 |
 |
 |
| |
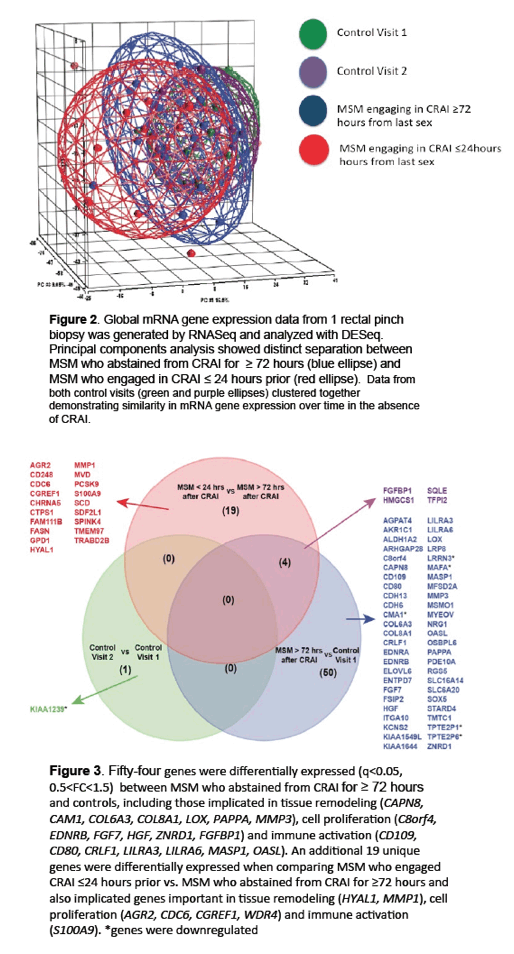
Genes Tied to Mucosal Injury/Repair in Rectum of MSM Having Receptive Anal Sex
|
| |
| |
- The Effect of Condomless Receptive Anal Intercourse on the Rectal Mucosa in MSM - The Relationship of the Rectal Microbiota, Mucosa, and Condomless Receptive Anal Intercourse in Men who Have Sex with Men
HIV Research for Prevention (HIVR4P), October 17-19, 2016, Chicago
----------------------------------------------------

⋅ The rectal mucosal microbiota of HIV negative MSM engaging in CRAI was enriched for Prevotellaceae that was associated with an increase in the relative abundance of the arachidonic acid metabolism pathway and with expression of rectal mucosal genes associated with mucosal injury/repair.
⋅ We hypothesize that mechanical microtrauma occurs during CRAI with deposition of pro-inflammatory semen resulting in transient, subclinical damage of the mucosal epithelium. This, in turn, results in an inflammatory response and exposure of the lamina propria to the gut commensal microbiota and subsequent mucosal healing. Over time, the microbiota shifts to organisms (e.g. Prevotellaceae) better able to metabolize products of mucosal injury.
⋅ The composition of the microbiota may be an important variable to consider in the design of candidate HIV vaccines particularly for MSM at-risk of HIV infection, and therapeutic manipulation of the microbiota could potentially be utilized as adjunctive therapy to optimize mucosal immune responses to candidate HIV vaccines.
⋅ Future research is needed to examine mechanisms behind the shift in the microbiota composition and whether shifts in the microbiota are associated with mucosal susceptibility to HIV.
