 |
 |
 |
| |
Benefits of pre-exposure prophylaxis relative to drug resistance risk
|
| |
| |
Reported by Jules Levin
Durban IAC 2016 July 18-22
Robert Grant, Virginia Fonner, Michelle Rodolph, Teri Liegler, David Glidden, Rachel Baggaley

Abstract
Background:The risk of drug resistance (DR) with FTC/TDF or TDF pre-exposure prophylaxis (PrEP) occurs primarily in people who had acute HIV infection when starting or restarting PrEP. The risks and benefits of RNA testing before starting PrEP have not been determined.
Methods:The risk of DR in PrEP programs was compiled across 6 randomized clinical trials and one demonstration project. Resistance was measured using clinical genotypic tests.
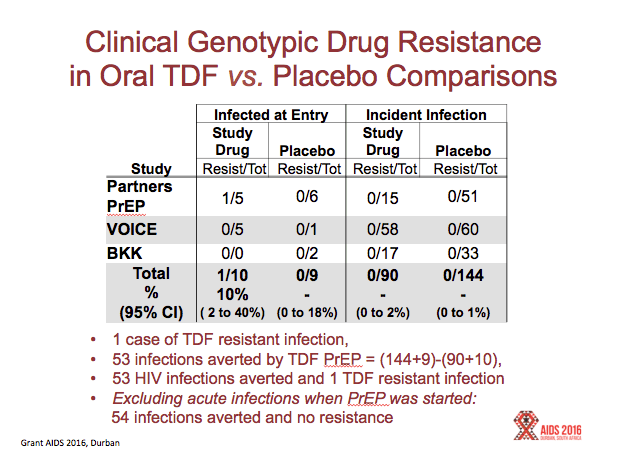
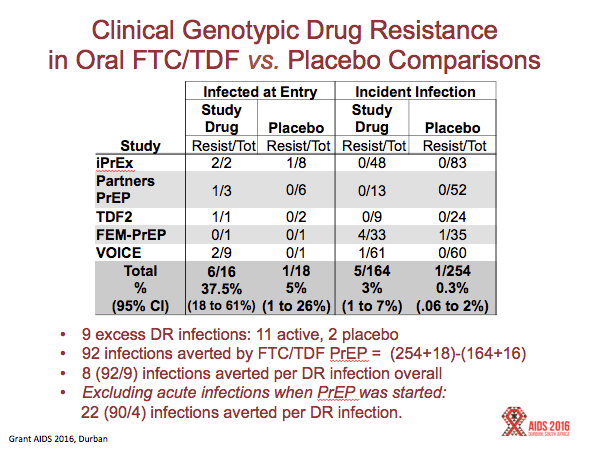
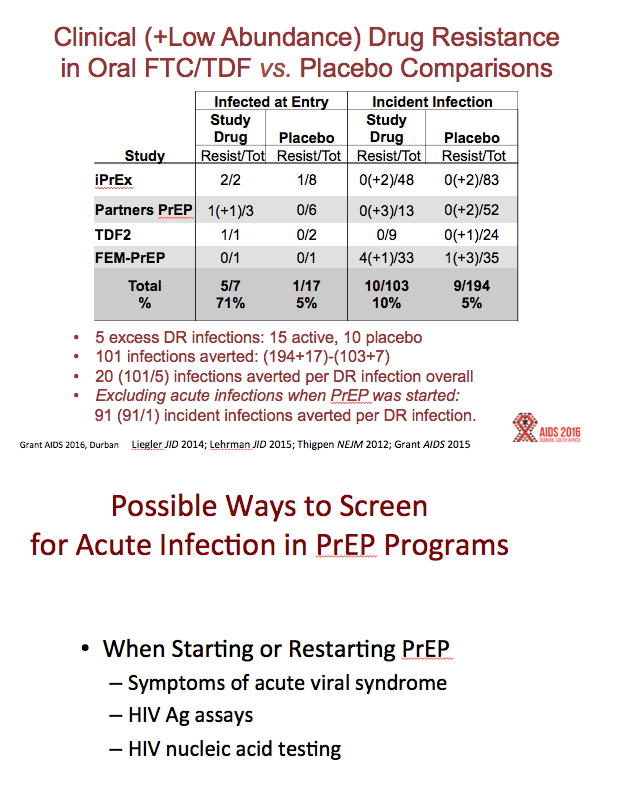
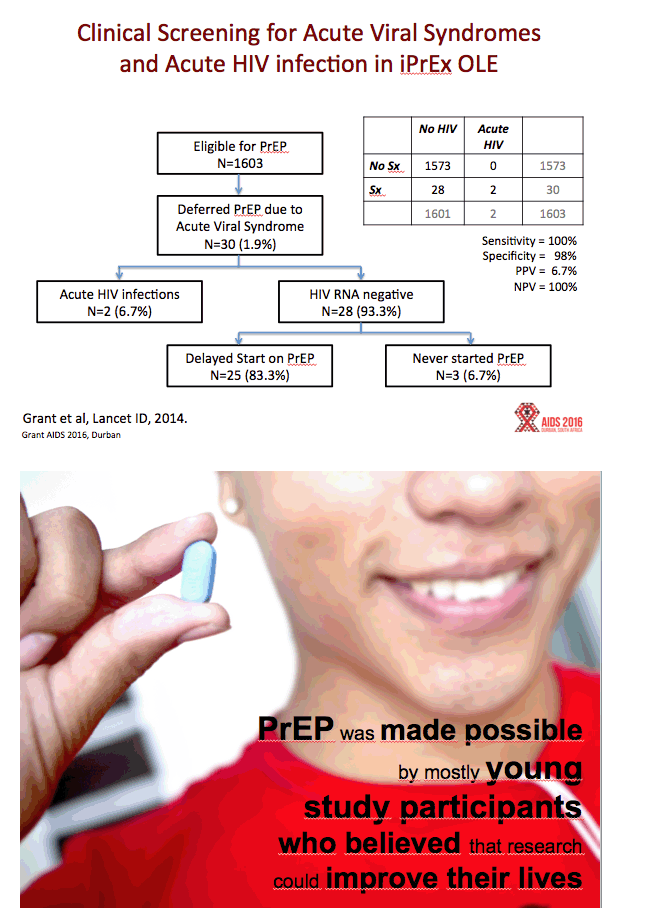
Results:All reviewed trials used rapid 2ndor 3rdgeneration antibody tests to guide PrEP initiation, and retrospectively analyzed baseline specimens for HIV RNA among seroconverters. FTC DR occurred in 10 participants who received FTC/TDF PrEP, including 33% (5/15) of participants acutely infected when starting PrEP, and in 3% (5/157) of participants with emergent infection. Including both baseline and emergent infections, there were 172 infections in the FTC/TDF arms compared with 270 among corresponding placebo controls, representing 98 infections averted and 10 (98/10) infections averted for every FTC resistant infection. Tenofovir resistance occurred in 1 participant who received TDF PrEP, including 10% (1/10) of