| |
Cost-effectiveness of Early Treatment of Hepatitis C Virus Genotype 1 by Stage of Liver Fibrosis in a US Treatment-Naive Population
|
| |
| |
.......By treating all stages of fibrosis vs waiting to treat at stages F3 and F4, the percentage of averted cases of liver transplant, decompensated cirrhosis, hepatocellular carcinoma, and liver death are 26%, 17%, 27%, and 25%, respectively........cost of treating 50% of patients with HCV genotype 1 during the next 5 years could be as high as $29 billion, partly offset by $3 billion in savings in the management of chronic HCV and advanced liver disease......and treating earlier was more cost-effective
Treating hepatitis C virus (HCV) infection at early stages of fibrosis not only improves health outcomes but is cost-effective, according to a new study. "Our analysis indicates that treating hepatitis C early averts advanced liver disease (liver failure and liver cancer) and increases the number of healthy life years for patients. This makes treating patients early cost-effective compared to waiting until they have higher degrees of liver fibrosis or cirrhosis, as many insurers currently require," lead author Harinder Chahal, PharmD, MSc, assistant adjunct professor in the department of clinical pharmacy at the University of California at San Francisco, told Medical Economics. The research also discovered that earlier treatment was more cost-effective. Among patients receiving eight or 12 weeks of sofosbuvir-ledipasvir, treating all fibrosis stages compared with treating stages F3 and F4 adds 0.73 quality-adjusted-life years (QALY) and $28 899, for an incremental cost-effectiveness ratio of $39,475 per QALY gained. Treating at stage F2 (portal fibrosis with rare septa) costs $19,833 per QALY gained versus waiting until stage F3. Treating at stage F1 (portal fibrosis without septa) gained $81,165 per QALY as compared with waiting until stage F2. Treating at stage F0 gained $187,065 per QALY as compared with waiting until stage F1. The high cost of HCV treatments makes it challenging for most health systems to treat all HCV-infected patients. "Insurance companies put into place prior authorizations that require patients to progress to higher levels of liver disease, thus effectively limiting access to a cure with new drugs. Payers may think that treating at severe fibrosis or cirrhosis is less expensive, and per our analysis, the costs are lower with later treatment initiation," Chahal said. "However, the cost-effectiveness assessment also takes into account the effects of this spending, including averted HCV disease. This results in some savings in medical costs, offsetting part of the initial expense, and improved health status, measured as QALYs.".....http://medicaleconomics.modernmedicine.com/medical-economics/news/earlier-hcv-treatment-improves-cost-effectiveness-care?page=0,1
----------------------------
at AASLD:
Regression of Advanced Fibrosis or Cirrhosis Measured by Elastography in Patients with Chronic Hepatitis C who Achieve Sustained Virologic Response after Treatment for HCV - (12/01/15)
AASLD: Hepatitis C Cure Leads to a Durable Decrease in Liver Stiffness..... "cirrhosis persisted in 58%" - (12/18/15)
The Cost of Making Hepatitis C a Rare Disease in the United States - (12/07/15)
Value of A Cure To Society: "$256.9 Bill relative to No Treatment" - (12/03/15)
The Association of Sustained Virological Response and Mortality After Interferon-based Therapy for Chronic Hepatitis C (HCV) in a Large U.S. Community-based Health Care Delivery System....SVR Reduces Mortality a Lot/Treating Early Before Cirrhosis Reduced Mortality vs in Cirrhotics - (01/13/16)
Treating Hepatitis C in the US: Measuring impact and value in the context of other major health interventions - (11/23/15)
Hepatitis C Disease Burden in the United States in 2015 and Beyond - (11/23/15)
Effect of HCV treatment on hospitalization rate: Chronic Hepatitis Cohort Study (CHeCS) - (11/19/15)
AASLD: Better Work Productivity and Activity in Patients On Ombitasvir/Paritaprevir/Ritonavirand Dasabuvir With or Without Ribavirin (RBV) in Treatment-Naïve Adults With Genotype 1 (GT1) Chronic Hepatitis C - (12/07/15)
----------------------
Cost-effectiveness of Early Treatment of Hepatitis C Virus Genotype 1 by Stage of Liver Fibrosis in a US Treatment-Naive Population - '$58 Bill to treat all' - (12/11/15)
JAMA Intern Med. Published online November 23, 2015
Harinder S. Chahal, PharmD, MSc1,2; Elliot A. Marseille, PhD2,3; Jeffrey A. Tice, MD4; Steve D. Pearson, MD, MSc5; Daniel A. Ollendorf, PhD5; Rena K. Fox, MD4; James G. Kahn, MD, MPH2,6
"We herein examined the health impact, cost, and cost-effectiveness of earlier treatment.
This analysis suggests that treatment with new HCV drugs is cost-effective when started with any evidence of fibrosis (stage F1).
By treating all stages of fibrosis vs waiting to treat at stages F3 and F4, the percentage of averted cases of liver transplant, decompensated cirrhosis, hepatocellular carcinoma, and liver death are 26%, 17%, 27%, and 25%, respectively.
For sofosbuvir-ledipasvir treatment for 8 and 12 weeks, treating all stages of fibrosis compared with treating stages F3 and F4 produced a QALY gain of 0.73 (Table) owing to a higher health state utility value after SVR in early fibrosis (69% of the QALY benefit) and to averted liver complications and death (Figure 1 and eTable 14 in the Supplement; 31% of the QALY benefit). Treating all stages of fibrosis compared with treating stages F3 and F4 increases drug costs by $33 721. An SVR lowers lifetime health care costs by about $5000, resulting in net increased costs of $28 899 for sofosbuvir-ledipasvir treatment (Table and eTable 15 in the Supplement). Treating all stages of fibrosis with sofosbuvir-ledipasvir compared with treating stages F3 and F4 only has net costs per QALY gained of $39 475 (Table).
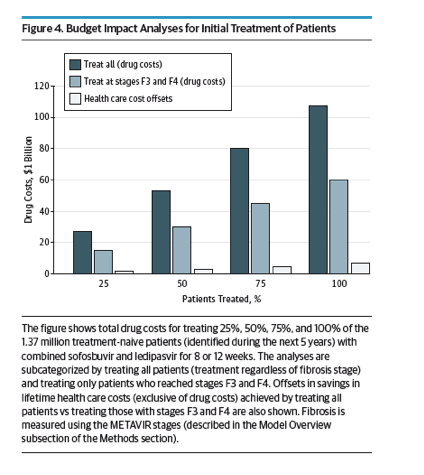
For budgetary considerations, if only 50% of eligible patients with HCV genotype 1 were to be treated with sofosbuvir-ledipasvir during the next 5 years, the cost of drugs in the United States would be $53 billion at current prices. Many payers negotiate prices, as has been seen with exclusivity deals with drug manufacturers.62- 65 If a mean 46% reduction in drug prices occurred, the cost of treating 50% of patients with HCV genotype 1 during the next 5 years could be as high as $29 billion, partly offset by $3 billion in savings in the management of chronic HCV and advanced liver disease. Figure 4 depicts the savings in lifetime health care costs exclusive of drug costs gained by treating all stages compared with treating stages F3 and F4, which is $3.3 billion with sofosbuvir-ledipasvir treatment."
|
|
| |
| |
|
|
|