----------------------------
at CROI 2016

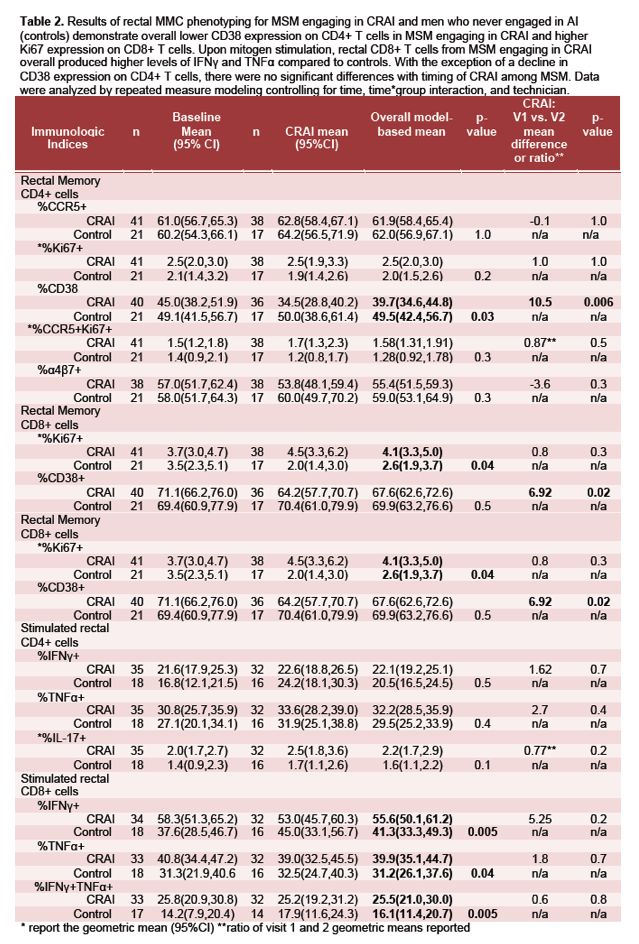
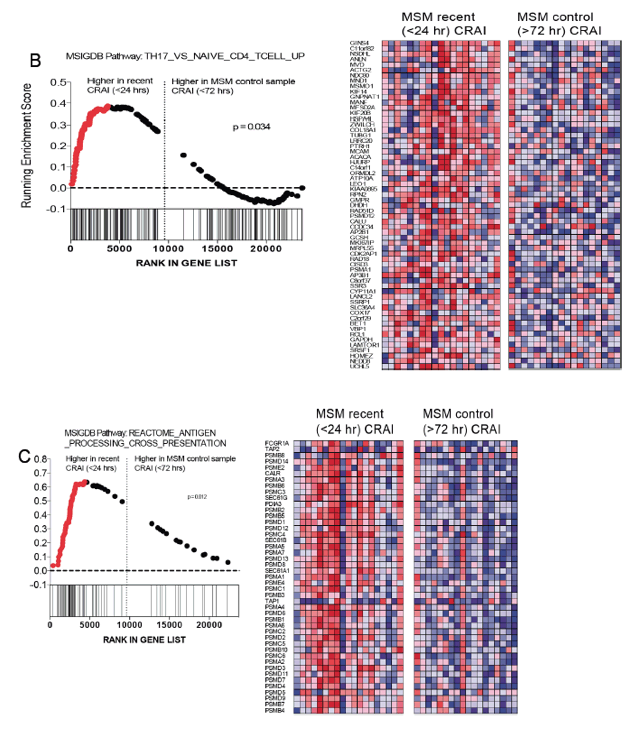
⋅ The rectal mucosa of HIV negative MSM engaging in CRAI showed a distinct mRNA gene expression and CD8+ T lymphocyte profile as compared to men who have never engaged in anal intercourse.
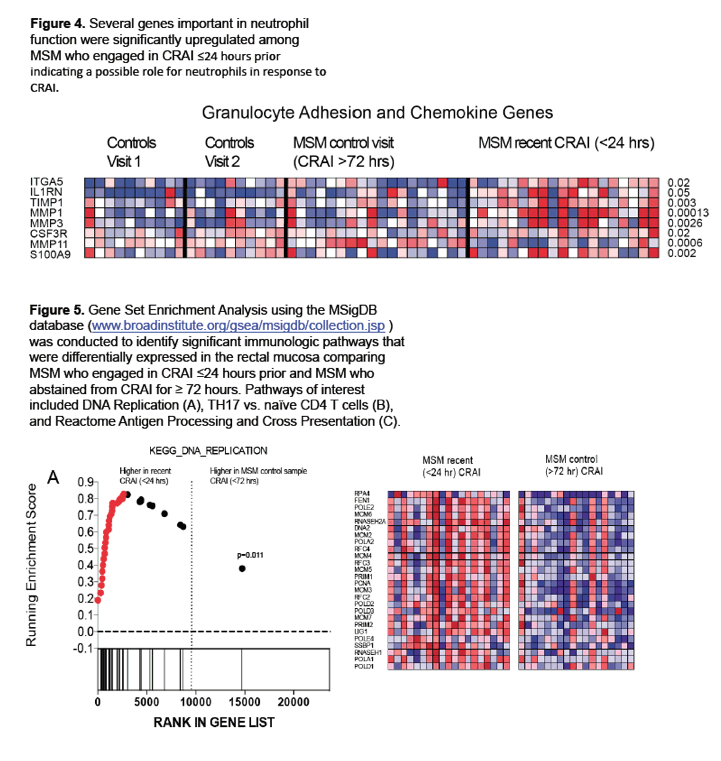
⋅ Our data show evidence of an acute inflammatory response to CRAI, possibly driven by neutrophils and monocytes, that may be the result of microtrauma/mucosal injury during intercourse with exposure to pro-inflammatory semen and the gut microbiota.