participants acutely infected when starting PrEP, and none (of 90) with emergent infection. There were 100 infections in the TDF arms and 153 infections in the placebo controls, representing 53 infections averted by TDF PrEP and 53 (53/1) infections averted for every tenofovir resistant infection. In the demonstration project, a screen for acute viral symptoms led to deferral of PrEP among 30 of 1603 (1.9%) participants, of whom 2 (6.7%) were subsequently found to be acutely infected with HIV. Overall, the absolute risk of excess drug resistance during FTC/TDF PrEP was 0.05% (5/9222).
Conclusions:This analysis supports recent World Health Organization PrEP implementation guidance suggesting that rapid 3rdgeneration antibody tests are sufficient to minimize the overall risk of DR from PrEP. DR risk is higher with FTC/TDF PrEP compared with TDF PrEP. RNA testing before starting PrEP minimizes DR risk further while increasing costs and the risk of HIV infection due to delayed PrEP initiation.


FTC/TDF PrEP averted 98 infections in the trials (270 infections in the placebo arms and 172 in the active arms). In these same studies, there occurred 11 infections with mutations associated with resistance to TDF, FTC, or both in the active arm, and 2 infections with drug resistance in the placebo arm, yielding an excess of 8 drug resistant infections. Hence, there were 12 new infections averted for every drug resistant infection associated with FTC/TDF PrEP (98/8 = 12)
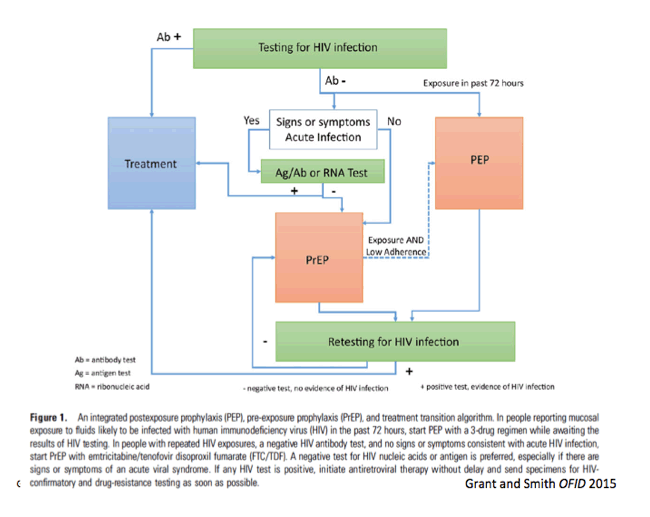
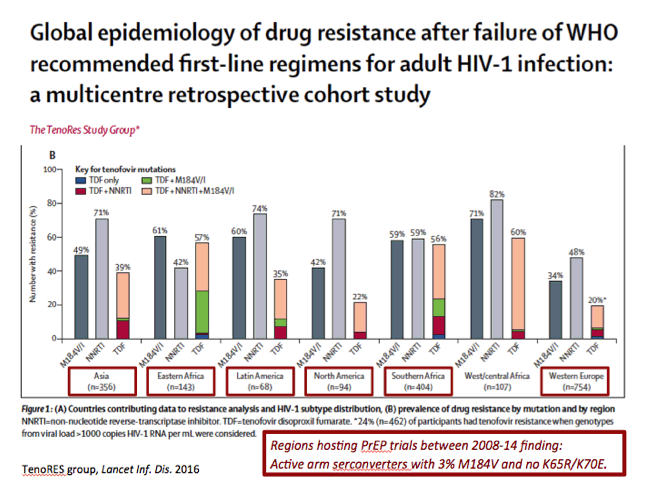
|
| |
|
 |
 |
|
|