| |
Baseline HCV NS5A Resistance / Retreatment
|
| |
| |
------------------
HCV DAAs Resistance / New HCV Drugs - Direct antiviral combination treatment according to presence of baseline RAVs in NS3, NS5A and NS5B leads to high SVR rates in non-cirrhotic and cirrhotic genotype 1 infected patients - (05/31/16)
EASL: Resistance Analysis in 1284 Patients With Genotype 1-6 HCV Infection Treated With Sofosbuvir/Velpatasvir in the Phase 3 ASTRAL-1, ASTRAL-2, ASTRAL-3, and ASTRAL-4 Studies - (04/15/16)
EASL: HIGH EFFICACY OF ABT-493 AND ABT-530 IN HCV GENOTYPE 1-INFECTED PATIENTS WHO HAVE FAILED DIRECT-ACTING ANTIVIRAL-CONTAINING REGIMENS: THE MAGELLAN-I STUDY - (04/15/16)
EASL: High Efficacy of Sofosbuvir/Velpatasvir/GS-9857 With or Without Ribavirin for 12 Weeks in Direct-Acting Antiviral-Experienced Patients With Genotype 1 HCV Infection - (04/15/16)
EASL: High Efficacy of an 8-Week, 3-Drug Regimen of MK-3682/Grazoprevir/MK-8408 in HCV Genotype 1, 2, or 3-Infected Patients: SVR24 Data from the Phase 2 C-CREST 1 and 2 Studies - (04/19/16)
------------------------------
Retreatment
EASL: Retreatment of Patients who failed DAA-Combination Therapies: Real-World Experience from a Large Hepatitis C Resistance Database - (05/27/16)
