| |
Evaluation of the Centers for Disease Control and Prevention Recommendations for Hepatitis C Virus Testing in an Urban Emergency Department......CDC Guidelines miss 25% with HCV
|
| |
| |
Download the PDF here
"universal 1-time testing might be more effective...CDC Guidelines miss 25% with HCV"....from jules: for years I have been telling CDC & everyone else CDC recommendations for screening overlooks big numbers
"This suggests the need to consider revision of the CDC recommendations for HCV testing in EDs and other episodic care settings. We propose expanding the age cutoff to all persons aged ≥18 years. Based on the estimated volume of HCV-infected patients, current workforce capacity for HCV care and treatment will unlikely be able to meet the increased demands associated with identifying known and previously undiagnosed HCV-infected individuals. Therefore, it is imperative to prioritize and integrate strategies for HCV testing and LTC while expanding the number of care providers with expertise in HCV care and treatment.
Seventy-five percent (n = 1814) of them would be identified through birth cohort testing and modified CDC risk-based testing together. However, 605 patients (526 with chronic infection) would be missed in an ED-based HCV testing program using current CDC testing recommendations that would be practical to implement in an ED (ie, 1945-1965 birth cohort screening and modified targeted testing based on history of IDU or known HIV positivity)."
-----------------------
Evaluation of the Centers for Disease Control and Prevention Recommendations for Hepatitis C Virus Testing in an Urban Emergency Department
Clinical Infectious Diseases May 1 2016
Yu-Hsiang Hsieh,1 Richard E. Rothman,1,2 Oliver B. Laeyendecker,3 Gabor D. Kelen,1 Ama Avornu,1 Eshan U. Patel,3 Jim Kim,1 Risha Irvin,2 David L. Thomas,2 and Thomas C. Quinn3
1Department of Emergency Medicine, 2Division of Infectious Diseases, Johns Hopkins University School of Medicine, Baltimore, and 3Division of Intramural Research, National Institute of Allergy and
Infectious Diseases, National Institutes of Health, Bethesda, Maryland
Abstract
Background. The Centers for Disease Control and Prevention (CDC) recommends 1-time hepatitis C virus (HCV) testing in the 1945-1965 birth cohort, in addition to targeted risk-based testing. Emergency departments (EDs) are key venues for HCV testing because of the population served and success in HIV screening. We determined the burden of undocumented HCV infection in our ED, providing guidance for implementation of ED-based HCV testing.
Methods. An 8-week seroprevalence study was conducted in an urban ED in 2013. All patients with excess blood collected for clinical purposes were included. Demographic and clinical information including documented HCV infection was obtained from electronic medical records. HCV antibody testing was performed on excess samples.
Results. Of 4713 patients, 652 (13.8%) were HCV antibody positive. Of these, 204 (31.3%) had undocumented HCV infection. Among patients with undocumented infections, 99 (48.5%) would have been diagnosed based on birth cohort testing, and an additional 54 (26.5%) would be identified by risk-based testing. If our ED adhered to the CDC guidelines, 51 (25.0%) patients with undocumented HCV would not have been tested. Given an estimated 7727 unique ED patients with HCV infection in a 1-year period, birth cohort plus risk-based testing would identify 1815 undocumented infections, and universal testing would identify additional 526 HCV-infected persons.
Conclusions. Birth cohort-based testing would augment identification of undocumented HCV infections in this ED 2-fold, relative to risk-based testing only. However, our data demonstrate that one-quarter of infections would remain undiagnosed if current CDC birth cohort recommendations were employed, suggesting that in high-risk urban ED settings a practice of universal 1-time testing might be more effective.
