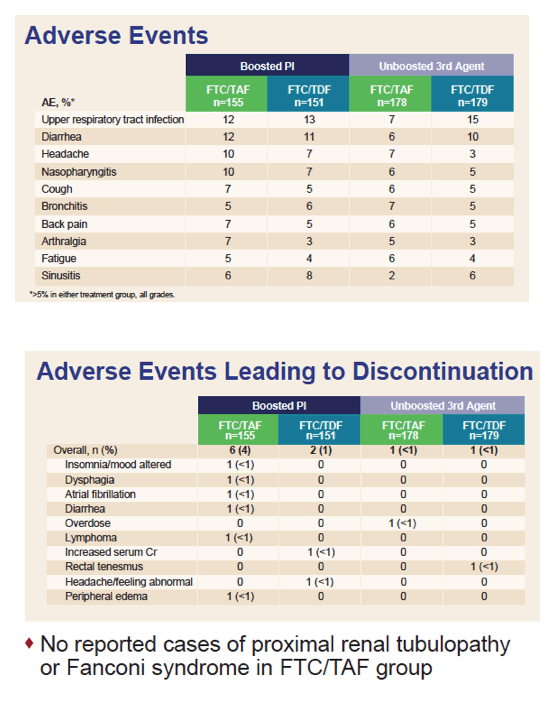
In the TAF and TDF arms, drug-related adverse events developed in 11% and 15% taking a boosted PI and in 8% and 9% taking a unboosted antiretroviral. Respective rates of discontinuation for adverse events were 4% versus 1% in the PI group and fewer than 1% in both arms of the non-PI group.
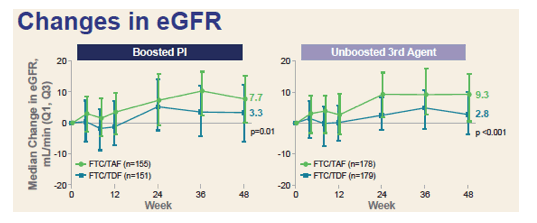
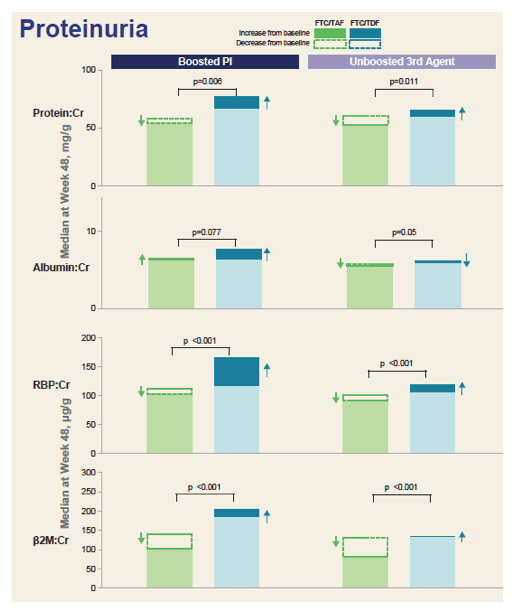
Median eGFR change through 48 weeks was significantly greater with TAF than TDF in the Boosted PI group (+7.7 versus +3.3 mL/min, P = 0.01) and in the non-PI group (+9.3 versus +2.8 mL/min, P < 0.001). Four measures of proteinuria [see graph below] were always better with TAF than TDF regardless of whether the third agent was a PI. In seven of these eight comparisons, the proteinuria difference favoring TAF was statistically significant (and nearly significant in the eighth comparison, P = 0.07). No cases of Fanconi syndrome or proximal renal tubulopathy developed.