⋅ With chronic exposure to CRAI, the frequency of IFNg producing CD8+ T cells increased, suggesting a pro-inflammatory microenvironment. Th17 cells, critical for mucosal defenses and inflammation, also likely play an important role in the acute and chronic response.
⋅ Further research will be needed to determine how these factors may impact HIV susceptibility or mucosal vaccine response in the rectal mucosa of HIV negative MSM and how the gut microbiota may contribute.
-----------------------------------------------
Mark Mascolini
HIV-negative men who have sex with men (MSM) engaging in condomless receptive anal intercourse (CRAI) express rectal mucosal genes linked to mucosal injury and repair, according to results of a study comparing men who practice CRAI and men who never had anal sex [1]. Rectal microbiota of MSM reporting CRAI had a relative abundance of the arachidonic acid metabolism pathway, a signal of inflammation.
Emory University researchers who conducted this study noted that HIV transmission risk is 12-fold higher with receptive anal intercourse than with other routes of sexual transmission. But most research on mucosal HIV transmission involves the female genital tract or nonhuman primates. In contrast, little is known about the biology of rectal mucosal transmission in MSM.
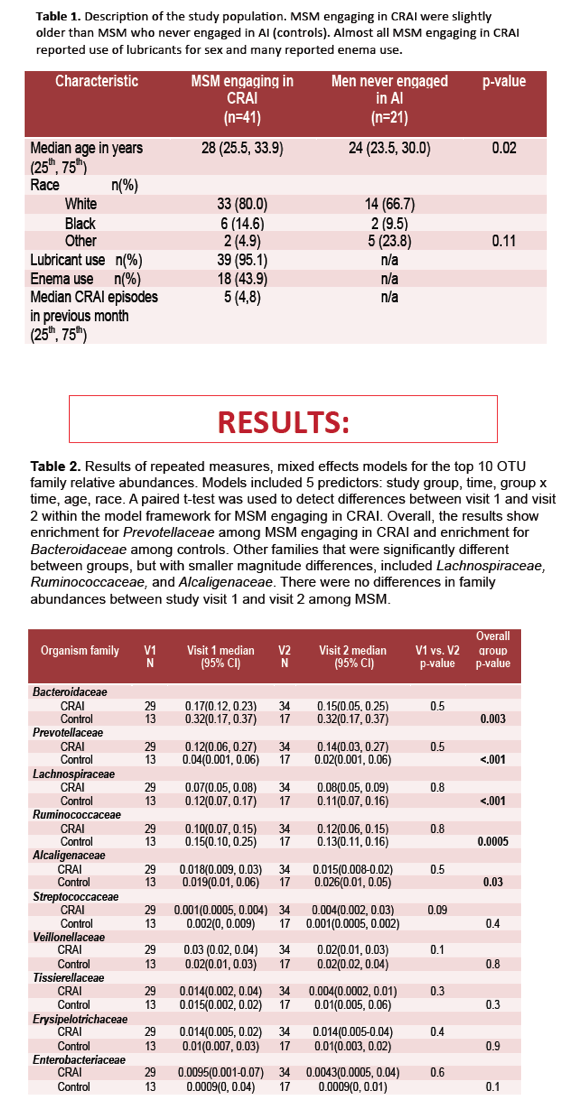
To begin filing that research gap, these investigators compared rectal mucosal microbiota in 41 HIV-negative MSM who practiced CRAI with an HIV-negative partner and 21 men who never had anal intercourse (controls). Study participants gave two rectal mucosal swab and biopsy samples separated by a median of 9 weeks. MSM did not have CRAI for 72 hours or longer before the first visit, and the CRAI group had receptive anal sex within 24 hours of the second visit. To determine relative prevalence of bacterial families, the researchers extracted and sequenced DNA from rectal mucosal swabs of 33 men in the CRAI group and 17 controls. They extracted RNA from biopsies of 18 men in the CRAI group and 10 controls and analyzed it for mRNA expression of genes involved in mucosal injury or repair (IL1RN, ITGA5, TIMP1, MMP11, CSF3R, S100A9, MMP1, MMP3, HYAL1, and CMA1).