Hepatitis C virus (HCV) infection poses a significant public health threat in the United States [1]. In the noninstitutionalized civilian population, 2.2-3.2 million (0.8%-1.2%) of Americans are living with HCV, and most of them are unaware of their infection status [2]. Since 1998, the Centers for Disease Control and Prevention (CDC) has recommended risk-based testing for persons considered to be at high risk for HCV infection, including individuals with human immunodeficiency virus (HIV); history of injection drug use (IDU), hemodialysis, transfusion, transplant, or use of clotting factor concentrates; recognized exposures at work; born to HCV-infected women; or persistent abnormal alanine aminotransferase level [3]. In 2012, the CDC augmented its recommendations to include 1-time testing for HCV for all individuals born during 1945-1965, in addition to risk-based screening [4]. In 2013, the US Preventive Services Task Force also recommended 1-time screening for HCV infection in this birth cohort (B recommendation) [5].
The burden of HCV infection in patients attending emergency departments (EDs) is high because EDs serve as a medical safety net for many Americans who are also at high risk for HCV. Seroprevalence studies have demonstrated extremely high prevalence of HCV antibody positivity (13% to approximately 18%) in some urban ED populations [6-8]. An estimated 73 000 US ED visits had an HCV-related diagnosis annually during 2001-2010 [9]. In 2013, 1 urban ED in Alabama identified 7% of patients with unrecognized chronic infection in an opt-out HCV screening of patients in the 1945-1965 birth cohort [10]. Since the CDC's revised HIV testing recommendations for healthcare settings were released [11], many EDs have had great success in implementing routine HIV testing to the population they serve over the past decade. This, coupled with the availability of effective therapeutics, makes EDs a key and strategic component of the national plan to expand HCV testing. The optimal strategy for HCV testing in ED settings remains unknown.
Few EDs have evaluated the underlying burden of known and unknown HCV infections in their populations, prior to implementing an HCV testing program. Here, we sought to determine the overall burden of undocumented HCV infection and evaluate the sensitivity of implementing the CDC recommendations for 1-time HCV testing of the 1945-1965 birth cohort in an urban ED.
METHODS
The study site was at Johns Hopkins Hospital Emergency Department (JHHED), Baltimore City, Maryland. JHHED serves a diverse and socioeconomically disadvantaged population with a prevalence of former or current injection drug users of up to 15% [12]. In 1988, the overall seroprevalence of HCV antibody in JHHED was 18% [6]. The overall HIV prevalence was 7.8% in 2007 and 5.6% in 2013 [13, 14].
Study Design
We conducted an 8-week identity-unlinked seroprevalence study from June to August 2013. All ED patients aged >17 years who had excess blood specimens during the study period were included. In brief, identity-unlinked seroprevalence study methodology involves (1) collection of excess blood sample obtained as part of routine clinical care; (2) assigning of a unique study code to each patient visit and the corresponding sample; (3) processing specimens and storing the aliquoted samples in a −80°C freezer for future laboratory testing; (4) collection of basic demographic and clinical data from administrative, programmatic, or clinical datasets into a demographic/clinical dataset; (5) permanent removal of all identifiers and protected health information from the dataset; (6) laboratory testing of stored specimen after de-identification of the demographic/clinical dataset; and (7) merging of the demographic/clinical dataset with the laboratory testing results via the unique study code. The study was approved by The Johns Hopkins University School of Medicine Institutional Review Board.
Data Collection
Sociodemographic information was obtained and abstracted from administrative databases and the electronic medical record (EMR). Diagnosis and laboratory testing of HCV (eg, HCV antibody and RNA testing) were also abstracted from the EMR. The research staff received standardized training in chart review, then performed pilot data collection trials with monitoring and oversight, prior to abstracting any data. A senior study coordinator performed systematic spot-checking of data collected by the research staff. Discrepancies were adjudicated by the study principal investigator.
HIV/HCV Serologic and Viral Load Analysis
Blood samples were tested for HIV by third-generation HIV enzyme immunoassay (Genetic Systems, Bio-Rad, Redmond, Washington), and positives were confirmed by Western blot (Bio-Rad). HCV infection was determined by an HCV enzyme immunoassay (GENEDIA HCV ELISA 3.0; GreenCross Life Science Corporation, South Korea), which has a sensitivity of 99.0% and a specificity of 100% [15]. HCV RNA was quantified using the Abbott RealTime HCV Amplification Reagent Kit (No. 04J86-90, Abbott Laboratories, Des Plaines, Illinois) on 100 randomly selected samples of HCV antibody-positive patients.