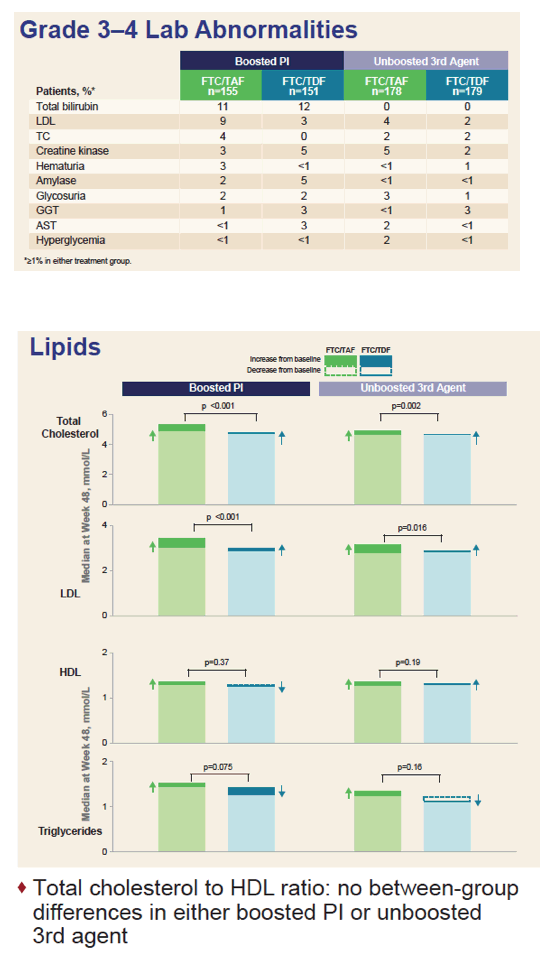
Average spine and hip bone mineral density [see graph below] improved with TAF and always dropped slightly with continued TDF, and these differences were always statistically significant in both the PI group and the non-PI group. Low-density lipoprotein cholesterol rose significantly more through 48 weeks with TAF than with TDF in both the PI group and the non-PI group, results reflecting the well-known lipid-lowering effect of TDF [2]. But total-to-HDL cholesterol ratio did not differ significantly with TAF versus TDF, regardless of the type of third agent.
The researchers believe their findings support use of TAF/FTC as a nucleoside backbone with safety benefits over TDF/FTC when taken with boosted PIs or unboosted third antiretrovirals.
References
1. Post F, Yazdanpanah Y, Schembri G, et al. Efficacy and safety of emtricitabine/tenofovir alafenamide (FTC/TAF) versus emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) as a backbone for treatment of HIV-1 infection in virologically suppressed adults: subgroup analysis by third agent. HIV Drug Therapy, Glasgow 2016. October 23-26, 2016. Abstract P093.
2. Santos JR, Saumoy M, Curran A. The lipid-lowering effect of tenofovir/emtricitabine: a randomized, crossover, double-blind, placebo-controlled trial. Clin Infect Dis. 2015;61:403-408.
--------------------
Efficacy and Safety of Emtricitabine/Tenofovir Alafenamide
vs Emtricitabine/Tenofovir Disoproxil Fumarate as a Backbone for Treatment of HIV-1 Infection in Virologically Suppressed Adults: Subgroup Analysis by 3rd Agent
Reported by Jules Levin
HIV Glasgow 2016 Oct 23-26
Frank Post,1 Yazdan Yazdanpanah,2 Gabriel Schembri,3 Adriano Lazzarin,4 Jacques Reynes,5 Franco Maggiolo,6 Mingjin Yan,7 Michael Abram,7 Cecilia Tran-Muchowski,7 Andrew Cheng,7 Martin Rhee7
1King's College Hospital, London, UK; 2Hôpital Bichat-Claude Bernard, Paris, France; 3The Hathersage Integrated Contraception, Sexual Health and HIV Service, Manchester, UK; 4Ospedale San Raffaele, Milano, Italy; 5Centre Hospitalier Universitaire de Montpellier, France; 6ASST Papa Giovanni XXIII, Bergamo, Italy; 7Gilead Sciences, Inc., Foster City, California, USA



|
| |
|
 |
 |
|
|