Median age stood at 28 in MSM who practiced CRAI and 24 in men who never had anal sex (P = 0.02). Respective proportions of whites were 80% and 67%, blacks 15% and 10%, and other races 5% and 24% (P = 0.11). Almost all CRAI participants (95%) used a lubricant, and 44% used enemas. These MSM had a median of 5 CRAI encounters in the past month (interquartile range 4 to 8).
A prior report on the men involved in this study detailed evidence of an acute inflammatory response to CRAI "that may be the result of microtrauma/mucosal injury during intercourse with exposure to pro-inflammatory semen and the gut microbiota" [2]. Specifically, they found increasing frequency of interferon-gamma-producing CD8 cells with ongoing CRAI, a result "suggesting a pro-inflammatory microenvironment."
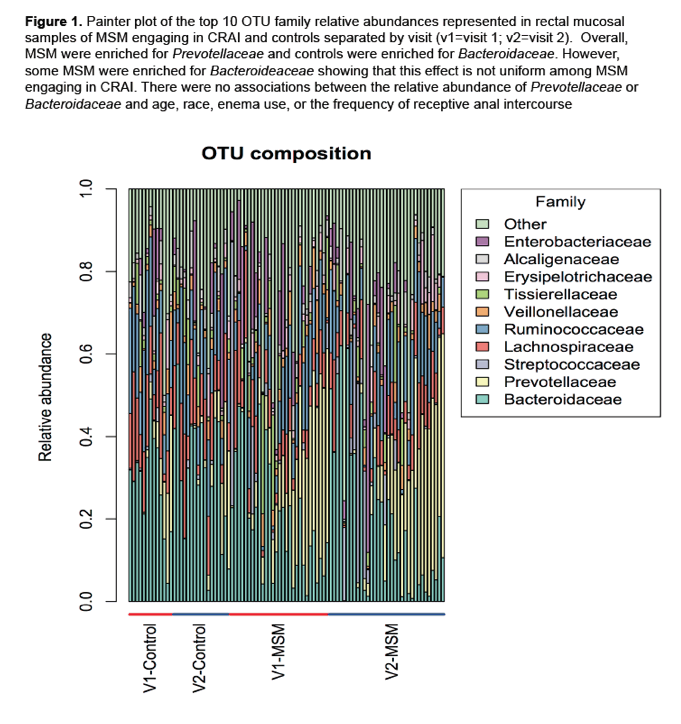
In the new analysis [1], a model adjusted for study group, time, group x time, age, and race indicated a significant enrichment of Prevotellaceae in MSM who practiced CRAI versus men who did not have anal sex (P < 0.001). Prevotellaceae are gram-negative bacteria that thrive in anaerobic conditions and break down protein and carbohydrates [3]. In contrast, control men had a significant enrichment in Bacteroidaceae, a family of anaerobic, possibly pathogenic bacteria (P = 0.003). Among MSM, abundance of these bacterial families did not differ substantially between study visit 1 and 2. Painter plot analysis of rectal mucosal samples confirmed a relative abundance of Prevotellaceae in MSM practicing CRAI and Bacteroidaceae in controls. But some men in the CRAI group had abundant Bacteroidaceae, a finding demonstrating that bacterial family enrichment is not uniform in MSM.