Data Analysis
Patients whose samples tested HCV antibody positive were further categorized as having "undocumented HCV infection," operationally defined as the presence of HCV antibody in the absence of evidence of HCV infection anywhere in the patient's EMR. Among the criteria of the CDC risk-based testing, in most ED practices only history of IDU or known HIV infection is routinely asked at triage. Therefore, we operationally defined "modified CDC risk-based testing" to apply to all patients with either history of IDU or known HIV infection, whereas "birth cohort testing" applied to all patients born during 1945-1965, regardless of risk. The number of patients with chronic HCV infection was estimated as a proportion of all HCV antibody-positive patients, based on a prevalence of HCV RNA detected in a subset of 100 randomly selected HCV antibody-positive patients. The age of a patient at the time of his or her ED visit was collected, but birth year was not. We back-calculated and operationally defined the 1945-1965 birth cohort as those who were 47-68 years of age. Using the same back-calculation approach, "patients 18 years and older" was defined as those born before 1995, and "patients 35 years and older" as those born before 1978. Descriptive statistical analysis was performed to describe the demographics of the study population. The χ2 test was performed to determine differences in prevalence of HCV antibody or undocumented HCV infection by sociodemographic status using SAS version 9.4 (SAS Institute, Cary, North Carolina).
Ethical Approval
The Johns Hopkins University School of Medicine Institutional Review Board approved the identity-unlinked seroprevalence study.
RESULTS
During the study period, there was a total of 10 715 ED visits from 8593 unique individuals. Of those 8582 patients aged >17 years, 4713 unique patients had excess blood specimens available, and were included in this analysis. The characteristics of these 4713 patients are summarized in Table 1. There were 1793 (38.0%) patients in the 1945-1965 birth cohort. Among those patients included for analysis, significantly higher proportions were female (55.0% vs 52.8%) and older (median age, 46 years [interquartile range {IQR}, 31-58 years] vs 37 years [IQR, 26-51 years]), but lower proportions were African American (63.1% vs 68.5%) compared with those who were not included (all P values <.05).
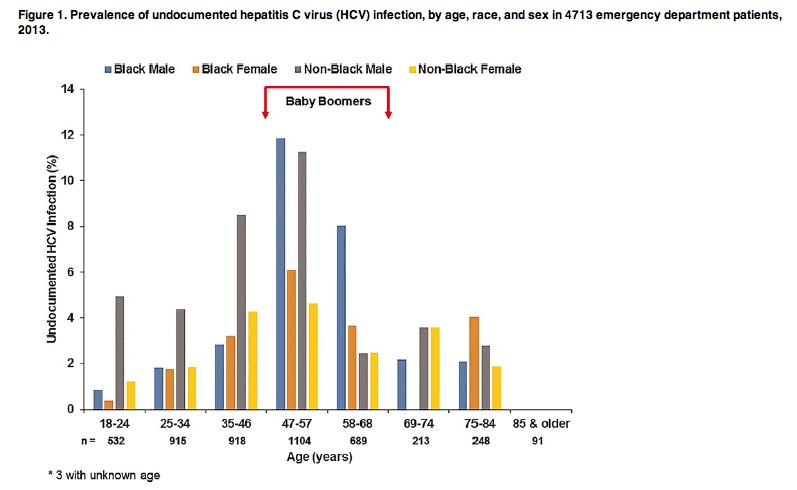