EASL: OMBITASVIR/PARITAPREVIR/R, DASABUVIR, AND SOFOSBUVIR TREATMENT OF PATIENTS WITH HCV GENOTYPE 1-INFECTION WHO FAILED A PRIOR COURSE OF DAA THERAPY: THE QUARTZ-I STUDY....95-100% SVR - (04/29/16)
EASL: C-SWIFT RETREATMENT FINAL RESULTS: HIGHLY SUCCESSFUL RETREATMENT OF GT1-INFECTED PATIENTS WITH 12 WEEKS OF ELBASVIR/GRAZOPREVIR PLUS SOFOSBUVIR AND RIBAVIRIN AFTER FAILURE OF SHORT-DURATION ALL-ORAL THERAPY - (04/29/16)
HIGH EFFICACY OF RETREATMENT WITH LEDPASVIR AND SOFOSBUVIR IN HCV PATIENTS WHO FAILED INITIAL SHORT COURSE THERAPY WITH COMBINATION DAA REGIMENS (NIH SYNERGY TRIAL) - (04/28/15)
------------------------------
EASL: Effect of Baseline Resistance-Associated Variants on SVR With the 3D Regimen Plus RBV - (04/18/16)
CROI: HCV resistance to the daclatasvir/sofosbuvir combination across different HCV genotypes in the real life French Cohort - (03/24/16)
EASL: Prevalence and Impact of Baseline Resistance-Associated Variants (RAVs) on the Efficacy of Ledipasvir/Sofosbuvir or Simeprevir/Sofosbuvir Against GT1 HCV Infection: HCV-TARGET Interim Analysis - (04/21/16)
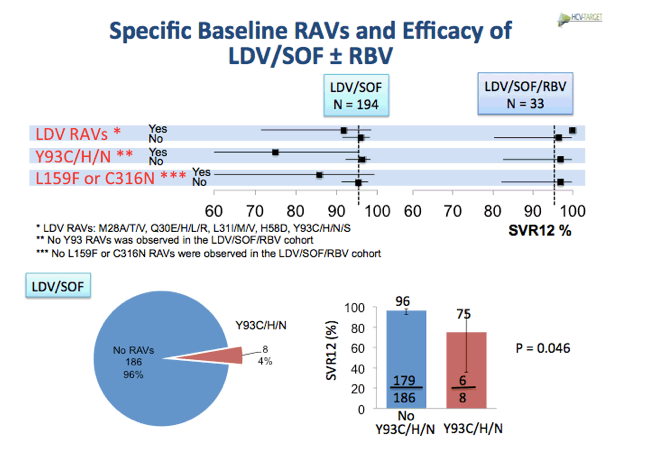
http://www.natap.org/2015/AASLD/AASLD_47.htm
Zepatier: 98% SVR in study without baseline RAVs, 100% with 16/18 weeks +RBV
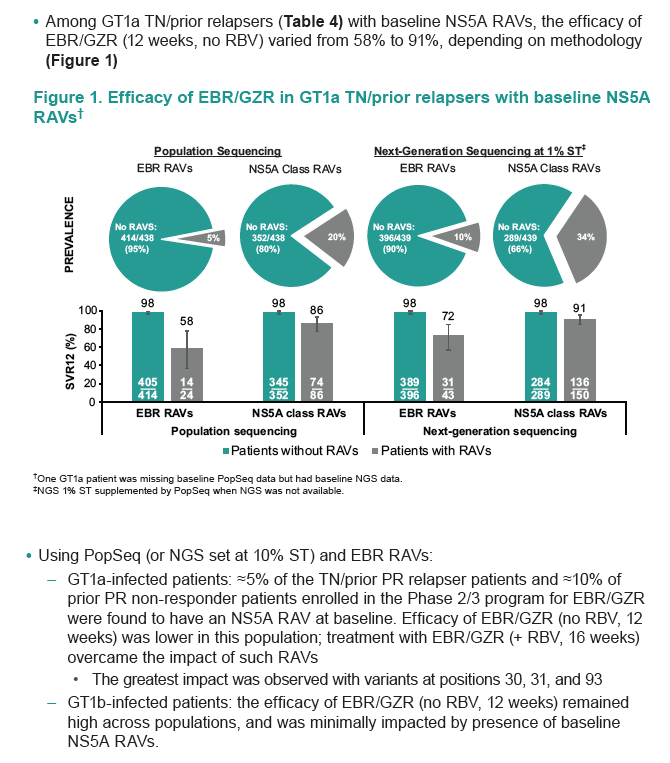
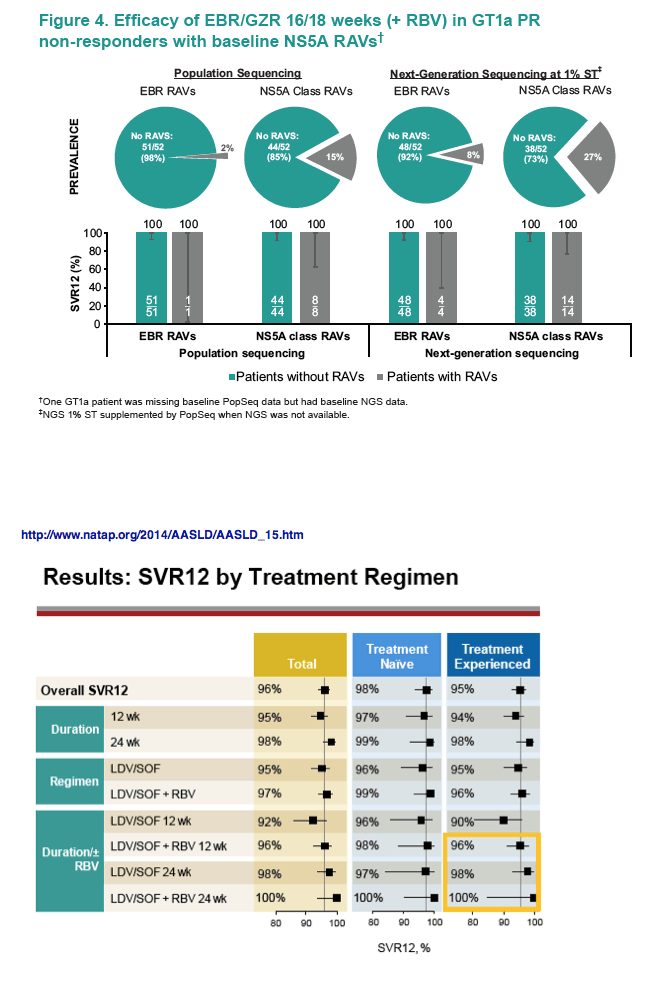
http://www.natap.org/2014/AASLD/AASLD_15.htm
|
|
| |
| |
|
|
|