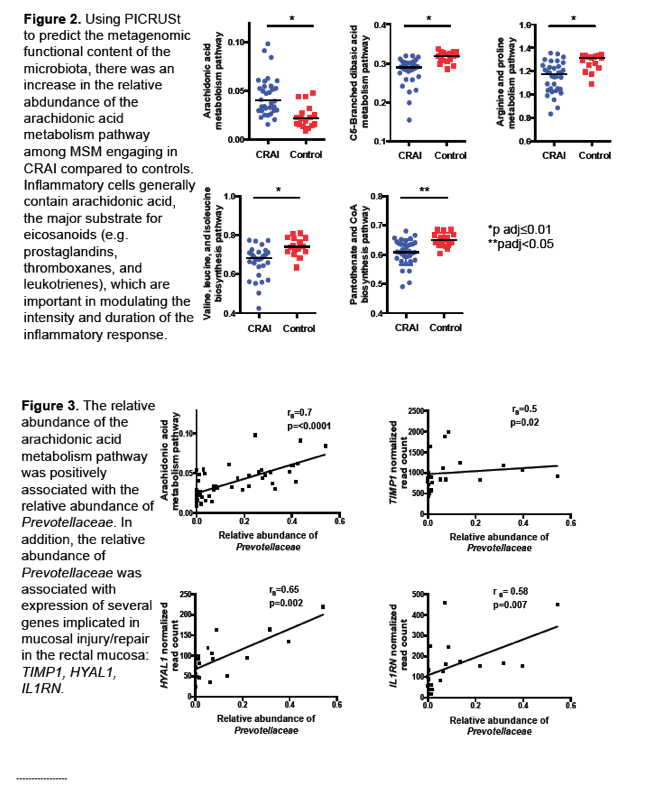
Using PICRUSt (software that predicts marker gene content), the Emory team detected a relative abundance of the arachidonic acid metabolism pathway in anal microbiota of MSM practicing CRAI compared with controls. The investigators noted that inflammatory cells usually contain arachidonic acid, which is the major substrate for eicosanoids that modulate intensity and duration of the inflammatory response.
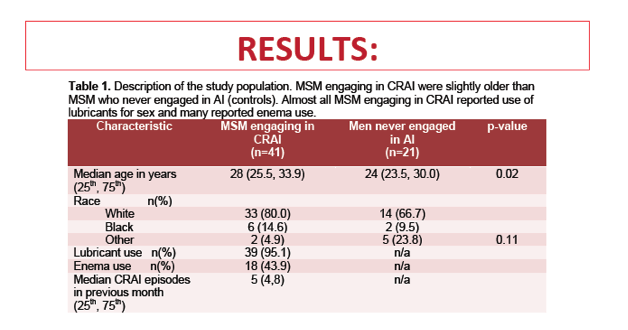
Relative abundance of the arachidonic acid metabolism pathway in CRAI practitioners was positively associated with relative abundance of Prevotellaceae (rs = 0.7, P < 0.0001). And relative abundance of Prevotellaceae was positively associated with three genes implicated in rectal mucosal injury and repair: TIMP1 (rs = 0.5, P = 0.02), HYAL1 (rs = 0.65, P = 0.002), and IL1RN (rs = 0.58, P = 0.007).
On the basis of these findings, the researchers hypothesized that tiny abrasions during condom-free receptive anal sex and exposure to inflammation-inducing semen result in "transient, subclinical damage of the mucosal epithelium." That damage ignites an inflammatory response and exposes gut lamina propria to commensal microbiota and subsequent healing of the mucosa. Over time, they proposed, rectal microbiota shift to organisms--such as Prevotellaceae--that can better metabolize products of mucosal injury. The researchers believe that rectal microbiota may be worth considering in designing HIV vaccines, especially vaccines for at-risk MSM. Therapeutically manipulating microbiota, they added, could help optimize the mucosal immune response to an HIV vaccine.
References
1. Kelley C, de Man T, Ingersoll J, et al. The relationship of condomless receptive anal intercourse, mucosal injury, and the rectal microbiota in men who have sex with men. HIV Research for Prevention (HIVR4P 2016), October 17-19, 2016, Chicago. Abstract P18.10.
2. Kelley CF, Duphare C, Lee HW, et al. The effect of condomless receptive anal intercourse on the rectal mucosa in MSM. Conference on Retroviruses and Opportunistic Infections (CROI), February 22-25, 2016, Boston. Abstract 286. http://www.croiconference.org/sites/default/files/posters-2016/286.pdf
3. Rosenberg E. The family Prevotellaceae. The Prokaryotes. Springer 2014. http://link.springer.com/referenceworkentry/10.1007%2F978-3-642-38954-2_131
----------------
2 posts below at HIVR4P & CROI




|
| |
|
 |
 |
|
|