Of 4713 patients, 652 (13.8%) were HCV antibody positive, of whom 204 (31.3%) had undocumented HCV infection (ie, 4.3% of the population who had excess blood samples). When we adjusted the estimate based on differences in the age, sex, and race of the sampled vs actual ED population, we found a prevalence of HCV antibody of 9.8%. HCV seropositivity was associated with age, sex, race, HIV infection, and IDU (Table 2). The 1945-1965 birth cohort patients had a higher overall HCV prevalence compared with patients who were not born during 1945-1965 (24.8% vs 7.1%; P < .05) and a higher undocumented HCV prevalence (7.1% vs 2.6%; P < .05). The prevalence of undocumented HCV infection varied by age, sex, and race (Figure 1). Notably, the prevalence of undocumented HCV infection in nonblack men born after 1965 and nonblack women born during 1966-1978 was greater than the national prevalence of 3.25% in the 1945-1965 birth cohort. Prevalence of HCV antibody at JHHED by sex and race in 3 different birth cohorts—1945-1965 (baby boomers), 1945-1978 (≥35 years to baby boomers), and 1945-1995 (≥18 years to baby boomers)—as well as that among the 1945-1965 birth cohort of the National Health and Nutrition Examination Survey (1999-2008) is presented in Table 3. Prevalence of detectable HCV RNA in a random subset of 100 of 652 HCV antibody-positive patients was 87% (95% confidence interval [CI], 79%-93%). If we assumed there was a similar prevalence in those without blood specimens, an observed seroprevalence of 13.8% would extrapolate to 7727 unique HCV antibody-positive patients, or 6722 (95% CI, 6104-7186) unique patients with chronic HCV infection (assuming that 87% of those with antibodies had chronic infection), in our ED in 1 year among 55 936 unique ED patients.
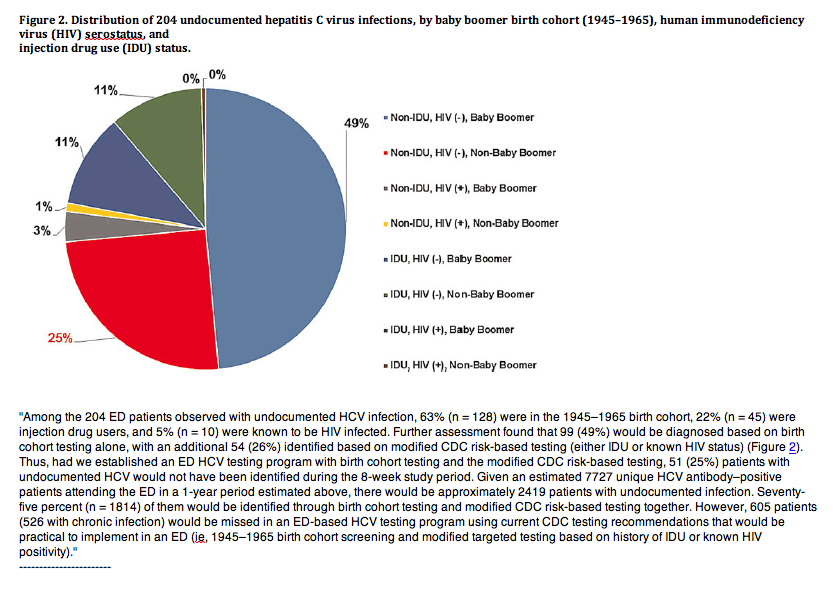
Among the 204 ED patients observed with undocumented HCV infection, 63% (n = 128) were in the 1945-1965 birth cohort, 22% (n = 45) were injection drug users, and 5% (n = 10) were known to be HIV infected. Further assessment found that 99 (49%) would be diagnosed based on birth cohort testing alone, with an additional 54 (26%) identified based on modified CDC risk-based testing (either IDU or known HIV status) (Figure 2). Thus, had we established an ED HCV testing program with birth cohort testing and the modified CDC risk-based testing, 51 (25%) patients with undocumented HCV would not have been identified during the 8-week study period. Given an estimated 7727 unique HCV antibody-positive patients attending the ED in a 1-year period estimated above, there would be approximately 2419 patients with undocumented infection. Seventy-five percent (n = 1814) of them would be identified through birth cohort testing and modified CDC risk-based testing together. However, 605 patients (526 with chronic infection) would be missed in an ED-based HCV testing program using current CDC testing recommendations that would be practical to implement in an ED (ie, 1945-1965 birth cohort screening and modified targeted testing based on history of IDU or known HIV positivity).
DISCUSSION
Similar to our previous reports, the JHHED population has a high prevalence of HCV. As expected, patients from the 1945-1965 birth cohort (ie, birth cohort testing approach) accounted for approximately half of undocumented HCV infections, followed by those who had risks due to history of IDU or HIV (ie, risk-based testing approach). Taken together, 75% of undocumented HCV infections would be identified. Notably, age, IDU history, and HIV positivity are easily asked and routinely part of our and many other EDs' triage evaluation. From an alternative perspective, alarmingly, a substantial number (25%) of undocumented infections in our ED would likely have been missed, had we implemented a routine screening approach based on these factors alone. Despite challenges with engaging patients in HCV care, our findings suggest that urban EDs should consider expanding CDC HCV testing recommendations to permit more robust identification of those patients with unknown HCV status.
The rationale for the CDC's addition of the baby boomer birth cohort testing was based on national data which found that screening persons born between 1945 and 1965 yielded optimal cost-effectiveness (which correlated with an HCV seroprevalence of 3.25%) [4]. In our ED, all demographic subgroups (black men, black women, nonblack men, nonblack women) born during 1945-1978 (ie, extending the birth cohort from 1945 to 1965, to those born as late as 1978, or age 35 years in this study) revealed a seroprevalence of HCV antibody of >3.25% (32.2%, 18.6%, and 20.0%, 8.3%, respectively), the threshold used by CDC. Further exploration found that nonblack men and nonblack women as young as 18-34 years (ie, born between 1979 and 1995) had a high HCV seroprevalence of 7.6% and 5.7%, respectively, although black men and women of the same age group had a lower HCV prevalence (2.3% and 1.6%, respectively). Thus, one logical strategy for HCV testing in our ED would be to expand birth cohort testing to those born from 1945 to 1978 (if one were to apply the same threshold used by CDC). Considering the impact and practicality of ED operations, a nontargeted HCV testing program that tests all ED patients born during 1945-1995 regardless of risks (ie, testing all adult patients born after 1945 once) might be most feasible and efficient given the availability of automatic "smart" alert systems that can be integrated into the EMR. The costs would be marginal considering that our ED already has a robust routine HIV screening program in place.
Another argument for nontargeted HCV testing extended to those born after 1965 is the recent HCV outbreak in the United States. Our findings on high HCV seroprevalence in young adult patients have become less surprising after a recent community outbreak of HIV/HCV in Indiana earlier this year [16]. This outbreak was linked with IDU of oxymorphone. There is also an increase in HCV infection associated with adolescent and young white adult injection drug users in states in central Appalachia in recent years [17]. Thus, our results confirm an alarming rate of nonblack adolescent and young adult risk trends that might put these individuals at high risk for HCV infection. When they come to ED, they might or might not disclose their IDU behavior to ED providers. Therefore, those reports and our data underscore the importance of broad screening beyond risk, and in some instances, beyond the birth cohort (1945-1965). Moreover, the observed age group-specific HCV prevalence data stratified by sex and race group demonstrate a marked epidemiologic shift in HCV infection in Baltimore City. These findings highlight an important role that EDs could serve, specifically as sentinel surveillance sites for identification of the emergence of HCV transmission in the communities they serve.
Our results also underscore the need for HCV management and treatment resources for HCV-infected individuals who frequent the ED. Based on an observed seroprevalence of 13.8% in our ED patients, we would expect approximately 8000 unique HCV antibody-positive patients, and up to approximately 6700 with chronic HCV infection (ie, assuming an 87% of those with antibodies had chronic infection) in our ED in 1 year. Because the majority of patients with chronic HCV infection are not in care [18], this represents a significant pool of individuals with newly diagnosed HCV infection who are in need of referral and linkage to care (LTC) services. Availability and reimbursement for confirmatory RNA testing as well as development of effective methods for LTC have been noted as the main barriers for ED-based HCV screening [10]. Currently, we have formed a partnership with case management in the in-house viral hepatitis clinic and have established a streamlined LTC pathway for known HCV-infected patients linking them to the viral hepatitis clinic for confirmatory RNA testing and subsequent care and treatment. Based on existing resources, many of these patient may be required to wait for their initial intake and subsequent HCV care and treatment. Current capacity for HCV care has been noted to be limited based on capacity of specialty care services (ie, infectious diseases and hepatology) across the nation [19, 20]. However, with the emergence of highly effective, safe all-oral HCV regimens, it is postulated that some HCV care may ultimately be shifted to primary care settings to meet patient care demands. Educational training for HCV management, treatment, and care in other specialties (eg, family medicine) and by primary care clinical providers might help mitigate these unmet needs and has been proposed by others as well [21-23].
Several limitations exist in our study. First, the prevalence of HCV infection is significantly associated with the local HCV epidemic, which is impacted significantly by rates of HIV coinfection, as well as local behavioral intervention programs such as needle exchange programs [24-26]. Therefore, our findings may not be generalizable to other urban EDs in the United States. Second, only ED patients who had blood drawn for their clinical procedures were included. Even though these consisted of 55% of all ED patients, the observed prevalence might not represent the true prevalence of HCV. However, our estimates were in line with the prevalence reported in our ED in the past [6] as well as those described more recently by other urban EDs [9, 27, 28], reflecting the fact that HCV is an important unmet healthcare burden, with a relatively high proportion of patients being unaware of their infection. Third, the precise number of patients with unknown HCV infection might be overestimated. This is because some patients that we coded as unaware of their infection status may in fact have known about their HCV serostatus, but this might not be reflected in the EMR. This information would not be available unless the ED providers routinely asked every patient in a standardized way about history of HCV infection. Although not 100% accurate, this paradigm faithfully reflects how a patient without HCV seropositivity might be selected for testing once an ED-based HCV testing program were implemented. Fourth, information regarding risk groups is also limited. We did not actually administer a screening tool for all risk factors, so it is possible that were CDC full risk profile information gathered, the actual percentage of patients who were missed by birth cohort and risk-based testing would be <25%. As noted, however, routinely asking all CDC risk questions at triage is not feasible, so expansion of screening to all adults may be more effective. Fifth, HCV RNA testing was not performed on all HCV-seropositive specimens. Therefore, we do not know the proportion of chronic HCV infection among all HCV-seropositive patients. Nevertheless, we can expect that the majority (up to 87%) of those HCV antibody-positive patients were chronically infected according to the estimate from HCV RNA testing in 100 randomly selected HCV-seropositive specimens.
In conclusion, we observed a high seroprevalence of undocumented HCV infection in our population, indicating that urban EDs could be a valuable venue for HCV testing. CDC-recommended birth cohort testing alone would augment risk-based identification of undocumented HCV infections 2-fold, relative to modified risk-based testing alone. However, one-quarter of infections would still remain undiagnosed applying current modified CDC recommendations (which are likely the only risk criteria that can be routinely implemented in a busy ED setting). This suggests the need to consider revision of the CDC recommendations for HCV testing in EDs and other episodic care settings. We propose expanding the age cutoff to all persons aged ≥18 years. Based on the estimated volume of HCV-infected patients, current workforce capacity for HCV care and treatment will unlikely be able to meet the increased demands associated with identifying known and previously undiagnosed HCV-infected individuals. Therefore, it is imperative to prioritize and integrate strategies for HCV testing and LTC while expanding the number of care providers with expertise in HCV care and treatment.
|
|
| |
| |
|
|
|